Research Article
Trypanocidal Activity of Some Sudanese Medicinal Plants against Experimental Trypanosoma evansi Infection
Central Veterinary Research Lab, Department of Biochemistry Nutrition and Toxicology, P.O. Box 8067, Alamarat, Khartoum, Sudan
Trypanosomosis is a group of diseases caused by flagellated protozoan parasites of the genus Trypanosoma, family Trypanosomatidae. They are widely distributed in Africa, South America, Asia and Middle East (Molyneux and Ashford, 1983). Trypanosoma evansi cause a disease referred to as Surra. It’s an important disease of livestock in Africa and Asia causing great economic losses in camels and water buffalos. Trypanosoma evansi is mechanically transmitted during feeding of blood sucking Diptera especially Tabanid flies (Leach and Robberts, 1981).
Trypanosomosis is a complex disease that directly and indirectly has impact on crop and livestock agricultural development and hence represents a major constraint to socio-economic development in areas affected (FAO, 1988).
Chemotherapy, by stopping the multiplication of the trypanosomes helps the immune system to overcome the infection. Treatment will be more effective in well-fed and rested animals, in which the immune system is not adversely affected by stress and lack of food. The management of African Animal Trypanosomosis (AAT) at farmer’s level has been predominately dependent on the use of the trypanocidal drugs (Diminazine, Homidium and Isometamidium). It is estimated that, about 35 million doses per year are used in Africa to cure the disease (Peregrine and Mamman, 1993).
Since 1961, no additional drugs for use against animal trypanosomosis have gone beyond the experimental stage. Drug resistance between diamidines and isometamidium group seems to exist (Williamson, 1979). At the recommended dose Cymelarsan® has been shown to be well tolerated (Biswas and Hunter, 1993). Control of the disease is based mainly on control of vectors whereas the seasonality f their abundance and patchy distribution make it difficult to adopt efficient programs for their control. Chemotherapy is however, the most commonly recommended method of disease control. Limited successes had been achieved, despite enormous effort by several workers in the field of chemotherapy and allied disciplines to discover and develop an ideal trypanocide (Jennings et al., 1993). In camels, a number of drugs have been tried for trypanosomosis caused by T. evansi. However, the extensive and long term use of small numbers of commercially available trypanocides result 4d in the appearance of drug resistance (Lang, 1985).
Herbal plants (cheaper availability and with less or no side effects) have emerged as a potential candidate (Karim et al., 2011). Plants used in indigenous medicine are considered to be potential source of development of alternative therapeutics (Cox and Balick, 1994). Since herbal treatment for various diseases in Africa is still wide spread (Anokbonggo, 1992), ethnobotonical approach in collaboration with traditional healers may prove to be a rich source of drug discovery in Veterinary medicine, Ishtiaq et al. (2006). O’Neill and Lewis (1993) stated that close to half the world’s best selling pharmaceuticals were either natural products or their derivatives. Therefore, investigation of natural remedies as a source of new drugs gained great interest in recent years. Some naturally occurring chemical compounds serve as models for a large percentage clinically proven drugs, and many are now being re-assessed as antimicrobial agents (Mahady et al., 2008). About 39% of the new drugs discovered during the period between 1983-1994 were either natural products or derivatives of them (Harvey, 2001).
Many natural plants of plant origin, with a wide range of different chemical structure, have been reported to have activities against different species of protozoan parasites including, Plasmodium, Trypanosoma, Leishmania and Entaoeba (Raz, 1998). In vitro anti-plasmodial effect of Crude Extracts of Diospyros melanoxylon was studied by Saxena et al. (2011). The in vitro antitrypanosomal activity of some African medicinal plants was found to have antitrypanosomal activity (Oliver-Bever, 1986; Assi and Guinko, 1991). Rahman et al. (2010) evaluate the potential of Argemone mexicana extract as antimicrobial agent against bacterial isolates originated from drinking water. Sudan is rich with plants used as herbal treatment. Elhardallou (2011) studied the cytotoxicity and biological activity of many Sudanese medicinal plants.
This work had been conducted to evaluate trypanocidal activity of certain plant extracts compared to standard drugs. It was also carried out to assess trypanocidal activity claimed in tradition medicine for certain plant extract in vivo using biological models.
Animals: Swiss albino mice (Mus domesticus), Swiss albino rats (Albino Wister) were obtained from the laboratory of experimental animals, unit of the central veterinary research laboratories-Suba, Khartoum Sudan. They were housed in laboratory cages, fed with pellets and fresh vegetables and were watered ad libitum throughout the experimental period.
Trypanosome and infection: The parasite was isolated from naturally infected camels at Abuzeid livestock market-Omdurman town. Infected blood was inoculated into a mouse for propagation. The blood of the infected mouse was cryopreserved in liquid nitrogen. The rapid matching wet-examination technique described by Herbert and Lumsden (1976) was used by examining a drop of mouse blood under the 40x magnification of a microscopic and counting the number of Trypanosoma in each field and matched with log figure obtained from true the reference table. Trypanosomes were injected I/P at dose of 5x105.
