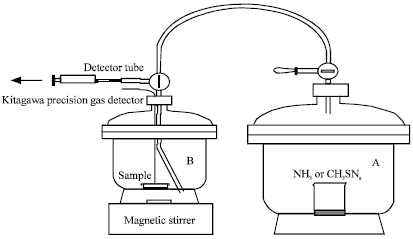
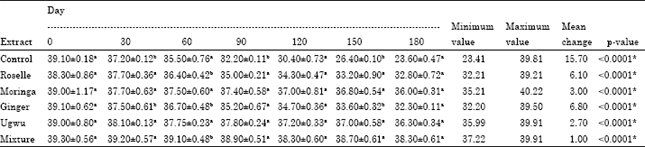
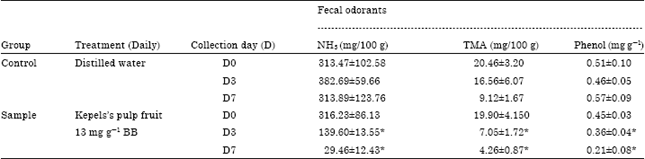
Research Article
Indonesian Kepel Fruit (Stelechocarpus burahol) as Oral Deodorant
Department of Anatomy, Physiology and Pharmacology, Faculty of Veterinary Medicine, Bogor Agricultural University, Indonesia
M. Rahminiwati
Department of Anatomy, Physiology and Pharmacology, Faculty of Veterinary Medicine, Bogor Agricultural University, Indonesia
S. Sadiah
Department of Anatomy, Physiology and Pharmacology, Faculty of Veterinary Medicine, Bogor Agricultural University, Indonesia
I. Batubara
Biopharmaca Research Center, Bogor Agricultural University, Jl Taman Kencana No. 3 Bogor, 16151, Indonesia
L.K. Darusman
Biopharmaca Research Center, Bogor Agricultural University, Jl Taman Kencana No. 3 Bogor, 16151, Indonesia
T. Mitsunaga
Faculty of Applied Biological Science, Gifu University, 1-1 Yanagido, Gifu, 501-1193, Japan