ABSTRACT
The GI tract of an individual which is inhabited by a large number of microbes, is under constant threat from pathogens and oxidative damage. In Indian system of medicine, several cucurbits have been widely documented for prophylactic and therapeutic purposes to ameliorate such situations. The total flavonoids, observed in the extract of three cucurbits, Lagenaria siceraria (Ls), Luffa cylindrica (Lc) and Cucurbita pepo (Cp), were 17.9±1.3, 6.3±0.2 and 2.1±0.5 mg g-1, respectively. The antioxidant activity, determined by DPPH assay, displayed that Ls, Cp and Lc had 77±1.3, 10±3 and 10±1.0% free radical scavenging activity, respectively. Reduction Potential (RP) assay revealed the antioxidant effects for Ls, Lc and Cp as 44±0.8, 22±.3 and 15±.8%, respectively. Maximum activity of quenching H2O2 was demonstrated by Lc (92±1%) as compared to Cp (78±2%) and Ls (76±1.6%). Antimicrobial activity of Lactobacillus rhamnosus measured through generation of inhibition zone against E. coli was augmented maximally in presence of Ls (4.4±0.3 cm), followed by Cp (4.2±.01 cm) and Lc (4.0±.03 cm). Biofilm formation by L. rhamnosus was maximally supported in presence of Lc (absorbance = 0.26±0) followed by Ls (0.21±.01) and Cp (0.20±.01). Thus, these dietary cucurbits showed appreciable antioxidant activity and enhance antimicrobial and barrier function of mucosa by L. rhamnosus and impart protection to gut without any toxic effects.
PDF Abstract XML References Citation
How to cite this article
URL: https://scialert.net/abstract/?doi=rjmp.2012.500.510
INTRODUCTION
The Reactive Oxygen Species (ROS) and free radicals have been implicated in a wide range of pathological conditions such as inflammations (Schetter et al., 2010), cancer, diabetes (Moussa, 2008), arthritis and neurodegenerative diseases. In the gut the free radicals are generated during cellular and metabolic activities of host or pathogens and also due to interaction between different components like various molecules of the pathogens and host and also the dietary molecules present inside the gut lumen. The exogenous agents such as heat, cold, ionizing radiation, oxidative drugs and pollutants are known to generate free radicals and sequentially the oxidative damage (Milan et al., 2011). Although free radicals and ROS are essential components of the biological system but their production and generation beyond the essential limit or their leakage at undesired sites, cause many perturbations in the constitutive and functional molecules leading to biological damage. The living system therefore constantly quenches leaked and free oxidative species through various endogenous scavenging mechanisms such as SOD, GSH, etc. (Olsvik et al., 2005).
The GI tract is indeed most vulnerable to various pathogens and free radical damage. The microbes are known to harbour the GI tract in huge numbers, extending from the stomodeum to proctodeum. In the GI tract, there are about 450-500 species of microbes belonging to about 40 genera ranging from useful (probiotic) to commensal and opportunistic to virulent pathogens but there is also an inter-specific pressure on various microbial populations. In the gut, the peak populations keep shifting and such a shift may sometimes cause pathogenic manifestations in the GI tract and the body. During various pathological conditions, the population of pathogens shifts towards dominance at the cost of probiotic population. For a healthy gut, the population of different bacteria in an inter-specific dialogue remains under over all control of the probiotic bacteria. The literature has a large number of citations deliberating on probiotics (Like L. rhamnosus, Bifidobacterium bifidum) and their symbiotic relationship with the cells and tissue of the GI tract of host. Probiotics are known to increase the break-down of certain polysaccharides, prevent injury and inflammation, protect gut from pathogenic microbes like Salmonella, Escherichia coli, Salmonella typhimurium, Staphylococcus aureus, Vibrio cholera, etc. by secreting antimicrobial agents (Premalatha and Dhasarathan, 2011). Different species of Lactobacillus produce several antibacterial compounds which display varying degree of effects against different bacteria. Sharaf and Al Harbi (2011) reported that antimicrobial agents produced by 40 species of lactic acid bacteria were effective against E. coli, S. aureus and Candida ablicans, etc. but not against methicilin resistant Streptococcus aureus. Nisin is one of the most effective antibacterial agent produced by Lactobacillus lactis and has been used as a natural preservative in food industry. Bisin is another antimicrobial agent produced by Lactobacillus and is a more potent antimicrobial agent. In this study we have attempted to evaluate the antibacterial activity of compounds produced by L. rhamnosus against E. coli using well diffusion assay.
