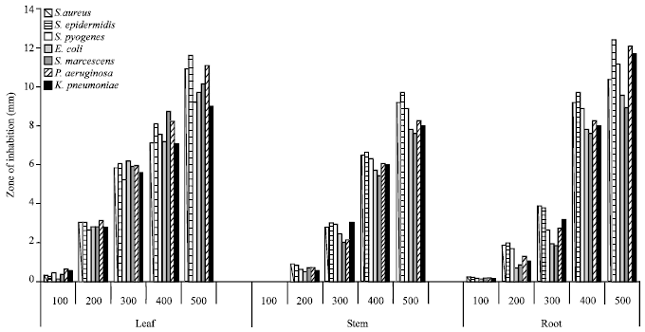
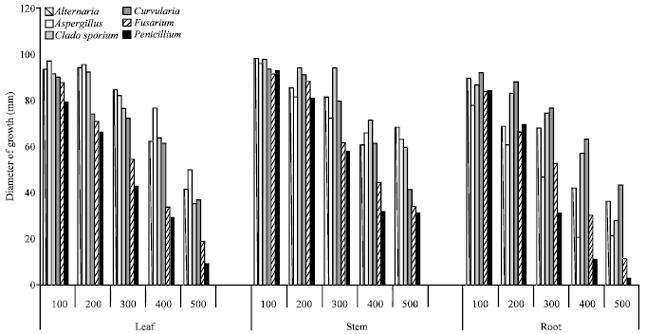
Research Article
Phytoconstituents and Their Influence on Antimicrobial Properties of Morinda citrifolia L.
R & D Centre, Robust Materials Technology Pvt. Ltd., Bengaluru, 560072, Karnataka, India
Parul Chatly
R & D Centre, Robust Materials Technology Pvt. Ltd., Bengaluru, 560072, Karnataka, India
Sayak Adhikari
R & D Centre, Robust Materials Technology Pvt. Ltd., Bengaluru, 560072, Karnataka, India
K.R. Ravikumar
R & D Centre, Robust Materials Technology Pvt. Ltd., Bengaluru, 560072, Karnataka, India