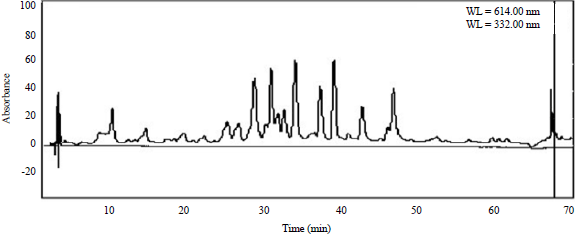
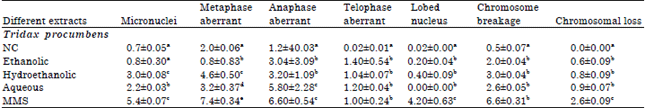
Research Article
Cytotoxicity and Genotoxicity of Tridax procumbens L. in Allium cepa
Laborat�rio de Fitotera Picos, Departamento de Ci�ncias Biol�gicas, Faculdade de Ci�ncias e Letras de Assis, Universidade Estadual Paulista (UNESP), Avenida Dom Ant�nio 2100, CEP: 19806-900, Assis, S�o Paulo, Brasil
Michelly Cristina Montenoti
Faculdade de Medicina de Mar�lia (FAMEMA), Mar�lia, S�o Paulo, Brasil
Vanessa Marques de Oliveira Moraes
Laborat�rio de Fitotera Picos, Departamento de Ci�ncias Biol�gicas, Faculdade de Ci�ncias e Letras de Assis, Universidade Estadual Paulista (UNESP), Avenida Dom Ant�nio 2100, CEP: 19806-900, Assis, S�o Paulo, Brasil
Luciana Pereira Silva
Funda��o Educacional do Munic�pio de Assis (FEMA), Assis, S�o Paulo, Brasil
Regildo Marcio Goncalves da Silva
Universidade Estadual Paulista (UNESP), Instituto de Qu�mica de Araraquara, Rua Prof. Francisco Degni 55, CEP: 14800-060, Araraquara, S�o Paulo, Brasil