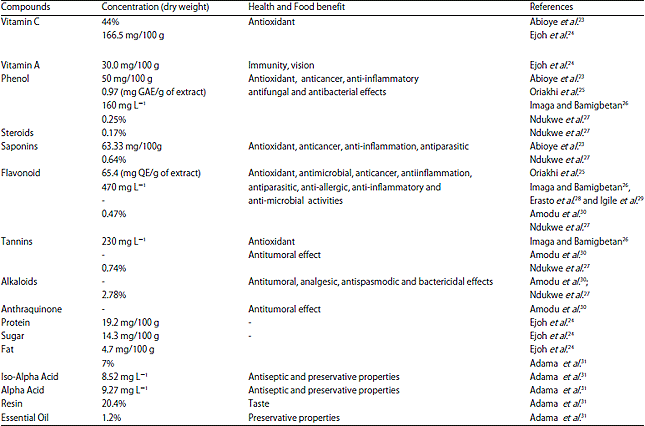
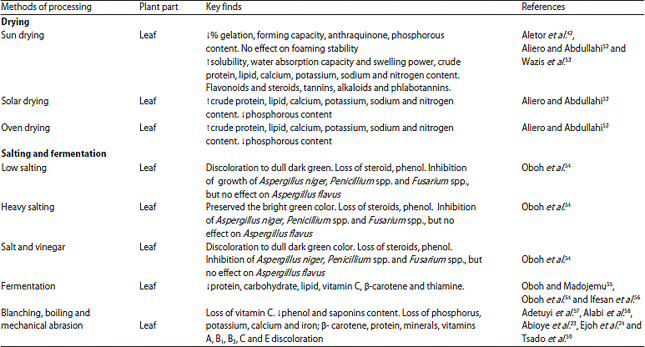
Review Article
Pre-harvest and Post-harvest Factors Affecting Bioactive Compounds From Vernonia amygdalina (Del.)
Department of Medical Biochemistry, Faculty of Basic Medical Sciences, Federal University, Ndufu-Alike, Ikwo, PMB 1010, Abakaliki, Ebonyi State, Nigeria
LiveDNA: 234.16212
Macmanus Chinenye Ndukwu
Department of Agricultural and Bioresources Engineering, Michael Okpara University of Agriculture, Umudike, PMB 7267, Umuahia, Abia State, Nigeria