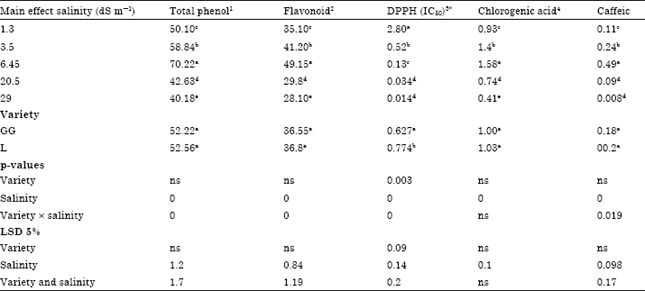
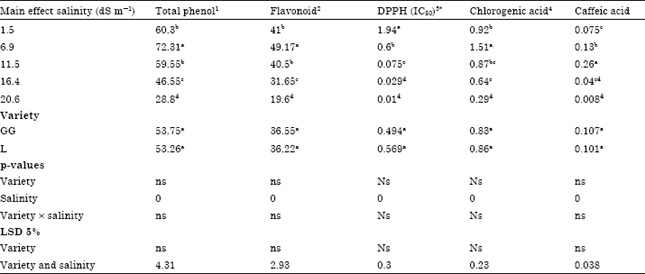
Research Article
Effect of Salinity on Phenolic Composition and Antioxidant Activity of Artichoke (Cynara scolymus L.) Leaves
Department of Horticulture, Gorgan University of Agricultural Sciences and Natural Resources, Gorgan, Iran
Azim Ghasemnezhad
Department of Horticulture, Gorgan University of Agricultural Sciences and Natural Resources, Gorgan, Iran
Mojtaba Barani
Department of Soil Science, Gorgan University of Agricultural Sciences and Natural Resources, Gorgan, Iran
Telmah Telmadarrehei
Department of Plant Protection, College of Agronomic Sciences, Sari Agricultural Sciences and Natural Resources University, Sari, Iran