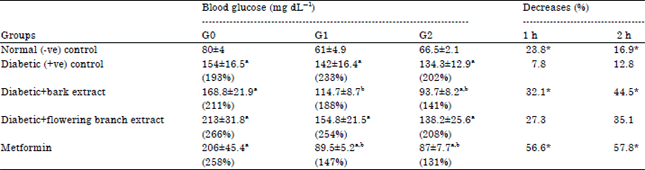
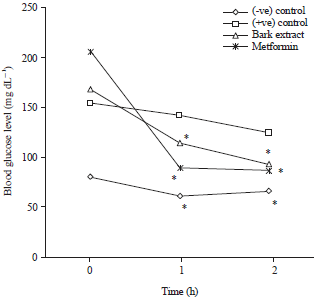
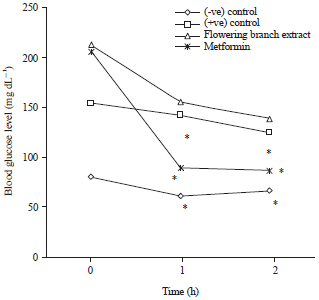
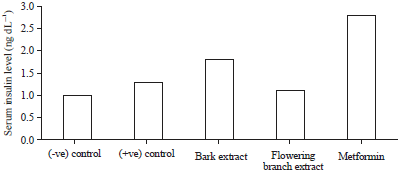
Research Article
Comparative Study of Biochemical Changes in Alloxan Induced Diabetic Mice Treated with Extracts of Spathodea campanulata Flowering Branch and Barks
Department of Biochemistry, Dubai Medical College, Dubai, UAE
Naglaa Ahmed
Department of Pharmaceutical Chemistry and Natural Products, Dubai Pharmacy College, Dubai, UAE