ABSTRACT
Plant as a source of herbal medicine is the oldest form of medicine known to mankind. It was the mainstay of many earlier civilization and still the most widely practiced form of medicine in the world currently. The GS-MS analysis of the leaf extract of Spilanthes uliginosa (Sw) was investigated. The analysis revealed the presence of six peaks from the chromatogram which showed six phytocompounds. The major phytocompounds identified in the leaf extract are hexadecanoic acid (8.68%), hepta-9, 10, 11-trienoic acid (19.36%), octadecenoic acid (8.14%), 5- hydroxylmethyl heptadecane (14.02%), docosane aldehyde (41.72%) and 1-ethoxyoctadecane (8.08%). The presence of these assorted chemicals may be responsible for the beneficial potentials of Spilanthes uliginosa (Sw) in tradomedicine.
PDF Abstract XML References Citation
How to cite this article
URL: https://scialert.net/abstract/?doi=rjmp.2016.42.54
INTRODUCTION
Plant, source of herbal medicine is the oldest form of medicine known to mankind. It was the mainstay of many earlier civilizations and in present time, it is still the most widely practiced form of medicine in the world (Sunday et al., 2014). Plants are considered as one of the most valuable sources of food, medicine and drug for prevention of illness and maintenance of human health (WHO., 2003). In Nigeria, many indigenous plants are broadly consumed as food or home remedies especially for the treatment or management of common diseases and some of these ailments have no cure with the use of synthetic drugs (Uraku et al., 2015a). The importance of plants in medicine remains even of greater relevance with the current global shift to obtain drugs from plant sources, as a result of which attention has been given to the medicinal value of herbal remedies for safety, efficiency and economy (Uraku et al., 2015b).
The claimed therapeutic value of plants lies in their bioactive principles. The identification of these bioactive constituents of plants is crucial in unviering some essential compounds of economic values for production of multifarious chemical substances and also for finding their therapeutic agents (Achi and Ohaeri, 2015).
Spilanthes uliginosa (Sw), commonly known as Acmella uliginosa (Sw) is an annual herb or short-lived perennial which grow up to 30-60 cm high in swampy, damp sites and road sites. It is cultivated in large scale in the forest zone from Senegalto Ghana and also in Cameroun, Tanganyika and in Caribbean (Shanthi and Amudha, 2010). It is an annual weed with soft branching stems; leaves opposite with wavy margins, flowers bright yellow in terminal heads. It grows at fast rate but exhibit perennial growth pattern under favorable conditions. The flowers are borne in head inflorescence. At one glance, the shape of these flower head looks like human eyeball. Thus, this herb is colloquially known as eyeball plant. The interesting plant name is given with respect to its typical use as an age old remedy for relieving toothache (Uraku and Ogbanshi, 2015). It is a perennial herb in the tropics and sub-tropics, but it grows as an annual herb in the temperate regions. About sixty species of Spilanthes have been reported from various parts of the world including India and they have characteristic flower heads which distinguishes individual species. It originated from Africa and South American tropics but is distributed in tropics and sub-tropics of the world (Uraku and Ogbanshi, 2015). The genus occurs widely in damp pastures, at swamp margins, on rocks near the sea and as a weed of road-sides, near swage discharge areas and cultivations (Ramsewak et al., 1999). The seed germinates vigorously in about 12 days under greenhouse condition (21-32°C). Damp and cool conditions are held responsible for rotting of seeds. The plant is grown indoor or in the greenhouse, so that the seedlings become well established at the time of transplanting to the garden. Regular watering and shady conditions are essential for the proper growth of the plant genus (Sharma et al., 2010).
Spilanthes uliginosa (Sw) is a heavy feeder, preferring rich soils and an occasional side dressing of organic compost. As an ornamental, it is propagated by seed or by cuttings taken from plants in the vegetative phase stem. It is grown as a vegetable in Madagascar and Comoros and also in Réunion and Mauritius. It is sold in markets in Madagascar throughout the year with peak supplies from November-March. The leaf and buds may be harvested on an ongoing basis, as often as the plant can afford (Sharma et al., 2010).
