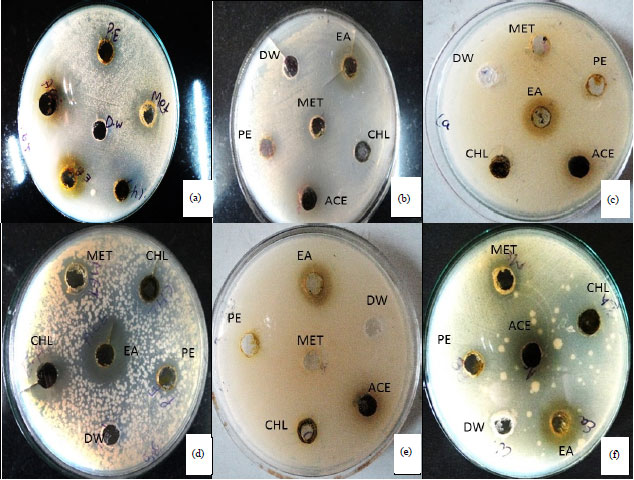
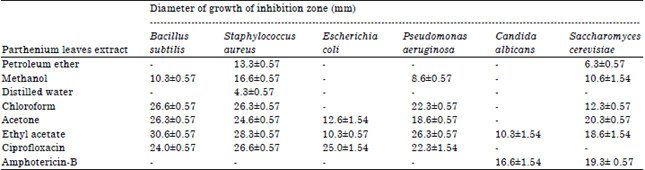
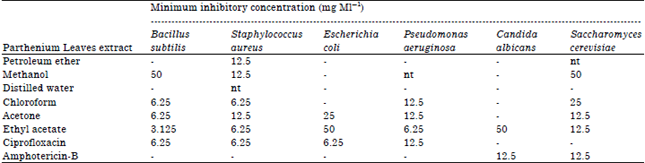
Research Article
Antimicrobial Activity of Medicinal Plant: Parthenium hysterophorus L.
Department of Microbiology, Kurukshetra University, Kurukshetra, 136119, India
LiveDNA: 91.15611
N.K. Aggarwal
Department of Microbiology, Kurukshetra University, Kurukshetra, 136119, India
R. Dhiman
Department of Microbiology, Kurukshetra University, Kurukshetra, 136119, India