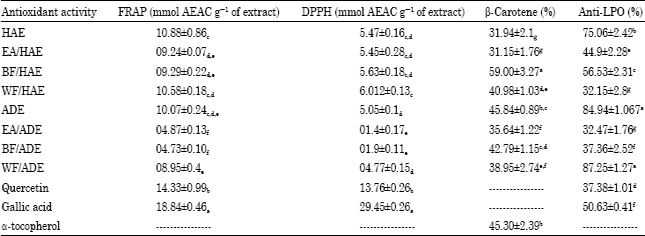
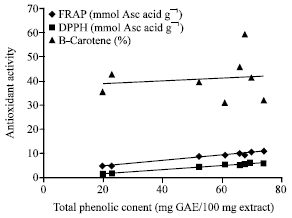
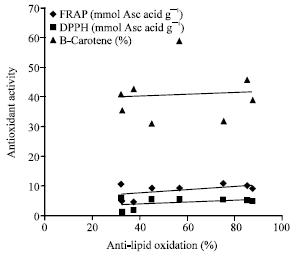
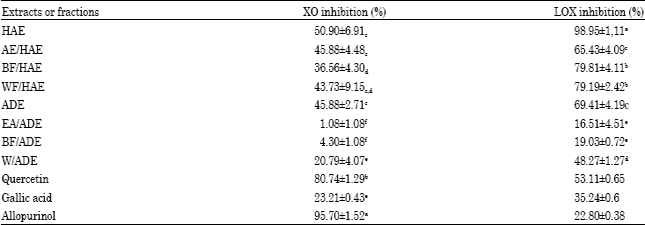
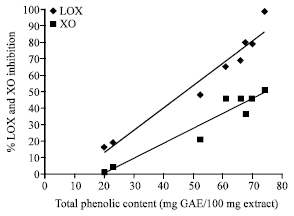
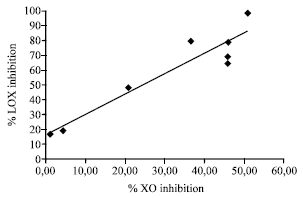
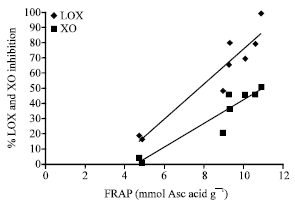
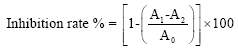Research Article
Antioxidant and Anti-inflammatory Activities from Galls of Guiera senegalensis J.F. Gmel (Combretaceae)
Laboratoire de Biochimie et de Chimie Appliquees (LABIOCA), UFR-SVT, Universite de Ouagadougou, 09BP 848 Ouaga 09, Burkina Faso
A. Hilou
Laboratoire de Biochimie et de Chimie Appliquees (LABIOCA), UFR-SVT, Universite de Ouagadougou, 09BP 848 Ouaga 09, Burkina Faso
C. Mounier
BioMed, Departement des Sciences Biologiques, Universite du Quebec a Montreal, 141 Avenue du President Kennedy, Montreal, Quebec H2X3Y7, Canada
A.Y. Coulibaly
Laboratoire de Biochimie et de Chimie Appliquees (LABIOCA), UFR-SVT, Universite de Ouagadougou, 09BP 848 Ouaga 09, Burkina Faso
M. Kiendrebeogo
Laboratoire de Biochimie et de Chimie Appliquees (LABIOCA), UFR-SVT, Universite de Ouagadougou, 09BP 848 Ouaga 09, Burkina Faso
J.F. Millogo
Laboratoire de Biologie et Ecologie vegetales, UFR/SVT, Universite de Ouagadougou, 09BP 848 Ouaga 09, Burkina Faso
O.G. Nacoulma
Laboratoire de Biochimie et de Chimie Appliquees (LABIOCA), UFR-SVT, Universite de Ouagadougou, 09BP 848 Ouaga 09, Burkina Faso