Research Article
Cytotoxicity and Biological Activity of Selected Sudanese Medicinal Plants
Institute of Food Science and Technology, University of Gezira, Wad Medani, Sudan
Cytotoxicity via the brine shrimp test was studied in order to reveal new anticancer compounds (Harborne, 1998). Toxicity to brine shrimps has a good correlation with anti-tumor activity in man (McLaughlin, 1991) since the brine shrimp responds similarly to the corresponding mammalian system (Solis et al., 1993).
Taxol, the new anti-cancer hospital drug from the bark of Taxus brevifolia, was discovered in this way. Alternatively, crude plant extracts can be first assayed for particular activities and the active fractions then analyzed phytochemically. Varieties of bioassays are now available for the phytochemist to use in such work (Hostettmann, 1991).
Twenty one sudanese plants, used in traditional medicine, were evaluated using the Brine Shrimp lethality Test (BST) The most active plants contain alkaloids in high concentrations in the most active plant Argemone mexicana (seeds, roots and leaves) (Dahawi, 2009).
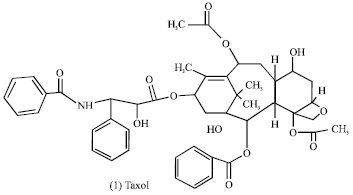
There are well-known drugs that are directly developed from plant species (Mans et al., 2000). The famous cytotoxic drugs are Vinblastine and Vincristine from Catharanthus roseus, the first cures in human cancer. Besides the cytotoxic plant compounds, the non cytotoxic drugs as Aspirin (Analgesic,anti-nflammatory) from Filipendula ulmaria, Benzoin (Oral disinfectant) from Styrax tonkinensis, Morphine (Analgesic) from Papaver somniferum and Quinine (For malaria prophylaxis) from Cinchona pubescens.
Development of natural products: The World Health Organization estimates that approximately 80% of the world's population relies primarily on traditional medicines as sources for their primary health care (Farnsworth et al., 1985). Over 100 chemical substances that are considered to be important drugs are either currently in use or have been widely used in one or more countries in the world have been derived from a little under 100 different plants. Approximately 75% of these substances were discovered as a direct result of chemical studies focused on the isolation of active substances from plants used in traditional medicine (Cragg and David, 2001).
Thirty-nine percent of the 520 new drugs approved during the period 1983 through 1994 were either natural products or derivatives of natural products (Harvey, 2001). Over 60% of antibacterial and antineoplastics were again either natural products themselves or based on structures of natural products (Grabley and Sattler, 2003).
As mentioned in previous studies, natural products today are most likely going to continue to exist and grow to become even more valuable as sources of new drug leads. This is because the degree of chemical diversity found in the natural products is broader than from any other source and the degree of novelty of molecular structure found in natural products is greater than that determined from any other source (Cragg et al., 1997,1999; Harvey, 2001; Sandsborg and Rolfsen, 1999).
Also, it was mentioned that, the revival of interest in the use of medicinal plant products for the treatment of various ailments is mainly due to increase awareness of the limited horizon of synthetic pharmaceutical products to control major diseases, high cost of currently available synthetic medicines, reported cases of adverse side-effects of modern medicines and perceived gentleness of natural medicines (Choudhary and Atta-Ur-Rahman, 2005).
Many of these plants are used as natural medicines by the natives to treat several tropical diseases including leishmaniasis, malaria, fungal and bacterial infections, benign and malignant tumors (Table 1).
Research into the use of plant- derived natural products alone in just the field of medicine covers a broad spectrum of activities (Dahanukar et al., 2000). Natural-products-based anticancer drug discovery continues to be an active area of research throughout the world (Da Rocha et al., 2001; Mann, 2002; Mehta and Pezzuto, 2002).
In addition, there will be need for the permanent search and development of new natural drugs. This need also arise from the advantage of certain natural products in controlling some diseases that chemicals fail to do. For example, the annonaceous acetogenins, extracted from pawpaw tree (Asimina triloba Dunal) were found to be the best effective against selected tumor type, e.g., squamtacin is selective against the human prostate carcinoma cell line (PC-3) and a series of 9-carbonyl compounds work best against the human pancreatic tumor cell line (PaCa-2) (Ahammadsahib et al., 1993).
The developing countries have also now opportunities to derive economic benefit by using their natural resources of medicinal plants by using traditional knowledge, which has often played a role in the development of modern science (Choudhary and Atta-Ur-Rahman, 2005).