The genus is a plant of choice for many health related disorders (Uraku and Ogbanshi, 2015). The raw leaves are used as flavouring for salads, soups and meats in Brazil and India. In central and South America, it is widely used as spice, food ingredient and as medicine (Shanthi and Amudha, 2010). The leaves were suggested to control the symptoms of various types of infections and stimulation of immunity in general. Hexanic extracts of Spilanthes acmella plants in rats is reported to induce full tonic-clonic convulsion accompanied by typical electrographic seizures in the EEG (Ratnasooriya and Peiris, 2005). Again, Spilanthes calva extracts at 2 mg per plate inhibit tobacco-induced mutagenesis (evaluated by Ames salmonella/microsome assay) by 86.4% (Rani and Murty, 2006). The flowers and leaves of Spilanthes acmella L. have a gungent taste accompanied by tingling and numbness and have been used as fork medicine for stammering, toothache, stomatities, throats complain and to stimulate flow of saliva (Sialagogue) (Ratnasooriya and Peiris, 2005).
In many countries of the world, traditional healers used the plant in treatment of many ailments such as dental and gum care (Gokhale and Bhide,1945), itching and psoriasis and tuberculosis (Peiris et al., 2001). It is one of the few herbs which exhibit antimalarial, antifungal, antiviral, immunostimulating activity, diuretic activity and the ability to dissolve urinary calculi (Tanwer et al., 2010). The plant is also used as snake bite remedy and in the treatment of articular rheumatism. In case of earache and ear infections, the immune enhancing activity of the Echinacea was enhanced by follow up with antibacterial influence of the Spilanthes tincture to cure the problem (Uraku and Ogbanshi, 2015) (Fig. 1).
This study was designed to evaluate the chemical compounds present in S. uliginosa (Sw) using GCMS analysis.
 | |
| Fig. 1: | Spilanthes uliginosa (Sw) plant |
MATERIALS AND METHODS
Collection and identification of plant material: Fresh leaves of S. uliginosa (Sw) were obtained from Ogboji-Ezzagu in Ishielu L.A.G. of Ebonyi State, Nigeria in the month of August, 2014. The plant samples were identified and authenticated by Professor Onyekwelu, S.S.C. a taxonomist in the Department of Applied Biology, Ebonyi State University, Abakaliki, Ebonyi State, Nigeria.
Preparation of plant material: The leaves of S. uliginosa (Sw) were sorted, washed thoroughly with distilled water to remove dirt and debris, cut into smaller pieces before it was shade dried for 3 weeks at room temperature (28±3°C). The dried leaves were pulverized into fine powder using electric blender (CORONA-REF. 121, Landers and Qlink blender, Model No. OBL-15L40). The powdered materials were stored in air tight polyethene bags protected from direct sunlight until required for use. The study was carried out between the months of September-November, 2014.
Plant sample extraction: Forty grams of the powdered leaves were extracted with 100 mL of 40% methanol overnight in a stopped bottle and with occasional stirring at room temperature (28±3°C). The sample was first sieved using muslin cloth and then filtered using Whatman No. 1 filter paper. This process was repeated three times. The filtrate was concentrated under reduced pressure at 40°C for 45 min in a rotary vacuum evaporator and then lyophilized to get a brown aromatic solid extract. The yield of the extract was expressed in terms of the percentage of the dry weight of initial plant material used (yield 35.37% w/w). The dry extract obtained was kept in a refrigerator at 4°C until required for use.