Biofilm formation is another important activity of Lactobacillus. Through this activity, probiotics provide specific health benefits to the host such as immunomodulation and denial to the pathogenic bacteria to adhere to mucosal cells by forming a physical barrier on the gut mucosa (Jones and Versalovic, 2009; Kullisaar et al., 2002). Lactobacilli also constitute a source of dietary antioxidants (Saide and Gilliland, 2005). It has also been reported that Lactobacillus fermentum contains glutathione, a potent cellular antioxidant and eliminates hydroxyl radicals. In fact Bifidobacterium and Lactobacillus spp. display a substantial impact on our enteric system. Therefore, to harness the maximum health benefits to the host gut the growth of probiotic population need to be sequentially supported. Some dietary products have the potential to improve the growth and useful activities of the probiotic bacteria and such molecules are known as prebiotics. In the present study, cucurbits were investigated for their prebiotic potential and subsequent therapeutic implications.
Many plants like Rhodiola imbricata, Hippophae rhamnoides, etc. have been reported for several therapeutic purposes against diseases caused by inflammation, free radicals and radiation etc. (Goel et al., 2006). Cucurbits, belonging to family Cucurbitaceae include about 118 genera and 825 species. In India, cucurbits are cultivated in a large number which constitute about 5.6% of the total vegetable production. The fruits of many plants of Cucurbitaceae family like Momordica charantia, Cucumis sativus, etc., have been widely used for medicinal purposes in India (Sahu et al., 2011). The health promoting ability of fruits to some extent might be related to the antioxidant properties of their constituents.
The common dietary cucurbits, such as Ls, Lc and Cp have been reported for the presence of not only essential nutrients such as carbohydrates, vitamins etc but also for containing secondary metabolites such as alkaloids, flavonoids, glycosides, steroids and saponins, etc. (Irshad et al., 2010). These compounds work as antioxidants (Oskoueian et al., 2011). Other important biomolecules present in different dietary cucurbits display a wide spectrum ranging from cucurbitacins, sterols (fuco-and compe-sterol), arepene and bryonolic acid. The biochemical, pharmacological and therapeutic information about various cucurbits has been extensively reported in the literature due to the pharmaco-therapeutic applications (Arora et al., 2011; Dhiman et al., 2012). Cucurbita has been very effective as anti-depressant (Umadevi et al., 2011) and antioxidant (Leman et al., 2007), Luffa renders antibacterial, antifungal (Devi et al., 2009), antioxidant and anti-inflammatory effects while Lagenaria manifests anti-proliferative (Shah et al., 2010), immuno-modulatory (Gangwal et al., 2008), anticancer and anti-oxidant properties (Erasto and Mbwamb, 2009; Onasanwo et al., 2011; Saha et al., 2011). The present investigations were taken up to explore the potential of cucurbits in amelioration of the stress generated by pathogens and free radicals in GI tract. These studies could ultimately lead to the development of various strategies to strengthen the synergy among the prebiotic, probiotic and host.
MATERIALS AND METHODS
Bacterial cultures: L. rhamnosus and E. coli were procured from Department of Biosciences, Jamia Milia Islamia, New Delhi and grown on MRS and EMB media respectively at 37°C in the sterilized environment and were harvested after 48 h. Both strains were purified by repeated plating in specific media and identified by gram reaction, cell morphology and catalase reaction.
Preparation of extracts: Fresh fruits of the cucurbits, L. siceraria (bottle gourd), L. cylindrica (sponge gourd) and C. pepo (pumpkin), were procured from the local market. The plant material was thoroughly washed with sterile distilled water several times and equal quantity of each plant material (50 g/100 mL) was homogenized separately in presence of solvent (absolute ethanol and triple distilled water; 50:50, v/v). After 24 h, the homogenate was filtered through a fine strainer having a spread of muslin cloth and extract was stored at 4°C in bottles with airtight cork.