The plants: The tree plants tested in the preset study were selected on the basis of information from traditional healers on their curative effect in the treatment of malaria or sleeping sickness.
Tinospora bakis (A. Rich) Miers in Hook. Niger EL: 215 (1849) family Menispemaceae was colleted from the Angasana hills in Eastern South of Sudan.
| • | Argemone maxicana of the family Papaveraceae was collected from khor Abuanga, Omdurman |
| • | Aristolachia bracteolate of the family Arsitolochiaceae was collected from juba region |
| • | Collection and identification of the plants was carried out at the Medicinal and Aromatic Plant Research Institute (MAPRI), Khartoum-Sudan |
The extracts: The plants were extracted for primary in vivo evaluation screening by extracting 20 g of dried coarsely powdered entire plants. Plants materials were successively extracted with chloroform, methanol and distilled water by percolation. A ten folds quantity of solvent in relation to the plant material was used for the extraction. For each solvent, extraction was performed three times at room temperature each time for 4 h. Thus, three extracts of increasing polarity were obtained from the plant. All extracts were filtered through or filter paper, the filtrates were then concentrated on a rotary evaporator ay 35°C under pressure and then dried. The solid extract obtained was removed, weighted and was kept as the stock solution for use. The methanolic extract was dissolved in distilled water while the chloroformic extract was dissolved in propylene glycol.
Experimental design: Groups of 10 rats each were used; they were aged 4-6 weeks, weighted 125-150 g and were divided as follows:
| Group1 | : | Infected untreated control |
| Group 2 | : | Infected ad treated with 0.25 mg kg-1 of Cylmalersan S/C |
| Group 3 | : | Infected and treated with 100 mg kg-1 BW of plant methanolic extract |
| Group 4 | : | Infected and treated with 250 mg kg-1 of plant methanolic extract |
| Group 5 | : | Infected and treated with 500 mg kg-1 of plant methanolic extract |
| Group 6 | : | Infected and treated with 100 mg kg-1 BW of plant chlorophormic extract |
| Group 7 | : | Infected and treated with 250 mg kg-1 BW of plant chlorophormic extract |
| Group 10 | : | Infected and treated with 500 mg kg-1 BW of plant chlorophormic extract |
The plant extract was given orally using nasogastric tube.
Sampling: Parasitaemia was checked daily for 30 days by examining a drop of blood obtained from each rat by cutting the tip of the tail with scissor tail disinfected with 70% ethanol.
Blood from ocular vein was collected once a week tills the end of the experiment for serum. The serum was kept at -20°C for analysis. Animals were dissected immediately after death or at the end of the experiment.
Cymelarsan was used as a standard drug in this experiment at a dose rate of 0.25 mg kg-1 BW. It was clear that there was an immediate cure as from the second day of treatment. All the rats became aparasitaemic till the end of the treatment period with percentage clearance 100%. The result of Tinospora bakis was shown in Table 1. The best effect of the extract was given with the high dose 500 mg kg-1 BW either with the methanolic or chloroformic extract. The initial trypanosome clearance occurred on day 8 with chloroformic extract and on day 10 with methanolic extract with percentage clearance of 50%. Relapse occurred between 10-15 day of treatment.
Argimone mexicana extract gave better result than Tinospora bakis where clearance of the parasite occurred with dose 250 mg kg-1 BW (Table 2). Initial trypanosome clearance occurred on day 8 with both extracts. The high dose 500 mg kg-1 BW has a good result where the initial trypanonome clearance occurred on day five with chloroformic extract and day 7 with methanolic extract. The percentage of the aparacitaemic rats was 60% with methanolic extract and 70% with chloroformic extract. Relapse occurred between 12-16 day of treatment.
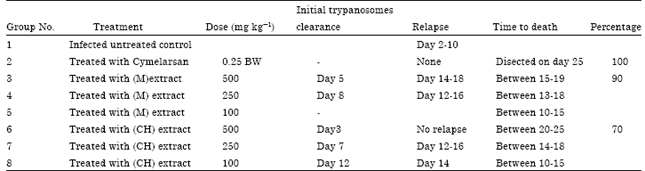
Aristolochia bracteolate extract gave a very good result especially whem given at the dose of 500 mg kg-1 BW of chloroformic extract (Table 3). The initial trypanosome clearance occurred on day 3 treatment and no relapse occurred till the end of the treatment period. The percentage of the aparasitaemic rats was 90%. On the other hand the 500 mg kg-1 BW of the methanolic extract gave an initial trypanosome clearance on day 5 with percentage 70% but relapse occurred between 14-18 day of treatment. The medium dose which is 250 mg kg-1 BW gave initial trypanosome clearance on day 7 with chloroformic extract and on day 8 with methanolic extract with percentage clearance of 60% for both extracts. Relapse occurred between 12-16 day of treatment.
| Table 1: | Antitrypanosomal activity of Tinospora bakis extracts compared to cymelarsan |
 | |
| Each group was composed of 10 rats each. The parasite was given at a dose rate of 5x105. M: Methanolic extract. CH: Chloroformic extract | |
| Table 2: | Antitrypanosomal activity of Argimone mexicana extracts compared to cymelarsan |
 | |
| Each group was composed of 10 rats each. The parasite was given at a dose rate of 5x105. M: Methanolic extract. CH: Chloroformic extract | |
| Table 3: | Antitrypanosomal activity of Aristolochia bracteolate extracts compared to cymelarsan |
 | |
| Each group was composed of 10 rats each. The parasite was given at a dose rate of 5×105. M: Methanolic extract. CH: Chloroformic extract | |
Of the three plants used in this study, Aristolochia bracteolata gave the best result. The smallest dose used which is 100 mg kg-1 BW has no effect on clearing T. evansi for the three plants. There was decrease in parasitaemia count but without clearance of the parasite.