Column fractionation of ethanol extract: The dry crude extract was subjected to column chromatography according to standard method. The sample for the column was prepared by adsorbing 20 g of the ethanol extract of S. uliginosa (Sw) with 60 g of silica gel G (60-120 mesh). The mixture was air dried and carefully layered on top of the packed silica gel in the column (14 cm length) using a glass funnel. The extract in the column was eluted with 100 mL of methanol at the rate of 1 mL min–1. The eluates were concentrated and labeled. The percentage yield of the fraction was recorded. The methanol fraction of S. uliginosa (Sw) leaves was analyzed by Gas Chromatography-Mass Spectrometry (GC-MS).
Gas Chromatography-Mass Spectrometry (GC-MS): The GC-MS analysis was carried out on a GC-MS (Model: QP2010 PLUS Shimadzu, Japan) comprising a AOC-20i auto-sampler and chromatograph interfaced to a mass spectrometer (GC-MS). The instrument is equipped with a VF 5 ms fused silica capillary column of 30 m length, 0.25 mm diameter and 0.25 μm film thickness. The temperatures employed were, column oven temperature 80°C, injection temperature 250°C at a pressure of 108.0 kPa, with total flow and column flow of 6.20 and 1.58 mL min–1, respectively. The linear velocity was 46.3 cm sec–1 and a purge flow of 3.0 mL min–1. The GC program ion source and interface temperature were 200 and 250°C, respectively with solvent cut time of 2.50 min. The MS program starting time was 3.00 min which ended at 30.00 min with event time of 0.50 sec, scan speed of 1666 μL sec–1, scan range 40-800 μ and an injection volume of 1 μL of the plant extract (split ratio 10:1). The total running time of GC-MS was 30 min. The relative percentage of the extract was expressed as percentage with peak area normalization.
Identification of phytocompounds: Interpretation on the mass spectrum was conducted using the database of National Institute Standard and Technology (NIST) having more than 62,000 patterns. The fragmentation pattern spectra of the unknown components were compared with those of known components stored in the NIST library (NIST., 2010). The compound bioactivity prediction is based on Dr. Duke’s Phytochemical and Ethnobotanical Databases (Duke, 2014). The relative percentage amount of each phyto-component was calculated by comparing its average peak area to the total area. The name, molecular weight and structure of the components of the test materials were ascertained.
RESULTS AND DISCUSSION
The various compounds present in the methanol leaf extract of Spilanthes uliginosa (Sw) were identified by GC-MS analysis (Fig. 2). These identified compounds confirmed the ethnomedical relevance of the plants in their use for the management and treatment of diseases. The curative potentials of this plant as aclaimed by local dwellers may be due a single effects or as well as interplay of the phytocompounds present in the plant. However, the definite mode of actions of the extract’s oil has not been established but the mechanism suggested here in the study can not be discarded (Ogunwande et al., 2010, Victor and David, 2015). This observation is in line with the report of Uraku et al. (2015a) and Balamurugan et al. (2012). The active principle, area of peak concentration (%), Retention Time (RT) Molecular Weight (MW) and Molecular Formula (MF) in the methanol extract as identified through the NIST database are listed in Table 1.
The GS-MS Chromatogram of Spilanthes uliginosa (Sw) methanol leaves extract detected six peaks and this signified six compounds. The results revealed that the following compounds hexadecanoic acid (8.68%), hepta-9, 10, 11-trienoic acid (19.36%), octadecenoic acid (8.14%), 5-hydroxylmethyl heptadecane (14.02%), docosane aldehyde (41.72%) and 1-thoxyoctadecane (8.08%) were found as the major compounds in the methanol extract of leaves of S. uliginosa (Sw) plant.