Chemicals: Analytical grade chemicals like DPPH (1,1-diphenyl-2-picrylhydrazyl), ascorbic acid, NaCl, acetone, H2O2, trichloroacetic acid were procured from M/S Qualigen (Mumbai) and potassium ferricyanide, ferric chloride (pH -7.4), KCl, KH2PO4, K2HPO4, NaH2PO4 and Na2HPO4 were procured from M/S Merck, Germany. MRS and EMB media were purchased from M/S Himedia, Mumbai.
Determination of total flavonoid contents (TFC): TFC was determined in all the three cucurbit extracts using the method described by Hassan and Hassan (2010).
One milliliter of each extract was mixed with 1.0 mL of aluminum trichloride in ethanol (20 mg mL-1) and a drop of glacial acetic acid and then diluted with ethanol to 25 mL. The absorption was read after 40 min using a Systronic double beam spectrophotometer. Blank was prepared using 1.0 mL extract and a drop of glacial acetic acid and then diluted to 25 mL with ethanol. The absorption of standard quercetin solution (0.5 mg mL-1 in ethanol) was measured under the same conditions. The amount of total flavonoids in extract, in quercetin equivalents was calculated by the following equation:
where, X is the flavonoid contents (mg g-1 extract in QE). A and A0 is the absorption of extract and quercetin solution, respectively. m is the weight of the cucurbits (mg) in 1 mL of cucurbit extracts and m0 is the weight of quercetin (mg) in 1 mL of quercetin solution.
Assessment of antioxidant activity
DPPH assay: The antioxidant activity of cucurbit extracts, on the basis of scavenging ability of the stable DPPH free radicals, was determined as described by Brand-Williams et al. (1995). Definite volume (50-250 μL) of an extract was taken in the test tubes and triple distilled water was added to make it to 1.0 mL. The 2.0 mL of DPPH solution (0.1 mM in methanol) was added to each test tube, mixed well and incubated at room temperature for 30 min. Absorbance of the mixture was measured at 517 nm using double beam spectrophotometer. Percent inhibition of DPPH radical was calculated by the following equation:
where, A0 referred to the absorbance of the control and A1 of the test sample.
Reduction potential assay: Reducing power of the extract was determined by the method of Oyaizu (1986). Known quantity of cucurbit extract (100-1000 μL) was mixed with sodium phosphate buffer (2.5 mL) and potassium ferricyanide (2.5 mL) and incubated at 50°C for 20 min. 2.5 mL of trichloroacetic acid was added to the mixture and centrifuged at 3000 rpm for 10 min. The upper layer of solution (2.5 mL) was mixed with distilled water (2.5 mL) and a freshly prepared ferric chloride solution (0.5 mL). The absorbance was measured at 700 nm. Percent increase in reduction potential was calculated as follows:
where, Atest showed the absorbance of test solution and Ablank of the blank.
Hydrogen peroxide (H2O2) scavenging assay: The hydrogen peroxide scavenging activity of plant extracts was evaluated following the method of Ruch et al. (1989). A solution of 40 mM H2O2 was prepared in 50 mM potassium phosphate buffer (pH 7.4). Different quantities of extracts (100-500 μL) were added to 2 mL of H2O2 solution (40 mM) and absorbance was taken at 230 nm after 10 min against a blank solution (phosphate buffer without H2O2). The percentage of scavenged H2O2 was calculated using following equation:
where, A0 was the absorbance of the blank and A1 of the sample.
Assessment of antimicrobial activity of L. rhamnosus against E. coli and its modulation by cucurbits extract
Well diffusion assay: Following the method described by Holder and Boyce (1994) and Raja et al. (2010), soft nutrient agar plates were prepared (in petri-dish of 9 cm diameter) and seeded with test organism (E. coli) and wells of equal size (2.5 cm in diameter) were prepared on each agar plate. Two milliliter of L. rhamnosus broth was poured in each well with or without cucurbit extract (500 μL). The plates were incubated for 24 h at 37°C in sterile environment. The Zones of inhibition were measured after 24 h.
