Research Article
Protective Effect of Avicennia alba Leaves Extract on Gastric Mucosal Damage Induced by Ethanol
Department of Biological Sciences, Faculty of Sciences, King Abdul Aziz University, P.O. Box 139109, Jeddah 21323, Saudi Arabia
INTRODUCTION
Gastrointestinal diseases are widespread among the inhabitants of many countries. Peptic ulcers are a common disorder of the entire gastrointestinal tract that occur mainly in the stomach and the proximal duodenum. Peptic ulcer is a major health hazard both in terms of morbidity and mortality. The incident rate of peptic ulcer has increased in recent years (Szabo and Vincze, 2000). Peptic ulcer disease is a chronic inflammatory disease characterized by ulceration in the regions of upper gastrointestinal tract where parietal cells are found and where they secrete Hydrochloric Acid (HCl) and pepsin. The anatomic sites where ulcer occurs commonly are stomach and duodenum, causing gastric and duodenal ulcer, respectively (Rang et al., 2003). Despite great advances in the understanding of the peptic ulcer illness, its etiology has not been completely elucidated. The basic physiopathological concept is that the peptic ulcer results from the imbalance between the mucosal defensive factors [mucus, bicarbonate secretion, prostaglandins, blood flow and the process of restitution and regeneration after cellular injury] and offensive factors [HCl-pepsin secretion, Helicobacter pylori, refluxed bile, increased free radicals and decreased antioxidants] (Bandyopadhyay et al., 2001; Bhattacharjee et al., 2002; Tulassay and Herszényi, 2010). Ulcerative lesions of the gastrointestinal tract are one of the major side effects associated with the use of Non-Steroidal Anti-Inflammatory Drugs (NSAIDS), alcohol, stress and ischemic reperfusion (Mizui et al., 1987; Kamsiah et al., 2005; Bahrami and Ali, 2010; Guldur et al., 2010). Although recent advances in our understanding have highlighted the multi-factorial pathogenesis of peptic ulcers, secretion of gastric acid is still recognized as a central component of this disease (Luiz-Ferreira et al., 2010).
A number of anti-ulcer drugs like gastric anti-secretory drugs-H2 receptor antagonists, anti-muscarinic agents, proton pump inhibitors, mucosal protective agents-carbenoxolone sodium, sucralfate and prostaglandin analogues are available which are shown to have side effects and limitations. Moreover, various reports have shown that commonly used drugs for peptic ulcers have danger of drug interaction, adverse effect and increased incidence of relapses during ulcer therapy (Bandyopadhyay et al., 2002; Goel and Sairam, 2002; Rao et al., 2004). In this context, the use of medicinal plants for the prevention and treatment of different pathologies is in continuous expansion all over the world (Mota et al., 2009). Medicinal plants are valuable natural resource and regarded as potentially safe drugs. Numerous natural products derived from plant sources have been evaluated as therapeutics for the treatment of various ailments like dysentery, influenza, vaginitis, tumors, diabetes, diuretics, jaundice, kidney stone, dyspepsia, anti-hepatotoxic, anti-hepatitis-B, anti-hyperglycemic and also as anti-viral and anti-bacterial (Ahmad et al., 2007; De Sousa Falcao et al., 2008). Natural products are gaining space and importance in the pharmaceutical industry as well as inspiring the search for new potential sources of bioactive molecules (Schmeda-Hirschmann and Yesilada, 2005). The anti-ulcerogenic activity of many plant products is reported due to an increase in mucosal defensive factors rather than decrease in the offensive factors (Dharmani et al., 2004; Narayan et al., 2004; Khushtar et al., 2009; Bahrami and Ali, 2010; Guldur et al., 2010).