| Table 1: | Compounds identified in methanol extract of Spilanthes uliginosa (Sw) leaves |
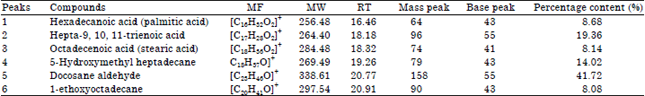 | |
| MF: Molecular formula, WW: Molecular weight, RT: Retention time | |
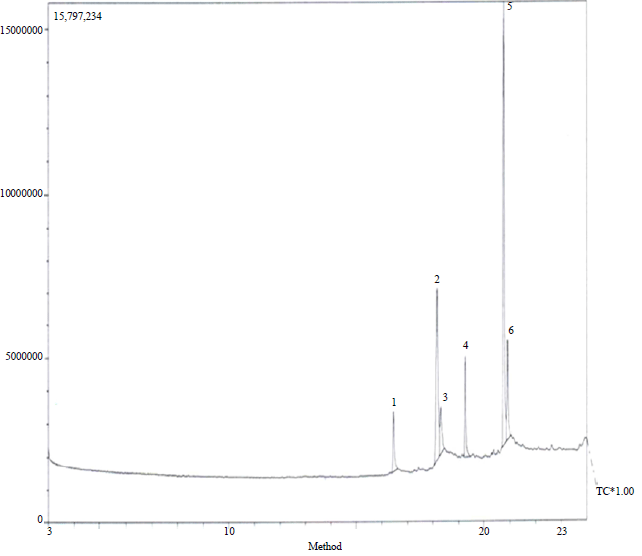 | |
| Fig. 2: | GS-MS chromatogram of the methanol extract of Spilanthes uliginosa (Sw) leaves |
| Table 2: | Activity of compounds identified from methanol extract of Spilanthes uliginosa (Sw) leaves |
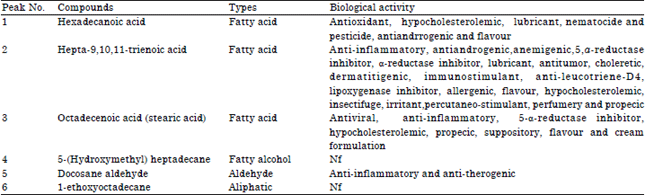 | |
| NF: Not found, Source: Dr. Duke’s phytochemical and ethnobotanical database | |
Table 2 listed the various phytochemical constituents which contribute to the medicinal activity of methanol extract of S. uliginosa (Sw). The beneficial actions of the phytocompound listed in Table 2 are based on phytochemical and ethnobotanical database by Jim Duke of the Agricultural Research Service/USDA.
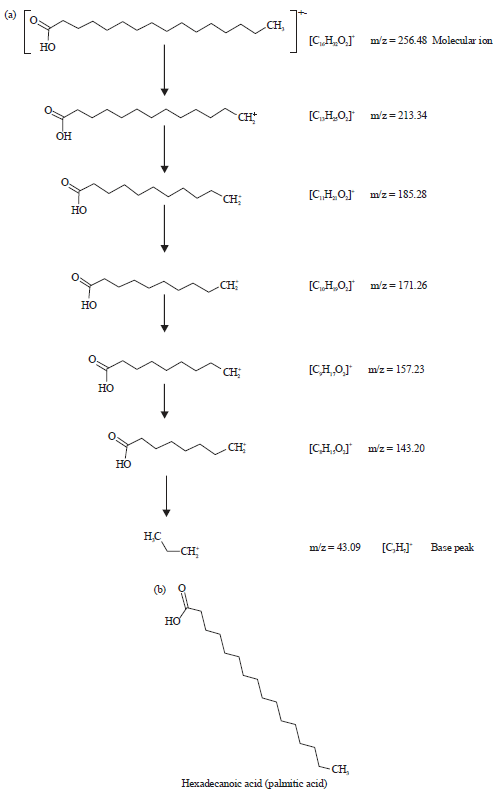 | |
| Fig. 3(a-b): | (a) Fragmentation patterns of the peak number 1 and large fragments at different m/z ratios of methanol extarct of leaves of Spilanthes uliginosa (Sw) and (b) Structure of the phytocompound of peak number 1 of the extarct of leaves |
The fragmentation patterns of the peaks and identified compounds of the plant were shown in Fig. 3-8. This indicated degeneration of outsized fragments into diminutive compounds giving rise to apperance of peaks at different m/z ratios.
