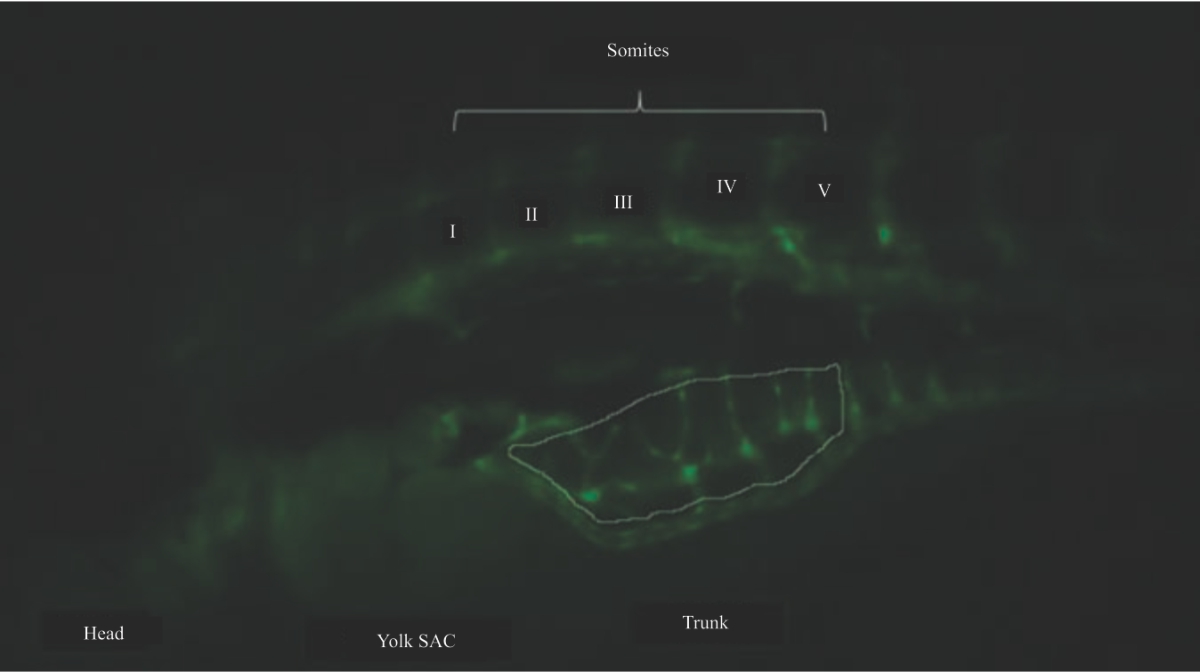
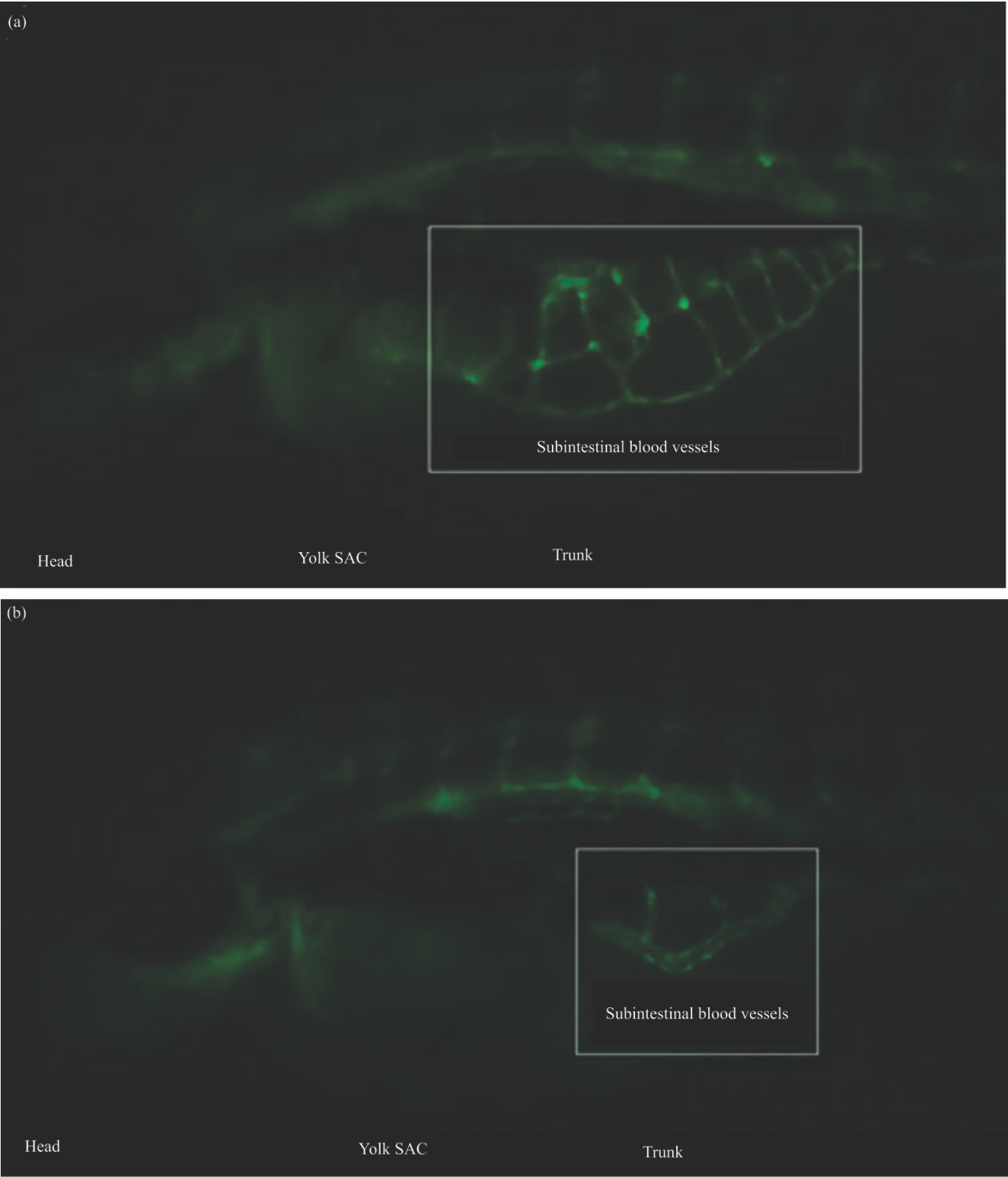
Research Article
Angiogenesis-Interfering Potential of Wound Healing Plants in Subintestinal Blood Vessels of Tg(fli1a:EGFP)y1/+Zebrafish Embryos
Department of Pharmacology, Faculty of Medical Sciences, Anton de Kom University of Suriname, Paramaribo, Kernkampweg 5-7, Paramaribo, Suriname
LiveDNA: 31.7708
Priya Magali
Department of Physiology, Faculty of Medical Sciences, Anton de Kom University of Suriname, Paramaribo, Suriname
Awinash Sardjoepersad
Department of Pharmacology, Faculty of Medical Sciences, Anton de Kom University of Suriname, Paramaribo, Kernkampweg 5-7, Paramaribo, Suriname