Short Communication
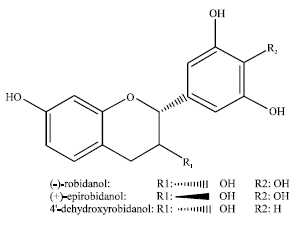
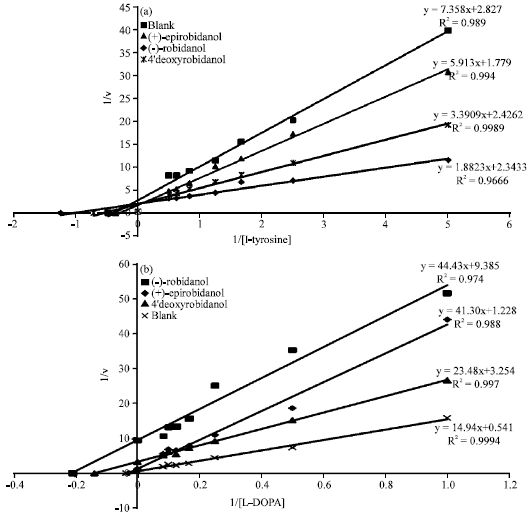
Tyrosinase Kinetic Inhibition of Active Compounds from Intsia palembanica
Biopharmaca Research Center, Bogor Agricultural University, Jl Taman Kencana No. 3, Bogor, 16151, Indonesia
I. Batubara
Biopharmaca Research Center, Bogor Agricultural University, Jl Taman Kencana No. 3, Bogor, 16151, Indonesia
T. Mitsunaga
Faculty of Applied Biological Science, Gifu University, 1-1 Yanagido, Gifu, 501-1193, Japan
M. Rahminiwati
Biopharmaca Research Center, Bogor Agricultural University, Jl Taman Kencana No. 3, Bogor, 16151, Indonesia
E. Djauhari
Biopharmaca Research Center, Bogor Agricultural University, Jl Taman Kencana No. 3, Bogor, 16151, Indonesia
K. Yamauchi
Faculty of Applied Biological Science, Gifu University, 1-1 Yanagido, Gifu, 501-1193, Japan