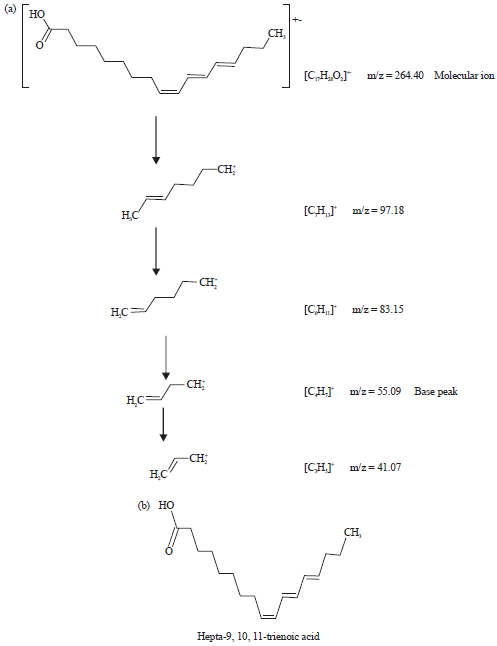 | |
| Fig. 4(a-b): | (a) Fragmentation patterns of the peak number 2 and large fragments at different m/z ratios of methanol extract of leaves of Spilanthes uliginosa (Sw) and (b) Structure of the phytocompound of peak number 2 of the extract of leaves |
The characterization of phytochemicals by Gass Chromatography-Mass Spectrometry (GC-MS) is a very crucial method to X-ray the possible therapeutic potentials of plant materials. Among the identified phytochemicals in Spilanthes uliginosa (Sw) methanol leaves extract are, hexadecanoic acid, hepta-9, 10, 11-trienoic acid, octadecenoic acid, 5-hydroxylmethyl heptadecane, docosane aldehyde and 1-thoxyoctadecane and each of these compounds has an important life action as stated in Table 2. The result of this study is in agreement with the study of Uraku et al. (2015b) on Gas Chromatography-Mass Spectrometry (GC-MS) analysis of essential oil from Hyptis spicigera leaves where they reported that the palnt contained six phytocompounds.
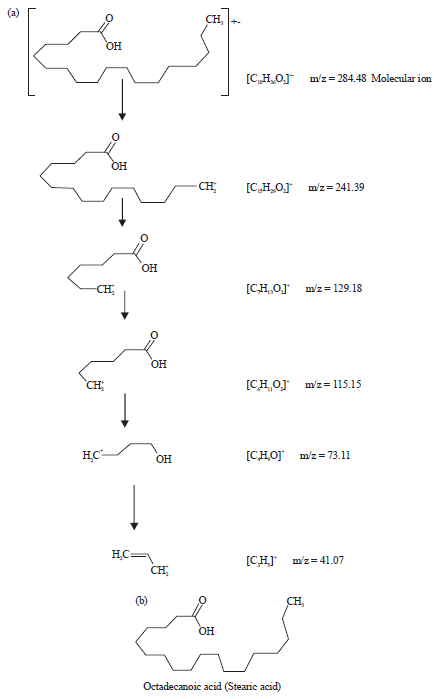 | |
| Fig. 5(a-b): | (a) Fragmentation patterns of the peak number 3 and large fragments at different m/z ratios of methanol extract of leaves of Spilanthes uliginosa (Sw) and (b) Structure of the phytocompound of peak number 3 of the extract of leaves |
Hexadecanoic acid posseses antioxidant activity (Mohan et al., 2014) as well as hypocholesrerolemic and antiandrrogenic activities (Gopalakrishnan and Udayakumar, 2014). Hepta-9, 10, 11-trienoic acid, octadecenoic acid and docosane aldehyde exhibit anti-inflammatory activities (Uraku, 2015) while the compounds hexadecanoic, hepta-9, 10, 11-trienoic and octadecanoic low the levels of cholesterol in the body (Sutha et al., 2012). This study conformed to the report of Gopalakrishnan et al. (2011) on GC-MS analysis of the methanolic extract of the leaves of Dipteracanthus patulus (Jacq.) Nees. Also, hepta-9, 10, 11-trienoic acid and octadecenoic acid has antitumor and antiviral property respectively.