Mangroves are widespread in tropical and sub tropical regions, growing in the saline intertidal zones of sheltered coastlines. The ability to survive in mangrove habitats, characterized by high salt concentrations, low aeration of waterlogged soil and frequently changing water levels due to tidal cycles, has clearly evolved several times independently within angiosperms (Ricklefs and Latham, 1993). Tomlinson (1986) grouped plants that occur in mangrove habitats into three categories, major, minor and associates, based upon the degree to which they are restricted to these habitats and their importance in these communities. Mangrove plants are containing biologically active anti-viral, anti-bacterial and anti-fungal compounds (Bhattacharya et al., 2003). The presence of compounds like tannins, alkaloids and polyphenols in mangrove plants which play an important role in the suppression of deleterious microorganisms (Ross et al., 1980; Nishiyama et al., 1987; Jamale and Joshi, 1998). Furthermore, mangrove plant extracts have been used for centuries as popular method for treating several health disorders plant-derived substances have recently become of great interest owing to their versatile applications. Avicenniaceae family is a member of true mangrove plants which has one genus (Avicennia), 11 species and several subspecies. Plants of Avicennia are trees and woody shrubs distributed in coastal and estuarine habitats in tropical and subtropical areas worldwide (Duke, 1991). Avicennia is considered a major or ‘>true mangrove’= element; these plants are endemic to mangrove habitats, play a predominant role in community structure and have the ability to form pure stands (Tomlinson, 1986). Additionally, Avicennia is the most species-rich and most frost tolerant of all mangrove genera; it is one of only two true mangrove genera that are distributed along coastal habitats in both the New and Old World. The taxonomic placement of Avicennia is contentious. In some classifications it has been placed in the family Verbenaceae, but more recently has been placed by some botanists in the monogeneric family Avicenniaceae. Recent phylogenetic studies have suggested that Avicennia is derived from within Acanthaceae and the genus is included in that family in the Angiosperm Phylogeny Group system. Avicennia alba is one of the most current species among these plants in Southeast Asia mangrove forests. Vadlapudi and Naidu (2009) showed that the plant extracts of A. alba have greater potential as anti-microbial compounds against microorganisms and that they can be used in the treatment of infectious diseases caused by resistant pathogenic. Ito et al. (2000) studied the chemical constituents of A. alba and identified three new naphthoquinones and their analogues, named avicequinone-A (1), -B (2), -C (3) and avicenol-A (4), -B (5), -C (6). Furthermore, Itoigawa et al. (2001) investigated the cancer chemopreventive activity of naphthoquinones and their analogs from Avicennia plant including A. alba and they reported that six natural and four synthetic naphthoquinones and five of their analogs were tested for their inhibitory activities against Epstein-Barr virus early antigen activation induced by 12-O-tetradecanoylphorbol-13-acetate in Raji cells. Some of the 1,4-naphthoquinones and their analogs were found to show remarkably potent activities, without showing any cytotoxicity. 1,4-Furanonaphthoquinone and its analog isolated from Avicennia plants having an alcoholic OH group on the dihydrofuran-ring, displayed the most potent activity. Furthermore, avicenol-A exhibited a marked inhibitory effect on mouse skin tumor promotion in an in vivo two-stage carcinogenesis test. Additionally, they reported that some of these 1,4-naphthoquinones and their analogs might be valuable as potent cancer chemopreventive agents. Kim et al. (2003) demonstrated that the naphthoquinone analog, termed 2,3-dichloro-5, 8-dihydroxy-1,4-naphthoquinone (DDN), induces apoptosis in human promyeloid leukemic HL-60 cells and shows anti-tumor activity in vivo. Moreover, Suntar et al. (2010) showed that the naphthoquinones and some flavonoids (hyperoside, isoquercitrin, rutin and epicatechin) possess remarkable wound healing and anti-inflammatory activities. With this information, the extract of A. alba leaves has been evaluated in the present study to find out its possible effects against ethanol induced gastric mucosal damage in female rats.
Plant material and extraction: Fresh leaves of A. alba were directly collected from mangrove area of Langkawi Island, Malaysia in September, 2010. The leaves were air-dried at room temperature and stored in dry plastic container until use for extract preparation. Two grams of leaves were powdered and mixed with 100 mL of cold water for 15 min using an electric blender. This mixture was later used for experimental supplementation at low (100 mg kg-1, BW) and high (300 mg kg-1 BW) doses.
Animals model: Female albino rats of Wistar strain used in this study were obtained from the Experimental Animal Unit of King Fahd Medical Research Center, King Abdul Aziz University, Jeddah, Saudi Arabia. The total number of rats used was 30 ranging in age from 6-7 weeks and weighing between 135-175 g, kept in standard cages at room temperature of 20±1°C, humidity 50-55% and 12 h day/night cycle. The experimental animals were fed standard rat chow and water. This study was conducted according to ethical guidelines of the Animal Care and Use Committee of King Abdul Aziz University.