The search for an active trypanocidal drug from a plant origin is a concern of many researchers. The study gave an indication of the activity of the three plants used with variation in the trypanocidal effect. For Tinospora bakis only the high dose for both methanolic and chloroformic extract appeared to clear the parasite although clearance was only 50%. The lower doses of the plant caused general decrease in the mean parasitaemia count without clearance from blood. This suggested that plant extract might clear the parasite from the blood if the concentration of the plant was increased. Plant extracts were always produced from dried plant material which may not be the procedure used traditionally and so disintegration of unstable bioactive compounds might have occurred. In many cases, plants are used in combination with others which may give rise to synergistic effects. The result of T. bakis was found to be similar to that obtained by De-Mesquita et al. (2005). They stated that T.bakis has a trypanocidal activity against T. cruzi. The result also found to resemble the result obtained by Ouattara et al. (2006). Who found that alkaloidal extracts from the roots of T. bakis has antimalarial activity.
On the other hand, Argimone mexicana extract produced a significant trypanocidal effect in rats infected with T. evansi although it didn’t completely eliminate the parasite from blood. It was observed that the chloroformic extract of the plant gave better result than that of the methanolic extract at the same dose either 250 or 500 mg kg-1 BW. With the chloroformic extract clearance of the parasite occurred on 60% of the rats used at the dose 250 mg kg-1 BW and 50% with methanolic extract. The high dose which is 500 mg kg-1 BW caused clearance percentage of 60% of the infected rats with methanolic extract and 70% with chloroformic extract. This result indicated that both methanolic and chloroformic extracts of A. mexicana produced significant trypanocidal activity. This result agreed with that of Nok et al. (1994) who studied the effect of Cannabis sativa, which is related to the family of A. mexicana. They found that the aqueous extract of the seeds of C. sativa when administered by injection at a dose of 50 mg kg-1 BW/day for five consecutive days, cured rats infected experimentally with T. brucei. The result of A. mexicana concurred with that of Freiburghaus et al. (1996) who studied the trypanocidal effect of A. mexicana in vitro. They found that none of the extracts of A. mexicana exhibited activity against T. brucei rhodeinse. This difference in between the present result and the other authors, may have been due to the fact that they used an in vitro test, where the metabolic transformation of inactive molecules to active ones or vice versa, may occur and thus activity might change under in vivo conditions.
Aristolochia bracteolate extracts produced the best result of the three plants used. The highest activity was found with the chloroformic extract especially when given at the dose rate of 500 mg kg-1 BW. The clearance occurred in 90% of the infected rats and no relapse occurred till the end of treatment period. With methanolic extract at the same dose, trypanosmes clearance occurred in 70% but relapse occurred on day 15. The high level of activity displayed by the chloroformic extract, as compared to the methanolic extract, indicates that the chloroform might be capable to extract the biological active principle (s) responsible for the trypanocidal effect of the plants used. Most of the work done in A. bracteolata was conducted to its effect on Plasmodium species which also blood parasites. Many workers found A. bracteolate to be effective against plasmodium species such as El-Tahir et al. (1999) who studied the in vitro effect of A.bracteolata against P. falciparum and found it effective. Ahmed et al. (2010) used the whole plant of A.bracteolate for screening against P. falciparum and found it to be effective. Almost every part of the plant have medicinal usage. Identifying bioactive compounds and establishing their health effects are active areas of scientific enquiry (Kris-Etherton et al., 2004).
In conclusion, both methanolic and chlorformic extracts of either Aristolochia bracteolate or Argimone mexicana produced significant dose-dependent Trypanocidal activity in rats experimentally infected with Trypanosoma evansi. The trypanocidal activity was more pronounced when the extract was administered at the higher dose (500 mg kg-1 BW). The use of medicinal plants are mostly applied by traditional healers for treatment of diseases, therefore, the knowledge of the information of traditional healing from people should be documented since the healthcare system of people should be negatively affected (Cheikhyoussef et al., 2011).
The Financial Support of the Animal Resources Research Corporation is gratefully acknowledged. The Medicinal and Aromatic Plants Research Institute (MAPRI), National Centre for Research Ministry of Science and Technology.
Samia Hussein Abdelrahman Reply
Thank you very much for publishing my article in Science Alert which has a very good reputation for the good quality of articles they accept and the prcise way of correction and good system they follow.