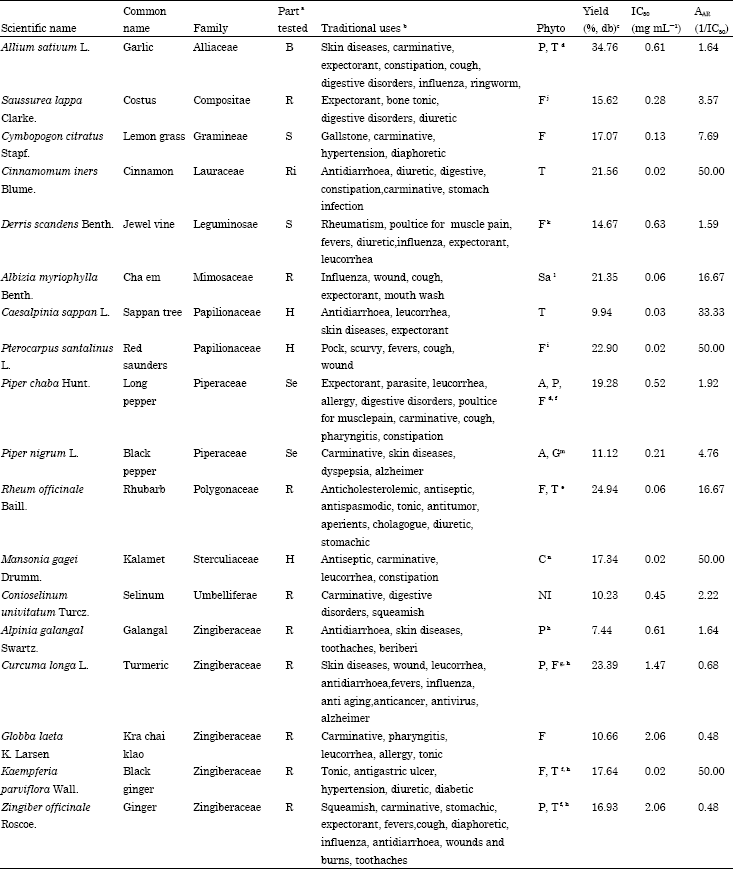
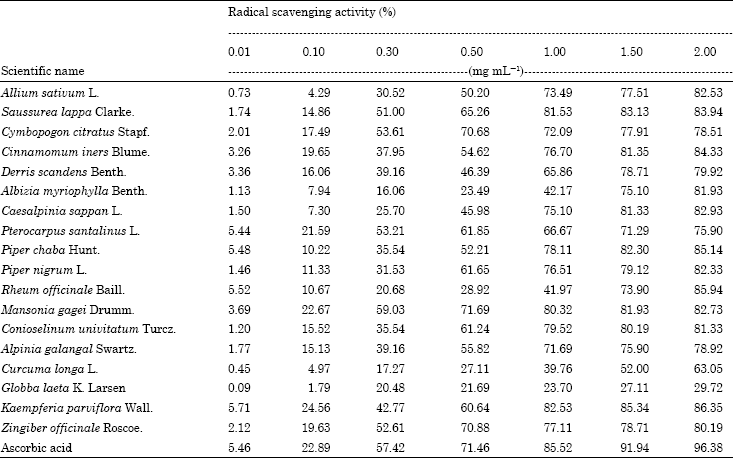
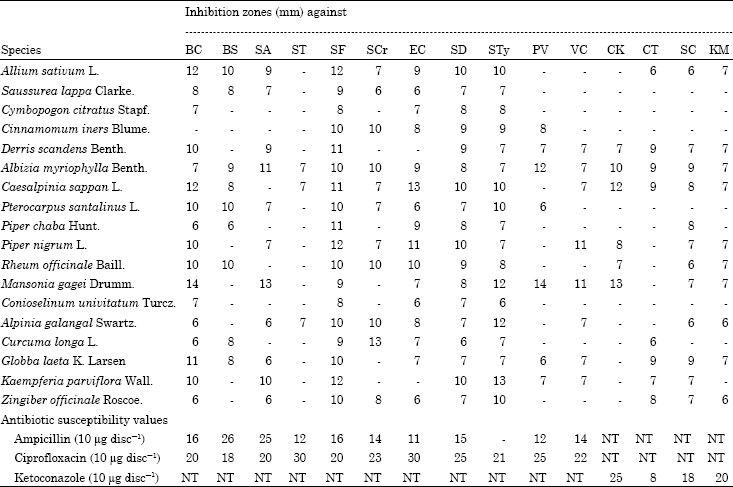
Research Article
In vitro Free Radical Scavenging and Antimicrobial Activity of Some Selected Thai Medicinal Plants
Laboratory Equipment Center, Division of Research Support and Academic Survice, Mahasarakham University, Maha Sarakham Province 44000, Thailand
S. Samappito
Department of Biotechnology, Faculty of Technology, Mahasarakham University, Mahasarakham 44000, Thailand