Assessment of effect of cucurbits on the biofilm formation by L. rhamnosus
Peg method: Biofilm formation by L. rhamnosus and its modulation by cucurbits was studied using peg method (Smith et al., 2008). One milliliter of L. rhamnosus broth was inoculated in 100 mL of MRS media with or without cucurbit extracts (1 mL) in different beakers. Pegs were prepared by micropipette tips (with micropipette stand), immersed in the media in beakers. Beakers were kept in the sterile environment for 24 h at 37°C. After 24 h, biofilm formed on pegs was washed with 2 mL of acetone and absorbance was taken at 600 nm using double beam spectrophotometer.
Statistical treatment: The data of at least three experiments, each conducted separately, was subjected to statistical analysis and results were presented in the form of MSE (Mean Standard Error).
RESULTS
Total flavonoid contents: Among the three cucurbits studied here, Ls contained highest amount of flavonoids (17%) followed by Lc (6.3%) and Cp (2.1%), respectively (Table 1). The presence of flavonoids correlated well with the DPPH scavenging and reduction potential of the cucurbits (Fig. 1, 2). Ls due to 17% flavonoids rendered maximum antioxidant activities as compared to Lc and Cp which had 6.3 and 2.1% flavonoids, respectively.
DPPH scavenging activity: Percent inhibition of DPPH radicals has been displayed in Fig. 1. Ls rendered the maximum inhibition of DPPH radicals from 26 to 72% when the extract volume was increased gradually from of 50 to 150 μL. Further increase upto 250 μL did not yield an appreciable increase in DPPH radicals scavenging activity which followed a plateau increasing from 72 to 77%. Lc and Cp revealed highly reduced antioxidant effects in comparison to Ls and the percent inhibition of DPPH radicals could increase from 2% at 50 μL to 10% at 250 μL of extract. Ls extract therefore, contains some strong antioxidant molecules in comparison to Lc and Cp.
Reduction potential (RP): The RP of the cucurbit extracts increased consistently with the increase in the volume of extract. The increase in the volume of the extracts from 100 to 500 μL correspondingly increased RP from 10 to 44% (Fig. 2).
| Table 1: | Total flavonoid contents measured as quercetin equivalent (QE) in the cucurbit fruit extracts |
 | |
| Each value is a mean of three experiments and has been expressed as Mean±SE | |
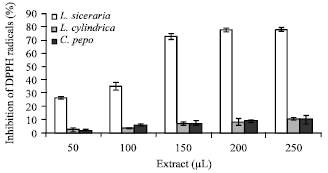 | |
| Fig. 1: | DPPH radical scavenging activity of different cucurbit extracts, each value is a mean of three experiments and has been expressed as Mean±SE |
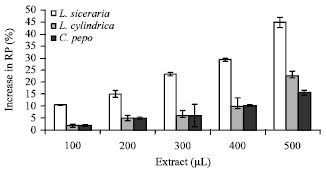 | |
| Fig. 2: | Reduction potential of different cucurbit extracts, each value is a mean of three experiments and has been expressed as Mean±SE |
At corresponding volumes of the extracts Lc and Cp rendered the increase from 2 to 22% and 2 to 15%, respectively.
Hydrogen peroxide scavenging activity: The cucurbit extracts revealed concentration dependent H2O2 scavenging activity. An increase in the total quantity of the extract rendered a proportionate increase in the scavenged H2O2 radicals (Fig. 3). Lc displayed maximum H2O2 radical inhibition from 38 to 92% with an increase in the volume of extract from 100 to 500 μL. At 100 μL, Ls displayed about 71% inhibition which increased gradually to 76% with the increase in the extract volume from 100 to 500 μL. The activity of scavenging peroxide radicals was enhanced from 23 to 78%, when the volumes of Cp extract were enhanced from 100 to 300 μL and maintained a plateau thereafter up to 500 μL.
Antimicrobial activity (well diffusion assay): L. rhamnosus rendered a zone of inhibition (ZI) of 4 cm diameter against E. coli. Addition of cucurbit extracts in the well enhanced the ZI, generated by L. rhamnosus. The cucurbits alone did not reveal any antimicrobial effect directly; however they augmented the antimicrobial activity of L. rhamnosus. Lc did not manifest any effect on the ZI since the ZI was 4 cm that was equal to the control. However, Ls and Cp increased the diameter of ZI which remained as 4.4 and 4.2 cm (Table 2).