In conclusion drug discovery can be significantly improved through the use of the knowledge to be gained from research into natural products (Gad, 2005).
Evaluation of natural products using bioassays: The plant kingdom represents an enormous reservoir of biologically active molecules and so far, only small fractions of plants with medicinal activity have been assayed. Nearly 50% of drugs used in medicine are of plant origin. There is therefore much current research devoted to the phytochemical investigation of higher plants that have ethanobotanical information associated with them. The phytochemicals (secondary metabolites) isolated are then screened for different types of biological activity (Harborne, 1998). In this regard, a simple bioassay was used for screening purposes (Hostettmann, 1991). Thus Artemia salina larvae (brine shrimps nauplii) has been used as a target organism to detect bioactive compounds in plant extracts and toxicity to this crustacean has a good correlation with anti-tumor (McLaughlin, 1991) and anti-trypanosoma (Zani et al., 1995) activities in man for the brine shrimp responds similarly to the corresponding mammalian system (Solis et al., 1993). Cytotoxicity via the brine shrimp test is studied in order to reveal new anticancer compounds (Harborne, 1998).
| Table 1: | Traditional uses of some sudanese flora |
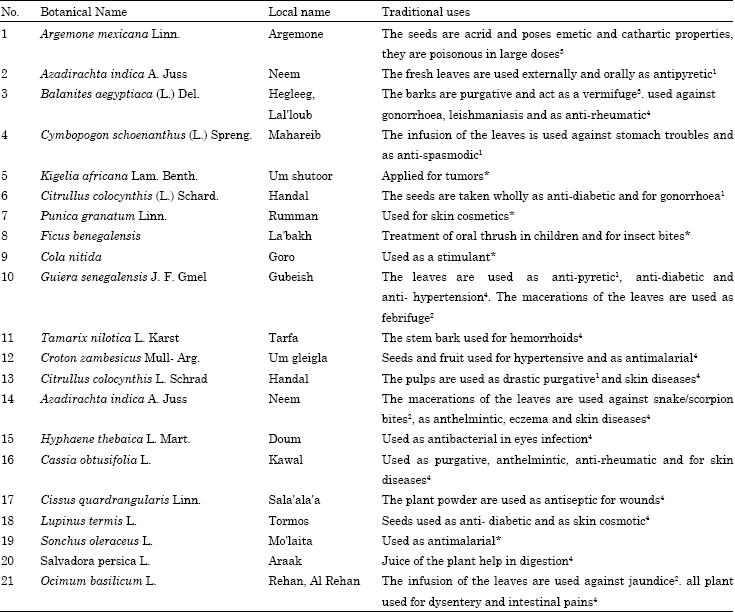 | |
| L: Leaves; R: Roots; S: Stem; Sb: Stem bark; Fl: Flower; Fr: Fruit; Sd: Seeds; Fr. Peri: Fruit pericarp; AP: Aerial parts; YL: Young leaves; M.L: Mature leaves; 1El Ghazali et al. (1997), 2El Ghazali et al. (1994); 3Broun and Massey (1929); 4El Ghazali (1997); *Traditional uses (not reported) | |
Another important bioassay to access the plant products cytotoxicity on human blood mononuclear cells for crude extracts was also used to evaluate botanicals.
 |
DEFINITION AND ORIGIN OF NATURAL PRODUCTS
Natural products are generally either of prebiotic origin or originate from microbes, plants, or animal sources (Nakanishi, 1999). Plants produce primary and secondary metabolites. Primary metabolites are those chemicals produced by plants that are directly involved in plant growth and development. They are essential for survival as glucose and chlorophyll. In contrast, secondary metabolites do not appear to be essential for survival. Most defensive chemicals in plants are considered secondary metabolites since a plant will grow perfectly well without them (Sumner, 2000).
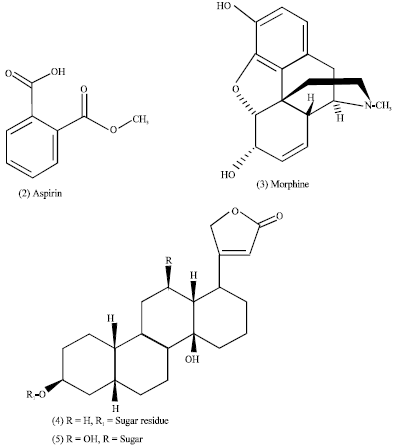
Plants are recognized for their ability to produce a wealth of secondary metabolites that are biosynthesized in plants (Fig. 1) (Cragg et al., 1999; Mann, 1987) for different purposes including growth regulation, inter and intra-specific interactions and defense against predators and infections (Verpoorte, 1998, 2000). The important secondary metabolites are alkaloids, terpenes, phenolic compounds and cyanogenic glycosides (Sumner, 2000).
