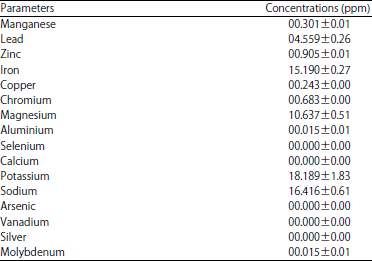
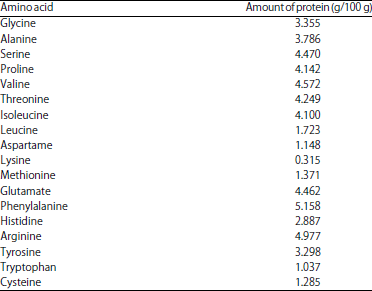
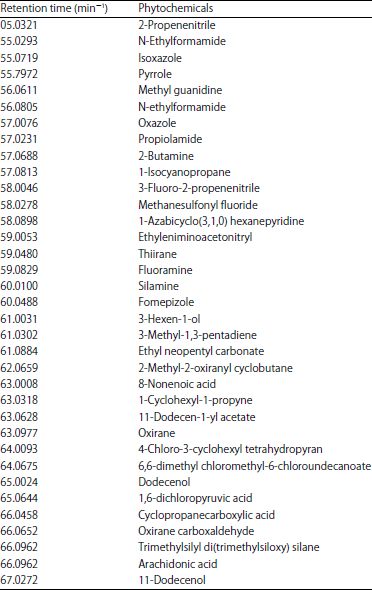
Research Article
Evaluation of the Medicinal Properties and Possible Nutrient Composition of Citrullus lanatus (Watermelon) Seeds
Department of Applied Biochemistry, Nnamdi Azikiwe University, Awka, Anambra State, Nigeria
LiveDNA: 234.30818
C.E. Oguazu
Department of Applied Biochemistry, Nnamdi Azikiwe University, Awka, Anambra State, Nigeria
LiveDNA: 234.32518
A.U. Odiakosa
Department of Applied Biochemistry, Nnamdi Azikiwe University, Awka, Anambra State, Nigeria
S.C. Okafor
Department of Applied Biochemistry, Nnamdi Azikiwe University, Awka, Anambra State, Nigeria