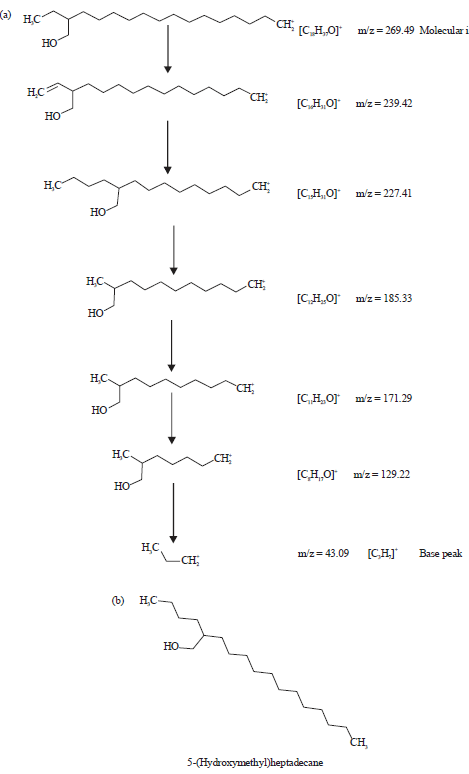 | |
| Fig. 6(a-b): | (a) Fragmentation patterns of the peak number 4 and large fragments at different m/z ratios of methanol extract of leaves of Spilanthes uliginosa (Sw) and (b) Structure of the phytocompound of peak number 4 of the extarct of leaves |
The result of this study is in concurrence with the research of Achi and Ohaeri (2015) on GC-MS determination of bioactive constituents of the methanolic fractions of Cnidoscolus aconitifolius. Also, the biological activites of the phytocompounds stated in this study agreed with the work of Mohan et al. (2014) on GC-MS analysis of bioactive components of tubers of Rucllia tuberosa L. (Acanthaceae) and that of Ambikapathy et al. (2011).
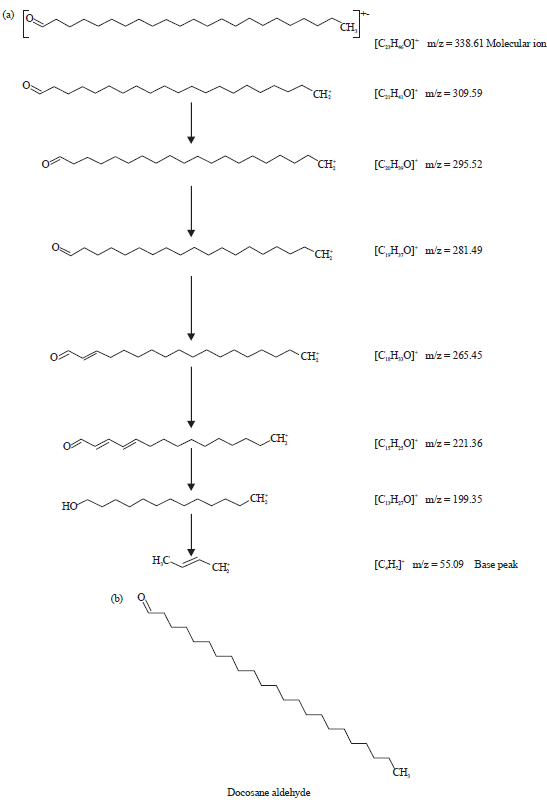 | |
| Fig. 7(a-b): | (a) Fragmentation patterns of the peak number 5 and large fragments at different m/z ratios of methanol extract of leaves of Spilanthes uliginosa (Sw) and (b) Structure of the phytocompound of peak number 5 of the extract of leaves |
The result of this analysis revealed that the plant contained promising novel class of pharmaceutical active compounds that might play beneficial roles in the treatment of diverse diseases in human.