Natural products as pharmaceuticals (non toxic drug): Plant-derived substances have traditionally played important roles in the treatment of human diseases. Today, about 80% of the world population residing in third world countries still rely almost entirely on plant products for their primary health care. The remaining 20% of individuals living in the first world use, in more than 25% of cases, pharmaceuticals which have been directly derived from plant products (Farnsworth, 1984; Cox, 1994). These range from common remedies such as aspirin (2) (originally isolated from the Rosacea; Filipendula ulmaria), to prescription drugs such as the analgesic morphine (3) and the cardiac glycoside digitoxin (4) and digoxin (5) (isolated from the Papaveracea; Papaver somniferum and the Apocynacea Digitalis purpula, respectively).
 |
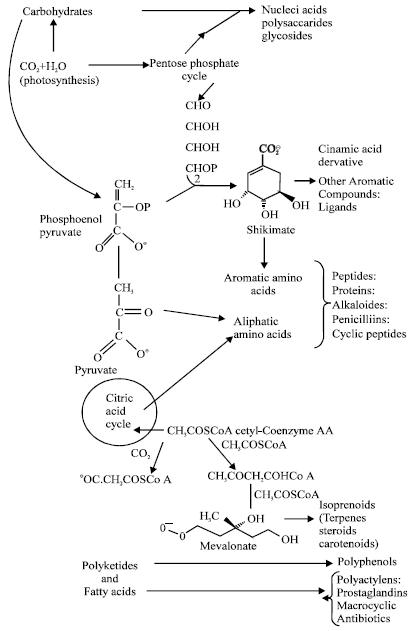 | |
| Fig. 1: | The process of the building blocks of the secondary metabolites |
Higher plants also produce a variety of different types of compounds, including biologically active proteins. Some of these types of compounds are even shared with other organisms and they include such chemical families as lectins, defensins, cyclotides and ribosome-inactivating proteins (O’Keefe, 2001). Ribosome - inactivating proteins are group of proteins exhibiting a wide spectrum of biological activities, including a ribonucleolytic activity for which the group is named. These compounds can be obtained from Panax ginseng and other plants have been reported to demonstrate antifungal and antiviral activities (Gad, 2005). (Table 2) shows a number of well-known drugs that are directly developed from plant species (Mans et al., 2000).
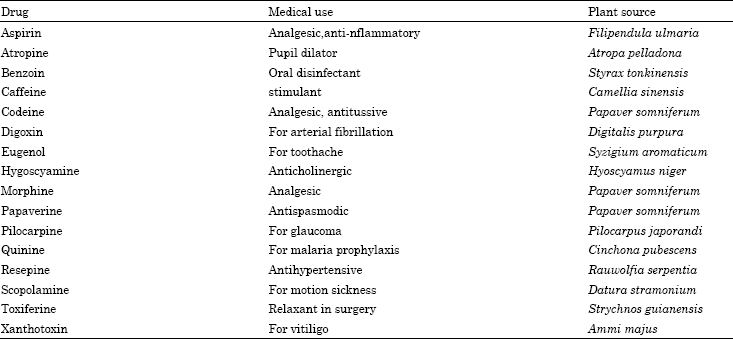
Natural products in cancer therapy (toxic drugs): Plant-derived compounds were also of great significance to cancer therapy (Table 3) (Mans et al., 2000). It was, for instance, only upon the addition of the Vinca alkaloid vincristine (6) and vinblastine (7) (isolated from Catharanthus roseus, Apocynaceae (Johnson et al., 1963) to mechlorethamine, prednisone and procarbazine that the first cures in human cancer (Hodgkin’s disease) were achieved (De Vita et al., 1970).
