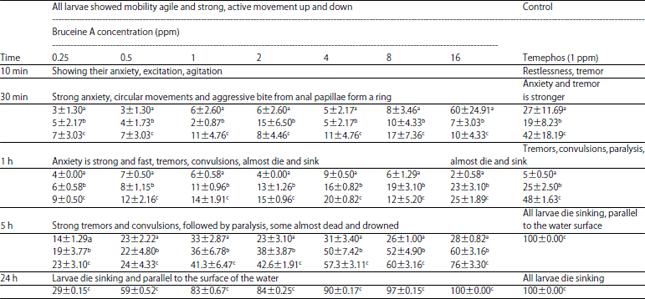
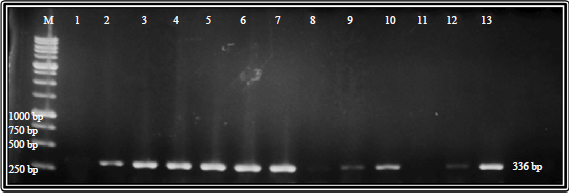
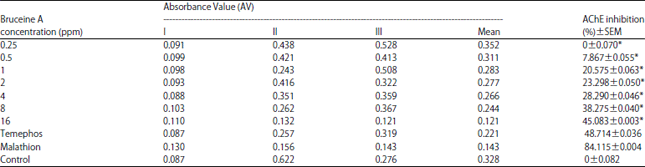
Research Article
Neurotoxic Mechanism of Bruceine A Biolarvicide Against Aedes aegypti Linnaeus Larvae
Department of Epidemiology and Tropical Disease, Faculty of Public Health, University of Diponegoro, Semarang, Indonesia
LiveDNA: 62.16151
Mustofa
Department of Pharmacology, Faculty of Medicine, University of Gadjah Mada, Yogyakarta, Indonesia
Tri Baskoro Tunggul Satoto
Department of Parasitology, Faculty of Medicine, University of Gadjah Mada, Yogyakarta, Indonesia
Edhi Martono
Departement of Plant Pest and Diseases, Faculty of Agriculture, University of Gadjah Mada, Yoyakarta, Indonesia