Research Article
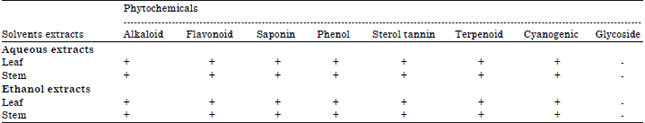
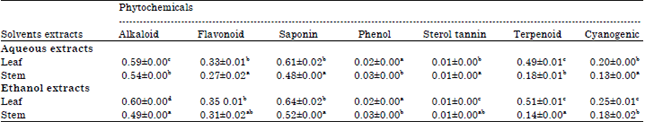
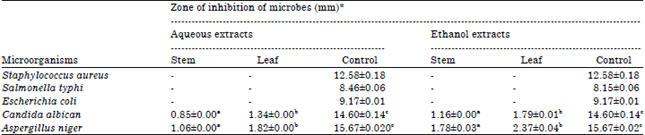
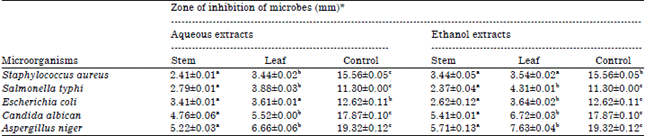
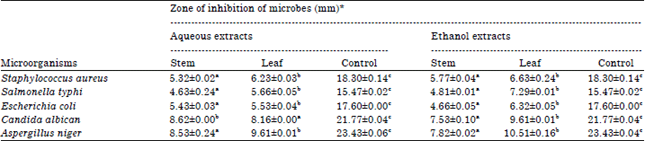
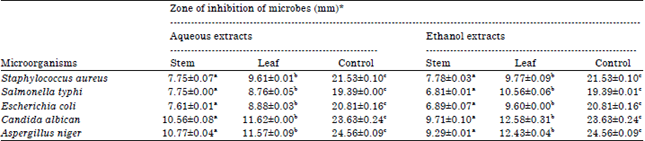
Phytochemical Screening, Antifungal and Antibacterial Activity of Aqueous and Ethanolic Leaf and Stem Extracts of Gnetum africanum Welw
Department of Botany, Nnamdi Azikiwe University, P.M.B. 5025, Awka, Anambra State, Nigeria
Rachael U. Ugwu
Department of Botany, Nnamdi Azikiwe University, P.M.B. 5025, Awka, Anambra State, Nigeria
Onyinye L. Nwokolo
Department of Botany, Nnamdi Azikiwe University, P.M.B. 5025, Awka, Anambra State, Nigeria
Maureen U. Chukwuma
Department of Biology, Nwafor Orizu College of Education Nsugbe, Anambra State, Nigeria
Ebele E. Akachukwu
Department of Biology, Nwafor Orizu College of Education Nsugbe, Anambra State, Nigeria