Experimental treatments: The animals were divided into six groups of five animals each. The animals were fasted for 24 h prior to the experiment. They were allowed free access to water. After 24 h, the water was withdrawn and the groups of rats were then subjected to one of the following treatments:
| • | Group 1: Rats were intragastrically received saline solution and served as control |
| • | Group 2: Rats were intragastrically received 1 mL of 99.9% ethanol, C2H5OH (Scharlab Co., Spain) |
| • | Group 3: Rats were intragastrically pretreated with 100 mg kg-1 BW of A. alba leaves extract and after 1 h they received 1 mL of 99.9% ethanol |
| • | Group 4: Rats were intragastrically pretreated with 300 mg kg-1 BW of A. alba leaves extract and after 1 h they received 1 mL of 99.9% ethanol |
| • | Group 5: Rats were intragastrically treated with 100 mg kg-1 BW of A. alba leaves extract |
| • | Group 6: Rats were intragastrically treated with 300 mg kg-1 BW of A. alba leaves extract |
Blood sampling: After 1 h of the experimental treatments, the animals were sacrificed under ether anesthesia. Blood samples were collected from orbital venous plexus into plain tubes and centrifuged. Blood sera were carefully separated and stored frozen. Serum creatinine, urea, uric acid, alanine aminotransferase (ALT), aspartate aminotransferase (AST) and Gamma Glutamyl Transferase (GGT) were later determined using an automatic analyzer (Reflotron® Plus System, Roche, Germany).
Histological evaluation: The stomach of each rat was removed and opened along the greater curvature and washed in physiological saline solution. Gastric tissues were harvested from all treated animals and the fragments from tissues were fixed in 10% buffered formalin solution for 48 h. The tissues were subjected to the dehydration process through ascending grades of alcohol, cleared in xylene, infiltrated and embedded in paraffin wax. Subsequently, 5 μ sections were stained with haematoxylin and eosin according to routine technique of light microscopy. Qualitative evaluation of prepared tissues and the obtaining of their photos were carried out using Motic digital microscope, DM-B1 series, Motic Company.
Statistical analysis: The statistical differences of all data were determined by the Student’s t-test. All values were expressed as mean±Standard Deviation (SD) for five observations. Statistical probability of less than 0.05 was used as a criterion for significance. All data were evaluated for statistical significance using the Statistical Package for Social Sciences, SPSS for windows, version 12.0.
Serum creatinine, urea, uric acid, ALT, AST and GGT values were used as indexes of kidney and liver functions in control and experimental animals treated with ethanol and A. alba leaves extract as shown in Table 1. Results show that ethanol treatment increased the levels of serum creatinine (+9.4%), ALT (+18.0%), AST (+12.4%) and GGT (45.6%) in-group 2, while the levels of urea and uric acid were statistically unchanged compared with control data. The levels of serum creatinine (+6.3%) ALT (+5.2%) and GGT (+24.7%) were significantly increased in rats treated with low dose of A. alba leaves extract plus ethanol (group 3), while the levels of urea, uric acid, AST and GGT were statistically unchanged compared with control group.