| Table 2: | Non cytotoxic drugs developed from plant sources |
 | |
| Table 3: | Some cytotoxic drugs developed from plant sources |
 | |
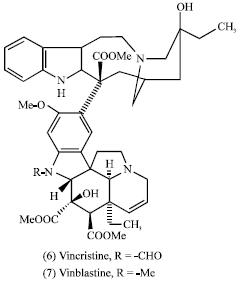
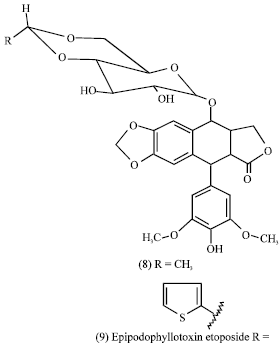
The combination of the epipodophyllotoxin Etoposide (8) and teniposide (9) is currently a highly active and curative regimen in testicular cancer (Williams et al., 1987). Etoposide is furthermore one of the most active agents against small cell lung carcinoma (Yarbro, 1992; Chabner, 1991; Harvey, 1999; Williams et al., 1987). The more recent development of the structurally and mechanistically novel taxanes, taxol (1) (extracted from the bark of Taxaceae Taxuus brevifolia, T. canadensis, or T. baccata (Wani et al., 1971) and the camptothecins (10) (derived from the bark and wood of the Nyssacea Camptotheca acuminata (Wall et al., 1966) in the 1990s represented a landmarkin cancer research because of their significant anti-solid tumor ovarian and breast carcinoma and has important activity against non-small cell lung cancer (McGuire et al., 1996).
 |
 |
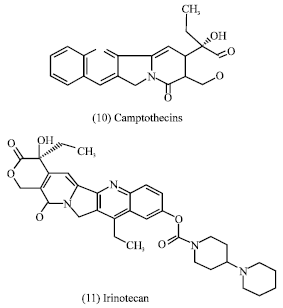
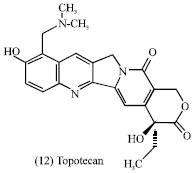
Irinotecan (11) and topotecan (12) are semi-synthetics from the lead compound camptothecin (10) which are approved for the treatment of advanced colorectal cancer (Bertino, 1997) and as second-line chemotherapy in ovarian carcinoma (Creemers et al., 1996), respectively. These agents are also active against several other solid malignancies such as carcinoma of the lung, cervix and ovary (Bertino, 1997; Creemers et al., 1996). In addition, some antitumor chemical classes from plants are monoterpenes, sesquiterpenes, diterpenes, triterpenes, steroids, cucurbitacins, saponins, cardenolides, lignan, quinones, pyrrolizidine and isoquinoline alkaloids from different plant families (Evans, 1989).
 |
 |
Natural products in food stuffs: The spices are common food adjuncts that impart color, flavor and aroma.
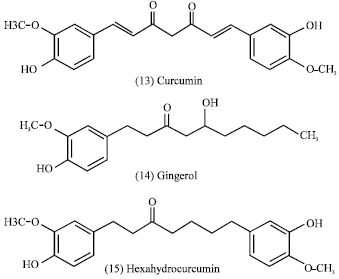
It shows potential health benefits, as they possess antioxidant activity e.g., in Indian cooking turmeric and ginger are used in major quantities. The active ingredient in turmeric is curcumin (13) and that in ginger are gingerol (14) and hexahydrocurcumin (15) Both these compounds prevent oxidation of oils and fats (Vankar et al., 2006).
 |
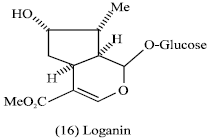
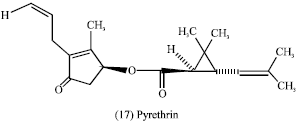
Natural products in pesticides: Groups of regular and irregular monoterpenes are used as natural insecticides such as iridoids and in particular loganin (16) and pyrethrin (17) (Evans, 1989).
 |
 |
Also rotenone (18) the isoflavone derivative, nicotine (19) the characteristic alkaloid of the genus Nicotiana have long been used as an effective insecticides (Evans, 1989).
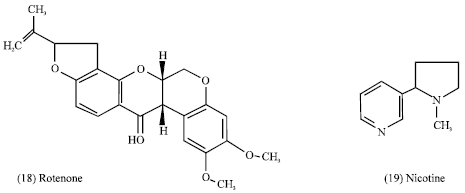 |
Additionally, secondary metabolites from higher plants have recently been used as pesticides or model for new synthetic pesticides, e.g., toxaphene (20) (insecticide and herbicide) and cinmethylin (21) (hebecide). These chemicals were developed from plant derived natural products such as terpenoids that can be found in the essential oil secreted by the glandular trichomes of Artemisia (Compositae) or closely related genera (Prates et al., 1998).