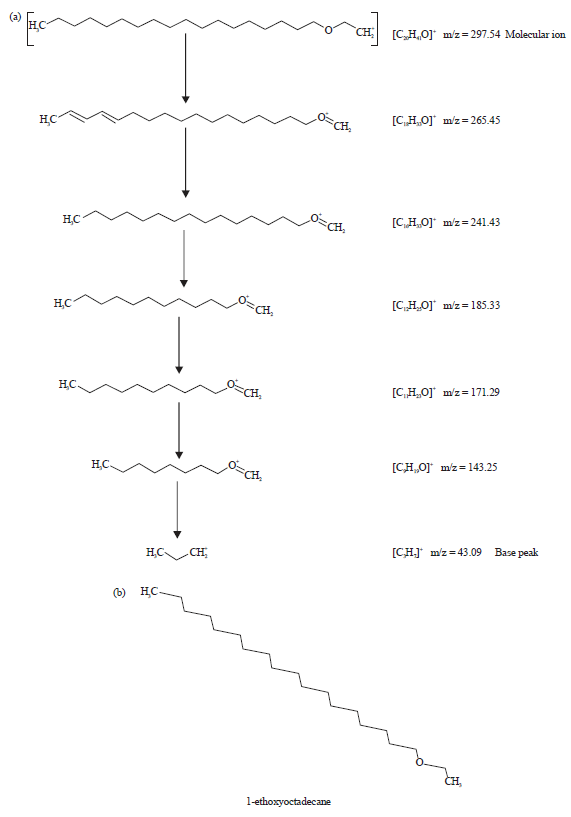 | |
| Fig. 8(a-b): | (a) Fragmentation patterns of the peak number 6 and large fragments at different m/z ratios of methanol extract of leaves of Spilanthes uliginosa (Sw) and (b) Structure of the phytocompound of peak number 6 of the extract of leaves7 |
Thus, this kind of GC-MS analysis is the first step towards understanding the nature of active principles in this medicinal plant and this type of study will be helpful for further detailed study. Further investigation into the pharmacological importance of S. uliginosa (Sw) and their diversity and detailed phytochemistry may add new knowledge to the information in the traditional medicinal systems.
CONCLUSION
The GC-MS method is a direct and fast analytical approach for identification of terpenoids and steroids and only few grams of plant material is required. The importance of the study is due to the biological activity of some of these compounds. The present study, which reveals the presence of components in tuber of S. uliginosa (Sw) suggest that the contribution of these compounds on the pharmacological activity should be evaluated.
The GC-MS analysis revealed that the leaf extract of S. uliginosa (Sw) contained appreciable high amounts of docosane aldehyde, hepta-9, 10, 11-trienoic acid and 5-(hydroxymethyl) heptadecane with low amounts of 1-ethoxyoctadecane, Octadecenoic acid and hexadecanoic acid. The presence of these phytocompounds may be responsible for its popular use in treatment of numerous diseases by traditional users.
ACKNOWLEDGMENT
This study was financially supported by Director of Research, Innovation and Commercialization (DRIC), EBSU and TETFund.
REFERENCES
- Sunday, A.G., O.E. Ifeanyi and O.F. Uzor, 2014. Wound healing potential of water and ethanol extract of fresh leaves and bark Ofgambia albidum in albino rats. IOSR J. Dental Med. Sci., 13: 16-22.
Direct Link - Uraku, A.J., A.N.C. Okaka, U.A. Ibiam, K.N. Agbafor and N.A. Obasi et al., 2015. Antiplasmodial activity of ethanolic leaf extracts of Spilanthes uliginosa, Ocimum basilicum (Sweet Basil), Hyptis spicigera and Cymbopogon citrates on mice exposed to Plasmodium berghei Nk 65. J. Biochem. Res. Rev., 6: 28-36.