| Table 1: | The values of serum creatinine, urea, uric acid, ALT, AST and GGT of control, ethanol, low dose of A. alba leaves extract (LD) plus ethanol, high dose of A. alba leaves extract (HD) plus ethanol, low dose of A. alba leaves extract (LD) and high dose of A. alba leaves extract (HD) treated rats (n = 5) |
 | |
| *Indicates a significant difference between control and treated groups, Percentage changes are included in parentheses | |
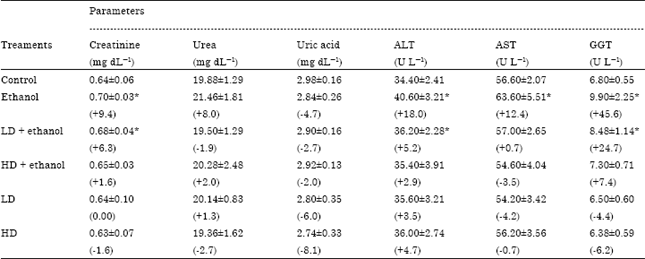
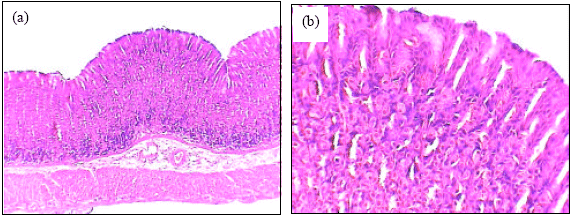
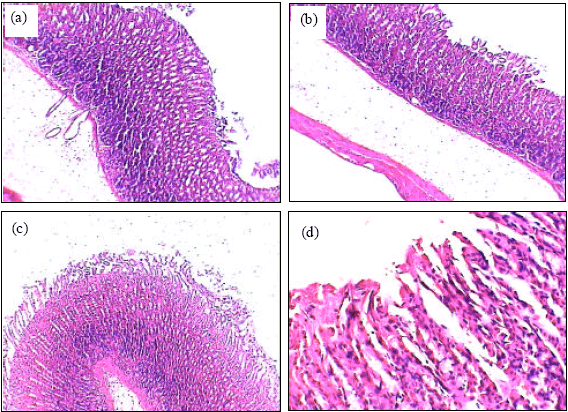
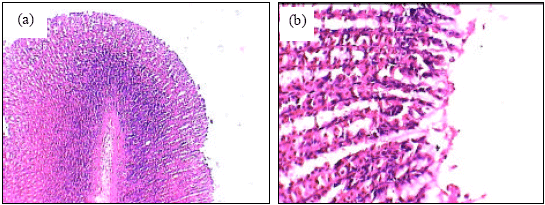
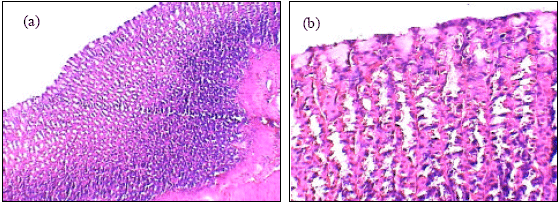
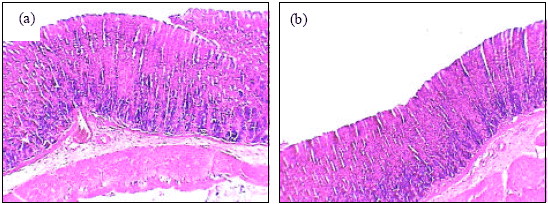
Insignificant changes of all serum parameters were noted in rats treated with high dose of A. alba leaves extract plus ethanol (group 4), low dose of A. alba leaves extract (group 5) and high dose of A. alba leaves extract (group 6). Figure 1(a, b) showed the normal structure of stomach tissues including serosa, muscularis, submucosa and mucosa layers in control group. Ethanol administration resulted in marked gross mucosal damages in rats of group 2 (Fig. 2a-d). Rats pretreatment with low dose of A. alba leaves extract (group 3) showed mild protection against ethanol-induced gastric mucosal damage (Fig. 3a and b). Animals pretreatment with high dose of A. alba leaves extract (group 4) showed significant protection against ethanol-induced gastric mucosal damage (Fig. 4a and b). Figure 5a and b demonstrated the normal structures of stomach in rats treated only with low (group 5) and high (group 6) doses of A. alba leaves extract.
It is generally accepted that alcohol consumption can induce dramatic changes in the physiological and biochemical processes of the whole organism and in the cells (Clemens and Jerrells, 2004; Cook et al., 2004; Poschl and Seitz, 2004; You and Crabb, 2004; Oba et al., 2005). Alcohol is regarded as the most commonly abused drug in the world with profound consequences, both societal and medical (Masters, 2007). In this study, ethanol administration induced severe renal and hepatic injuries evident as elevations of serum creatinine, ALT, AST and GGT values. These results are in agreement with previous experimental studies (Enomoto et al., 2003; Chung et al., 2005; Alsaif, 2007; Hussein et al., 2007; Arda-Pirincci et al., 2009; Habib-ur-Rehman et al., 2009; Das et al., 2010; Chen, 2010; Yurt and Celyk, 2010). The liver is the main organ involved in the metabolism of ethanol, but other extrahepatic tissues, i.e. the kidney, may also contribute to the ethanol metabolism. The enzyme activities of ALT, AST and GGT are considered to be a sign of alcohol abuse and are in general increased with alcoholism (Halmesmaki et al., 1992; Siddiqi et al., 2007). Ethanol associated endotoxaemia and subsequent release of inflammatory mediators may cause hepatocyte injury via oxyradicaldependent and-independent mechanisms. Ethanol manifests its harmful effects either through direct generation of reactive metabolites, including free radical species that react with most of the cell components, changing their structures and functions, or by contributing to other mechanisms that finally promote enhanced oxidative damage (Kato et al., 1990; Nordmann, 1994).