Modulation of L. rhamnosus generated biofilm by cucurbits: The modulating effect of the cucurbits on biofilm formation by L. rhamnosus has been shown in Table 3.
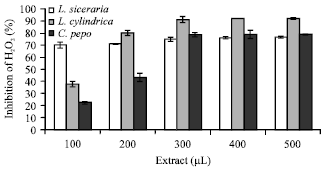 | |
| Fig. 3: | H2O2 scavenging activity of different cucurbit extracts, each value is a mean of three experiments and has been expressed as Mean±SE |
| Table 2: | Antimicrobial activity of L. rhamnosus and its modulation by cucurbit extracts |
 | |
| Each value is a mean of three experiments and has been expressed as Mean±SE | |
| Table 3: | Effect of cucurbit extracts on the biofilm formed on the plastic pegs by L. rhamnosus |
 | |
| Each value is a mean of three experiments and has been expressed as Mean±SE | |
The density of the bacterial cells (L. rhamnosus), dislodged from pegs, was determined by measuring the absorbance at 600 nm. The density of the cells of L. rhamnosus in biofilm generated without cucurbits, revealed the absorbance as 0.184. L. rhamnosus cells in the biofilm however increased in presence of cucurbit extracts. In presence of Lc, extract maximum absorbance observed was 0.26 however, in presence of Ls and Cp extract; the absorbance remained as 0.211 and 0.20, respectively (Table 3).
DISCUSSION
Cucurbits have several active biomolecules like cucurbitacins A, B, C, D, E, I, J, K, L and their glycosides, flavonoids and flavone glycosides (Torkey et al., 2009). The cucurbitacins are of great interest because of their wide spectrum of biological activities like cytotoxic, hepatoprotective, cardiovascular, antidiabetic, antioxidant, anti-inflammatory, analgesic and anti-proliferative activities. Gill and Bali (2011) have shown the anti-ulcer and antioxidant activities of tetracyclic triterpenoids (cucurbitacins) extracted from Cucurbita pepo seeds.
Flavonoid and phenolic contents are the other important bioactive molecules responsible for wide spectrum of biological characteristics as cell proliferation enhancement, immunomodulatory, antioxidant, anti-allergic and anti-inflammatory agents (Knekt et al., 2002; Pattanayak et al., 2012; Elzaawely and Tawata, 2012). Therefore, in the present study, the flavonoid contents in Ls, Lc and Cp were investigated and Ls was observed to contain the highest amount of flavonoids in comparison to Lc and Cp (Table 1).
DPPH assay is known to give reliable information concerning the antioxidant ability of the tested compounds (Huang et al., 2005). It depends on the color change of DPPH solution from purple to yellow as the radical is quenched by an antioxidant (Karagozler et al., 2008). The hydrogen or electron donation by antioxidants like cucurbits leads to the scavenging of free radicals. This reduces the number of DPPH radicals and protects the host body from oxidative damage. Ls displayed the DPPH radicals scavenging activity much higher than those displayed by Lc and Cp. Thus Ls having higher amount of flavonoids and DPPH scavenging activity could have more potential as an antioxidant out of three cucurbits studied here.
The antioxidant and reduction potential activity proceeds in a complementary manner, therefore reduction potential may be used as an indirect indicator of antioxidant activity (Yen et al., 2001). It is known that the presence of an antioxidant in a solution reduces Fe3+ to Fe2+ and the Fe2+ complexes so formed lead to the formation of ferric-ferrocyanide complex. The ability to reduce the Fe3+ to Fe2+ may be attributed to hydrogen donation by phenolic compounds (Shimada et al., 1992). The results of this study indicated a good correlation between the quantity of flavonoids (Table 1) and increase in reduction potential (Fig. 2).
Hydrogen peroxide is a weak oxidizing agent that crosses cell membrane barrier rapidly and inactivates a few enzymes directly. It can react with Fe2+ and possibly Cu2+ ions to form hydroxyl radicals and thus may be responsible for many of the toxic effects and oxidative damage (Wijeratne et al., 2005). Therefore, to protect the living system, it is necessary to remove hydrogen peroxide from the site for protection of the living system. Here, Lc displayed the maximum activity of peroxide scavenging as compared to Ls and Cp.
