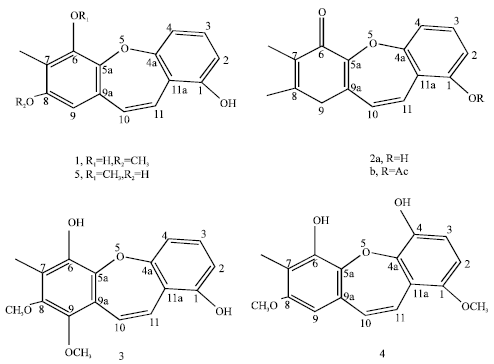
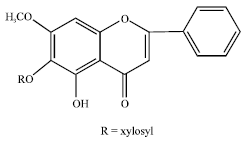
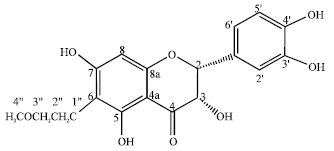
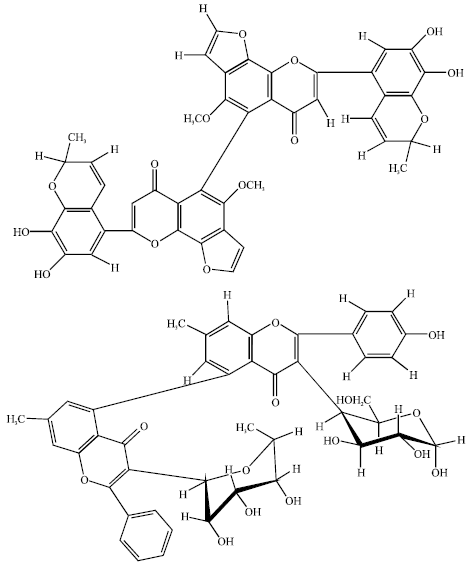
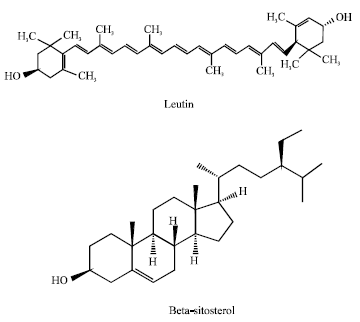
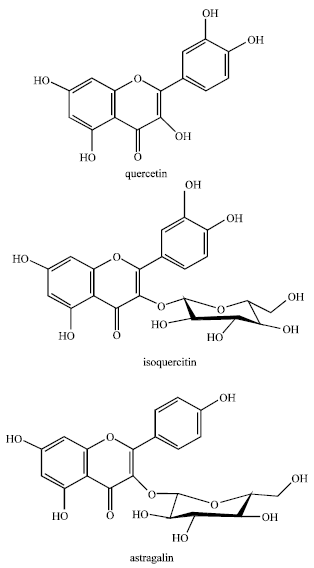
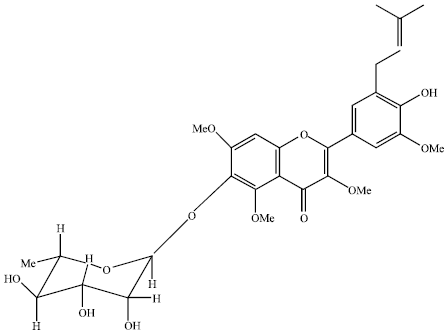
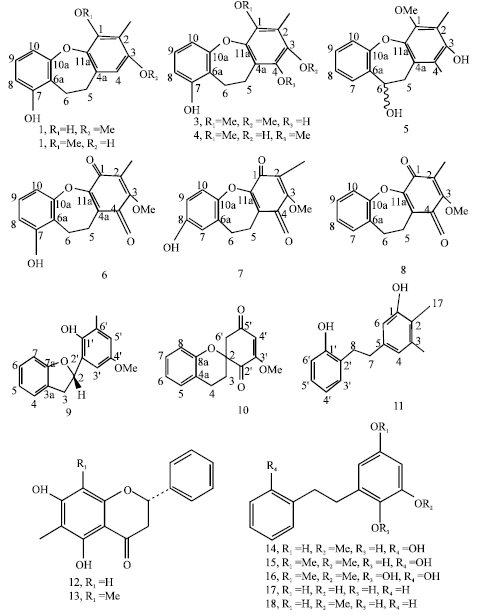
Review Article
Bauhinia purpurea Linn.: A Review of its Ethnobotany, Phytochemical and Pharmacological Profile
Department of Pharmaceutical Chemistry, RCPSR, Bhilai, 491 024, C.G., India
K.S. Chandrashekar
Department of Pharmacognosy, MIPS, Manipal, 457 6104, Karnataka, India