 | |
| Fig. 1: | The histology of stomach of normal female rats (a, X100). Gastric mucosal layer of normal female rats (b, X400) |
 | |
| Fig. 2: | The histology of stomach of ethanol treated female rats (a, b and c X100). Gastric mucosal layer of ethanol treated female rats (d, X400) |
The histopathlogical evaluations in the present study demonstrated that ethanol administration caused severe gastric mucosal damage. Various of experimental investigations showed similar histopathological observations in experimental animals treated with ethanol and NSAID (Coskun et al., 2004; Narayan et al., 2004; Kamsiah et al., 2005; Alhaider et al., 2006; Karumi et al., 2008; Li et al., 2008; Sehirli et al., 2008; Al-Rejaie, 2009; Zhao et al., 2009; Abdulla et al., 2010; Luiz-Ferreira et al., 2010).
 | |
| Fig. 3: | The histology of stomach of low dose of A. alba leaves extract plus ethanol treated female rats (a, X100). Gastric mucosal layer of low dose of A. alba leaves extract plus ethanol treated female rats (b, X400) |
 | |
| Fig. 4: | The histology of stomach of high dose of A. alba leaves extract plus ethanol treated female rats (a, X100). Gastric mucosal layer of high dose of A. alba leaves extract plus ethanol treated female rats (b, X400) |
 | |
| Fig. 5: | The histology of stomach of low dose of A. alba leaves extract treated female rats (a, X100). The histology of stomach of high dose of A. alba leaves extract treated female rats (b, X100) |
Ethanol serves as the most common ulcerogenic agent and when given intragastrically to rats it produces severe gastric hemorrhagic erosions (Shetty et al., 2000). Ethanol induced gastric mucosal injury is associated with extensive damage to mucosal capillaries and increased vascular permeability (Szabo et al., 1985; Bou-Abboud et al., 1988). Mucosal capillary necrosis, vascular congestion and thrombosis in the subepithelial microvasculature accompany disruption of the gastric mucosal barrier. In addition to the direct injurious effects of ethanol on gastric mucosa, other factors are also thought to be involved in the pathogenesis of injury (Konturek et al., 1996).
In the present study, administration of low and high doses of A. alba leaves extract significantly decreased the physiological and histopathological alterations induced by ethanol exposure. In rats treated with low dose of A. alba leaves extract following by ethanol administration, the levels of serum creatinine ALT and GGT were statistically evoked, while the levels of other serum biochemical parameters were notably unchanged compared with corresponding control group. Furthermore, the histopathological analysis of stomach showed that the pretreatment with low dose of A. alba leaves extract reduced the severe changes attributed to ethanol influence. These data confirmed the ameliorative role of low dose of A. alba leaves extract against ethanol-induced physiological and histological disturbances. In addition, the pretreatment with high dose of A. alba leaves extract significantly inhibited the ethanol-induced physiological and histopathological changes in rats, confirming its nephroprotective, hepatoprotective and gastroprotective influence. Moreover, insignificant changes of all physiological parameters and histopathological evaluations were observed in rats treated with only low or high doses of A. alba leaves extract, indicating its safety as selective concentrations in the present study. Thirunavukkarasu et al. (2010) studied the anti ulcer effect of Avicennia officinalis leaves extract in albino rats and they reported that the pretreatment with leaves extract (both hot water and cold water) caused a beneficial effect on NASID-induced gastric ulcer as evidenced by the reduction in the ulcer score. Also, they showed that these finding may be attributed to polyphenolic compounds found in mangrove plants. Moreover, the wound healing capacity of A. officinalis and other plant species such as Rizophora mangle and Excoecaria agallocha during ulcer is due to several mechanisms, such as coating the wound, forming complexes with proteins of cell wall, chelating free radicals and reactive oxygen species, stimulating the concentration of the wound and increasing the formation of new capillaries and fibroblasts (Tsukimi and Okabe, 1994; Perera et al., 2001, 2010; Thirunavukkarasu et al., 2009, 2010).