Lactobacilli produce various antimicrobial substances and protect gut from pathogens (Amdekar et al., 2010). Nisin is one of the most effective antibacterial agents, produced by Lactobacillus lactis which has been widely used as food preservative (Delves-Broughton, 2005). The antimicrobial agent produced by the strain of L. rhamnosus used here, has not been characterized, however, its antimicrobial effects were evaluated. The up-regulation of antimicrobial activity of L. rhamnosus in the presence of cucurbits was evaluated by monitoring zone of inhibition (ZI) generated for controlling the growth of E. coli. The results indicated that various cucurbits differed in their supportive mechanism that yielded antimicrobial up-regulation in L. rhamnosus. Different cucurbits contained different kinds of bioactive molecules which may be responsible for such variations in the magnitude of action. The molecules, responsible for such action, need to be investigated further for establishing structure-function relationship.
Probiotics like Lactobacillus protect the gut from pathogens and free radicals by forming a physical barrier (biofilm) on the gut mucosa (Jones and Versalovic, 2009; Kullisaar et al., 2002). Some probiotic bacteria attach themselves to the surface of epithelial cells while others prefer to remain embedded in the extracellular polymeric substances forming a matrix outside in absolute vicinity of the cells. Further, some of the probiotic bacteria occur mainly as dispersed bacterial cells while others form micro-colonies. For control of many diseases, the gut probiotic bacterial population is seriously jeopardized and require urgent replacement. The exogenous administration of Lactobacilli through probiotic capsules does increase the levels of loosely adherent Lactobacillus cells to some extent. However, administration of such probiotics is not often able to establish themselves as an integral part of gastro-intestinal biota and specially the biofilm. Some dietary supplementation like inulin-type fructans could offer a better alternative to stabilize the gut mucosal barrier (Kleessen and Blaut, 2005).
In our study, cucurbits have been shown to render positive effects on the biofilm (barrier) formation and augmentation of antimicrobial activity of L. rhamnosus, reduce inflammation and enhance immunity. Thus a common dietary substitution could be used as a sitotherapeutic agent for digestive disorders.
CONCLUSION
The present study explicitly reveals that dietary cucurbits, investigated here, have appreciable antioxidant activities and support the growth of L. rhamnosus and its antimicrobial activity. Therefore, these varieties of plants can be very useful in averting the free radical induced damage and enhancing immuno-competence and reducing inflammation mediated pathologies. However, in vivo study would be necessary before a formulation of some prophylactic, therapeutic and medicinal products from these cucurbits.
ACKNOWLEDGMENTS
The authors are thankful to Life Science Research Board, Defense Research and Developmental Organization, Ministry of Defense, Govt. of India for giving financial support through project No. DLS/81/48222/LSRB-195/ID/2009 under which, this study has been carried out. The authors are also thankful to the management of Amity University, Noida (U.P.) for providing all necessary infrastructure and facilities for conducting this study.
REFERENCES
- Amdekar, S., V. Singh, P. Roy, S. Kushwaha and D. Dwivedi, 2010. In vitro antibacterial activity of Lactobacillus casei against enteropathogens. WMC Microbiol., 1: 698-702.
Direct Link - Arora, R., M. Kaur and N.S. Gill, 2011. Antioxidant activity and pharmacological evaluation of Cucumis melo var. agrestis methanolic seed extract. Res. J. Phytochem., 5: 146-155.
CrossRefDirect Link - Holder, I.A. and S.T. Boyce, 1994. Agar well diffusion assay testing of bacterial susceptibility to various antimicrobials in concentrations non-toxic for human cells in culture. Burns, 20: 426-429.
CrossRefDirect Link - Devi, G.S., A.K. Muthu, D.S. Kumar, S. Rekha, M. Indhumathi and R. Nandani, 2009. Studies on the antibacterial and antifungal activities of the ethanolic extracts of Luffa cylindrica (Linn) fruit. Int. J. Drug Dev. Res., 1: 105-109.
Direct Link - Dhiman, K., A. Gupta, D.K. Sharma, N.S. Gill and A. Goyal, 2012. A review on the medicinally important plants of the family cucurbitaceae. Asian J. Clin. Nutr., 4: 16-26.
