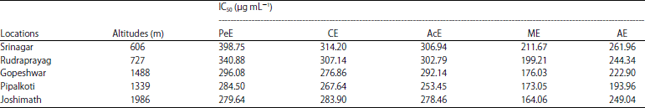
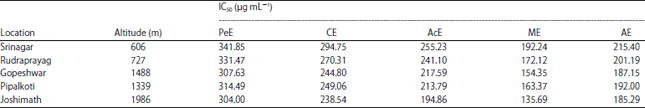
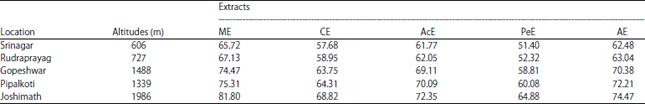
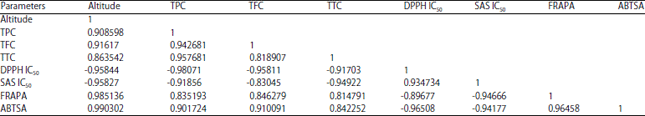
Research Article
Influence of Altitude on Secondary Metabolites and Antioxidant Activity of Coleus forskohlii Root Extracts
Department of Biotechnology, Hemvati Nandan Bahuguna (HNB) Garhwal University, Srinagar Garhwal (Uttarakhand), India
LiveDNA: 91.31895
Pooja Saklani
Department of Biotechnology, Hemvati Nandan Bahuguna (HNB) Garhwal University, Srinagar Garhwal (Uttarakhand), India
LiveDNA: 91.31909
Chandresh Chandel
Department of Biotechnology, Hemvati Nandan Bahuguna (HNB) Garhwal University, Srinagar Garhwal (Uttarakhand), India