Considerable scientific evidence suggested that under situations of oxidative stress Reactive Oxygen Species (ROS) such as superoxide, hydroxyl and peroxyl radicals are generated and the balance between antioxidation and oxidation is believed to be a critical concept for maintaining a healthy biological system (Davies, 2000). Oxygen free radicals are implicated in the pathogenesis of ethanol-induced gastric mucosal injury (Szelenyi and Brune, 1988; Hiraishi et al., 1999) apart from other mechanisms such as mucosal leukotriene release (Peskar et al., 1986), submucosal venular constriction (Oates and Hakkineu, 1988). Accumulation of activated neutrophils in the gastric mucosa may be a source for free radicals (Tepperman and Soper, 1990). The ethanol induced gastric mucosal damage was shown to be associated with the significant reduction in the non-protein sulphyldryl concentration in cultured rat gastric mucosa cells (Szabo et al., 1981). Oxygen derived free radicals have been implicated in the pathogenesis of a wide variety of clinical disorders and gastric damage is caused by physical, chemical and psychological factors that leads to gastric ulceration in human and experimental animals (Rao et al., 2000). Recently interest has been focused on the role of ROS in gastroduodenal pathogenesis related to gastric hypersecretion and gastroduodenal mucosal damage. Reports suggest that reactive ROS play an important role in the pathophysiological processes of acute gastric lesions (Parks, 1989; Vaananen et al., 1991; Jainu and Devi, 2004). Although the mechanism of ethanol, NSAIDS and other drug induced gastric lesions is unclear, accumulating neutrophils, oxygen free radicals, inhibition of prostaglandins play a crucial role (Perry et al., 1986; Szelenyi and Brune, 1988; Galvin and Szabo, 1992; Shetty et al., 2000; Liu et al., 2008). Ethanol induced ulcers are found mainly in the glandular part of the stomach are reported to potentiate the formation of leukotriene C4 (LTC4), mast cell secretory products and reactive oxygen species resulting in damage to the rat gastric mucosa (Peskar et al., 1986; Mizui et al., 1987; Oates and Hakkinen, 1988). A copious amount of gastric mucus is secreted during superficial mucosal damage and provides a favorable microenvironment for repair by restitution. An increase in gastric motility, vagal overactivity, mast cell degranulation, free radical generation, decreased gastric mucosal blood flow and decreased prostaglandin synthesis are involved in the production of stress-induced ulcers (Cho et al., 1976; Cho and Ogle,1979; Rao et al., 2000). Ethanol induced ulcers were not inhibited by anti-secretory agents such as cimetidine, but are inhibited by agents that exhibit a gastroprotective action with an antioxidative cytoprotection (Arisawa et al., 2006). Most of the anti-ulcer compounds or extracts are known for their scavenging activities on free radicals in the process of ulcer healing (Onasanwo et al., 2010). Therefore, there is a possibility that the chemical constituents of A. alba leaves extract may possess antioxidative properties to protect the gastric mucosal layer from the severe injury induced by ethanol.
The present findings demonstrated that the leaves extract of A. alba appears to be effective against gastric mucosal damage induced by ethanol treatment. However, the gastroprotective activity of A. alba against ethanol treatment may be due to its effects on both offensive and defensive factors. Additionally, the present investigation might well suggest that the chemical constituents of A. alba leaves extract have nephroprotective, hepatoprotective, gastroprotective and antiulcerogenic effects, possibly by decreasing oxidative stress and increasing antioxidant enzyme activity. Further experimentation is needed to explore the exact mechanism of gastric mucosal and ulcer protection by A. alba leaves extract and to evaluate its chemical constituents effect as potential therapeutic and healing factors on gastric mucosal injury and ulcers induced by ethanol and other ulceration models.