CrossRefDirect Link - Elzaawely, A.A. and S. Tawata, 2012. Antioxidant activity of phenolic rich fraction obtained from Convolvulus arvensis L. leaves grown in Egypt. Asian J. Crop Sci., 4: 32-40.
CrossRefDirect Link - Erasto, P. and Z.H. Mbwamb, 2009. Antioxidant activity and HPTLC profile of Lagenaria siceraria fruits. Tanzania J. Health Res., 11: 79-83.
Direct Link - Gill, N.S. and M. Bali, 2011. Isolation of anti ulcer cucurbitane type triterpenoid from the seeds of Cucurbita pepo. Res. J. Phytochem., 5: 70-79.
CrossRef - Goel, H.C., M. Bala, J. Prasad, S. Singh, P.K. Agrawal and R.C. Swahney, 2006. Radioprotection by Rhodiola imbricata in mice against whole-body lethal irradiation. J. Med. Food, 9: 154-160.
PubMed - Huang, D., B. Ou and R.L. Prior, 2005. The chemistry behind antioxidant capacity assays. J. Agric. Food Chem., 53: 1841-1856.
CrossRefPubMedDirect Link - Irshad, M., I. Ahmad, H.C. Goel and M.M.A. Rizvi, 2010. Phytochemical screening and high performance TLC analysis of some cucurbits. Res. J. Phytochem., 4: 242-247.
CrossRefDirect Link - Jones, S.E. and J. Versalovic, 2009. Probiotic Lactobacillus reuteri biofilms produce antimicrobial and anti-inflammatory factors. BMC Microbiol., Vol. 9.
CrossRefDirect Link - Karagozler, A.A., B. Erdag, Y.C. Emek and D.A. Uygun, 2008. Antioxidant activity and proline content of leaf extracts from Dorystoechas hastata. Food Chem., 111: 400-407.
CrossRefDirect Link - Knekt, P., J. Kumpulainen, R. Jarvinen, H. Rissanen and M. Heliovaara et al., 2002. Flavonoid intake and risk of chronic diseases. Am. J. Clin. Nutr., 76: 560-568.
PubMedDirect Link - Kullisaar, T., M. Zilmer, M. Mikelsaar, T. Vihalemm, H. Annuk, C. Kairane and A. Kilk, 2002. Two antioxidative Lactobacilli strains as promising probiotics. Int. J. Food Microbiol., 72: 215-224.
PubMed - Leman, T., H.A. Kayali and R.O. Urek, 2007. In vitro antioxidant properties of Cucurbita pepo L. male and female flowers extracts. Plant Foods Hum. Nutr., 62: 49-51.
CrossRefDirect Link - Milan, P.B., D.M. Nejad, A.A. Ghanbari, J.S. Rad and H.T. Nasrabadi et al., 2011. Effects of Polygonum aviculare herbal extract on sperm parameters after EMF exposure in mouse. Pak. J. Biol. Sci., 14: 720-724.
CrossRefDirect Link - Moussa, S.A., 2008. Oxidative stress in diabetes mellitus. Rom. J. Biophys., 18: 225-236.
Direct Link - Olsvik, P.A., T. Kristensen, R. Waagbo, B.O. Rosseland, K.E. Tollefsen, G. Baeverfjord and M.H. Berntssen, 2005. mRNA expression of antioxidant enzymes (SOD, CAT and GSH-Px) and lipid peroxidative stress in liver of Atlantic salmon (Salmo salar) exposed to hyperoxic water during smoltification. Comp. Biochem. Physiol. C Toxicol. Pharmacol., 141: 314-323.
PubMed - Oskoueian, E., N. Abdullah, W.Z. Saad, A.R. Omar and S. Ahmad et al., 2011. Antioxidant, anti-inflammatory and anticancer activities of methanolic extracts from Jatropha curcas Linn. J. Med. Plants Res., 5: 49-57.
CrossRefDirect Link - Oyaizu, M., 1986. Studies on products of browning reaction: Antioxidative activities of products of browning reaction prepared from glucosamine. Jpn. J. Nutr. Diet., 44: 307-315.
