Research Article
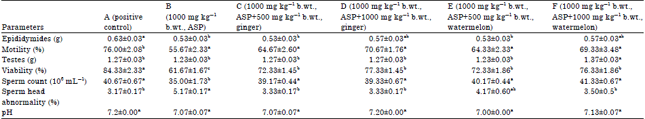
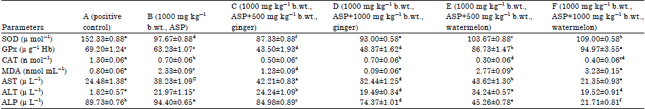
Potential of Ginger (Zingiber officinale) Rhizome and Watermelon (Citrullus lanatus) Seeds in Mitigating Aspartame-Induced Oxidative Stress in Rat Model
Department of Genetics and Biotechnology, University of Calabar, PMB 1115, Calabar, Nigeria
O.U. Udensi
Department of Genetics and Biotechnology, University of Calabar, PMB 1115, Calabar, Nigeria
LiveDNA: 234.3756
E.E. Ekerette
Department of Genetics and Biotechnology, University of Calabar, PMB 1115, Calabar, Nigeria
LiveDNA: 234.12502
U.H. Okon
Department of Genetics and Biotechnology, University of Calabar, PMB 1115, Calabar, Nigeria