Direct Link - Shanthi, P. and P. Amudha, 2010. Evaluation of the phytochemical constituents of Acmella calva (DC.) R.K. Jansen. Int. J. Pharma Bio Sci., 1: 308-314.
Direct Link - Uraku, A.J. and M.E. Ogbanshi, 2015. Nutritional and phytochemical potential of Spilanthes uliginosa (Sw) leaves. Am. J. Phytomed. Clin. Ther., 3: 111-119.
Direct Link - Ramsewak, R.S., A.J. Erickson and M.G. Nair, 1999. Bioactive N-isobutylamides from the flower buds of Spilanthes acmella. Phytochemistry, 51: 729-732.
CrossRefDirect Link - Ratnasooriya, W.D. and K.P.P. Pieris, 2005. Attenuation of persistent pain and hyperalgesia by Spilanthus acmella flowers in rats. Pharmaceut. Biol., 43: 614-619.
CrossRefDirect Link - Rani, S.A. and S.U. Murty, 2006. Antifungal potential of flower head extract of Pilanthes acmella Linn. Afr. J. Biomed. Res., 9: 67-69.
Direct Link - Tanwer, B.S., R. Choudhary and R. Vijayvergia, 2010. In vitro and in vivo comparative study of primary metabolites and antioxidant activity in Spilanthes acmella Murr. Int. J. Biotechnol. Biochem., 6: 819-825.
Direct Link - Victor, O.O. and S.M. David, 2015. GC-MS analysis of phyto-components from the leaves of Senna alata L. J. Plant Sci., 3: 133-136.
CrossRefDirect Link - Uraku, A.J., C.E. Offor, E.J. Itumoh, C.E. Ukpabi and P.M. Aja et al., 2015. Gas Chromatography-Mass Spectrometry (GC-MS) analysis of essential oil from Hyptis spicigera leaves. Am. J. Biol. Chem., 3: 45-56.
Direct Link - Balamurugan, K., A. Nishanthini, S. Lalitharani and V.R. Mohan, 2012. GC-MS determination of bioactive components of Melastoma malabathricum L. Int. J. Curr. Pharmaceut. Res., 4: 24-26.
Direct Link - Mohan, V.R., N.R. Kumar and K. Vasantha, 2014. GC-MS analysis of bioactive components of tubers of Ruellia tuberosa L. (Acanthaceae). Am. J. Phytomed. Clin. Therapeut., 2: 209-216.
Direct Link - Gopalakrishnan, K. and R. Udayakumar, 2014. GC-MS analysis of phytocompounds of leaf and stem of Marsilea quadrifolia (L.). Int. J. Biochem. Res. Rev., 4: 517-526.
Direct Link - Uraku, A.J., 2015. Determination of chemical compositions of Cymbopogon citratus leaves by Gas Chromatography-Mass Spectrometry (GC-MS) method. Res. J. Phytochem., 9: 175-187.
CrossRefDirect Link - Sutha, S., V.K. Devi and V.R. Mohan, 2012. GC-MS determination of bioactive components of Alstonia venenata R. Br. Res. J. Pharmaceut. Biol. Chem. Sci., 3: 291-296.
Direct Link - Gopalakrishnan, S., K. Saroja and J.D. Elizabeth, 2011. GC-MS analysis of the methanolic extract of the leaves of Dipteracanthus patulus (Jacq.) Nees. J. Chem. Pharm. Res., 3: 477-480.
Direct Link - Achi, N.K. and O.C. Ohaeri, 2015. GC-MS determination of bioactive constituents of the methanolic fractions of Cnidoscolus aconitifolius. Br. J. Pharm. Res. Int., 5: 163-172.
CrossRefDirect Link - Ambikapathy, V., R. Mahalingam and A. Panneerselvam, 2011. GC-MS determination of bioactive compounds of Enicostemma littorale (Blume). Asian J. Plant Sci. Res., 1: 56-60.
Direct Link