CrossRefDirect Link - Pattanayak, S.P., P.M. Mazumder and P. Sunita, 2012. Total phenolic content, flavonoid content and In vitro antioxidant activities of Dendrophthoe falcata (L.f.) ettingsh. Res. J. Med. Plant, 6: 136-148.
CrossRefDirect Link - Premalatha, M. and P. Dhasarathan, 2011. Probiotic action of lactobacillus isolated from the milk sample against some human pathogens. Res. J. Immunol., 4: 31-37.
CrossRefDirect Link - Raja, A., P. Prabakaran and P. Gajalakshmi, 2010. Isolation and screening of antibiotic producing psychrophilic actinomycetes and its nature from rothang hill soil against viridans Streptococcus sp. Res. J. Microbiol., 5: 44-49.
CrossRefDirect Link - Saha, P., S.K. Sen, A. Bala, U.K. Mazumder and P.K. Haldar, 2011. Evaluation of anticancer activity of Lagenaria siceraria aerial parts. Int. J. Cancer Res., 7: 244-253.
CrossRefDirect Link - Sahu, R.K., A. Jain and S. Nayak, 2011. Momortia charantia linn: A mini review. Int. J. Biomed. Res., 2: 579-587.
Direct Link - Saide, J.A.O. and S.E. Gilliland, 2005. Antioxidative activity of lactobacilli measured by oxygen radical absorbance capacity. J. Dairy Sci., 88: 1352-1357.
CrossRefDirect Link - Schetter, A.J., N.H.H. Heegaard and C.C. Harris, 2010. Inflammation and cancer: Interweaving microRNA, free radical, cytokine and p53 pathways. Carcinogenesis, 31: 37-49.
CrossRefDirect Link - Shah, B.N., A.K. Seth and R.V. Desai, 2010. Phytopharmacological profile of Lagenaria siceraria: A review. Asian J. Plant Sci., 9: 152-157.
CrossRefDirect Link - Sharaf, E.F. and R.M. Al Harbi, 2011. Isolation, Identification and antimicrobial activity of some local isolates of lactic acid bacteria. Res. J. Microbiol., 6: 826-838.
CrossRefDirect Link - Shimada, K., K. Fujikawa, K. Yahara and T. Nakamura, 1992. Antioxidative properties of xanthan on the autoxidation of soybean oil in cyclodextrin emulsion. J. Agric. Food Chem., 40: 945-948.
CrossRefDirect Link - Smith, K., A. Perez, G. Ramage, D. Lappin, C.G. Gemmell and S. Lang, 2008. Biofilm formation by Scottish clinical isolates of Staphylococcus aureus. J. Med. Microbiol., 57: 1018-1023.
CrossRef - Torkey, H.M., H.M. Abou-Yousef, A.Z. Abdel Azeiz and H.E.A. Farid, 2009. Insecticidal effect of cucurbitacin E glycoside isolated from Citrullus colocynthis against Aphis craccivora. Aus. J. Basic Applied Sci., 3: 4060-4066.
Direct Link - Umadevi, P., S. Murugan, S.J. Suganthi and S. Subakanmani, 2011. Evaluation of antidepressant like activity of cucurbita pepo seed extracts in rats. Int. J. Curr. Pharma. Res., 25: 108-113.
Direct Link - Wijeratne, S.S.K., S.L. Cuppett and V. Schlegel, 2005. Hydrogen peroxide induced oxidative stress damage and antioxidant enzyme response in Caco-2 human colon cells. J. Agric. Food Chem., 53: 8768-8774.
CrossRefDirect Link - Brand-Williams, W., M.E. Cuvelier and C. Berset, 1995. Use of a free radical method to evaluate antioxidant activity. LWT-Food Sci. Technol., 28: 25-30.
CrossRefDirect Link - Onasanwo, S.A., N. Singh, A.A. Saba, A.A. Oyagbemi, O.A. Oridupa and G. Palit, 2011. Anti-ulcerogenic and in vitro antioxidant activities of Lagenaria breviflora (LB) whole fruit ethanolic extract in laboratory animals. Pharmacognosy Res., 3: 2-8.
CrossRefDirect Link