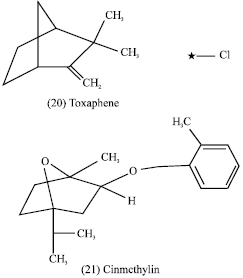 |
The secondary metabolites of trichomes of several plant species play central role in plant protection against plant feeding insects (Prates et al., 1998). So large amounts of natural pesticides were produced by natural pesticides factories (trichomes and root hairs) of the epidermis of aerial and rhizosphere plant parts (Duke et al., 2000). Monoterpenes occurring in trichomes have an insecticidal effect, particularly on termites (Harborne, 1997; Baker and Walmsley, 1982).
THE USE AND ADVANTAGE OF BIOLOGICAL ASSAYS
As mentioned by McLaughlin et al. (1998), today's work in natural product chemistry must incorporate bioassays. Extracts must be screened for biological activity, the active extracts selected, fractionations directed with bioassays and the bioactive compounds identified and then exploited. Therefore, for evaluation, botanicals in modern chemistry four bench top bioassays have been adopted which do not require higher animals to screen and direct the fractionation of botanical extracts in drug discovery efforts. These are (1) The brine shrimp lethality test (BST), (a general bioassay); (2) The inhibition of crown gall tumor on discs of potato tubers (an anti-tumor bioassay); (3) The inhibition of frond proliferation in duckweed (a bioassay for herbicides and plant growth stimulants) and (4) The yellow fever mosquito larvae lethality test (a bioassay for pesticides). The BST is especially suggested as an inexpensive, simple and rapid means of standardization of bioactivity in heterogeneous botanical products (McLaughlin et al., 1998).
Brine Shrimp Lethality Test (BST): Toxicity to Artemia salina larvae has a good correlation with anti-tumor, pesticidal (McLaughlin, 1991) and anti-trypanosoma (Zani et al., 1995) activity. The brine shrimp responds similarly to the corresponding mammalian systems. For example, the DNA – dependant RNA polymerases of A. salina have been shown to be similar to the mammalian type (Birndorf et al., 1975).
Bioassays offer a special advantage in the standardization and quality control of heterogeneous botanical products that contain mixtures of bioactive components from the same or from mixed botanical sources. Standardization of the products by biological assays will then generate reproducible benefits and the resulting consumer confidence (McLaughlin et al., 1998).
A positive correlation is found between Brine Shrimp toxicity and 9KB (human nasopharyngeal carcinoma) cytotoxicity (McLaughlin et al., 1993).
Uses of brine shrimp lethality bioassay: Over 300 novel antitumor and pesticidal natural products have now been isolated in the Laboratory at Purdue University using this bioassay as the prescreen (McLaughlin, 1991; McLaughlin et al., 1993). Thus, it is possible to detect and then monitor the fractionation of cytotoxic, as well as 3PS (p-388) (in vivo murine leukemia) active extracts using this bioassay rather than more tedious and expensive in vitro and in vivo antitumor assays. It is as convenient in house prescreen to the more expensive mechanism-based, cytotoxicity, or in vivo antitumor assays (McLaughlin et al., 1998). In addition, the brine shrimp test is an effective screening method for pesticides as in the discovery of the Annonaceous acetogenins as a new class of natural pesticides (Alkofahi et al., 1989; McLaughlin et al., 1997). These compounds are also powerful as in vivo active antitumor agents (Ahammadsahib et al., 1993). Brine shrimp have been previously utilized in various bioassay systems. Among these applications have been the analysis of pesticide residues, mycotoxins, stream pollutants, anesthetics, dinoflagellate toxins, morphine–like compounds, toxicity of oil dispersants, co- carcinogenicity of phorbol esters and toxicants in marine environments (Meyer et al., 1982).
BIOACTIVE COMPOUNDS
Secondary metabolites are produced by plants allegedly because they play certain biological and/or ecological roles towards combating other plants, animals, insects and man (Mann, 1987).
Many of these natural products have been shown to present interesting biological and pharmacological activities and are used as chemotherapeutic agents for centuries to treat a variety of diseases or serve as the starting point in the development of modern medicines (Verpoorte, 1998, 2000).
Alkaloids: The activities of these metabolites arise from their biological role in their origin. Some alkaloids act as growth regulators and others as insect’s repellants or attractants. In plants, Nicotine (19) protects tobacco plant from browsing insects; so it is used as an insecticide (Mann, 1987). Whereas for animals the pyrrolizidine alkaloids exhibit dramatic hepatic toxicity to livestock and cause serious diseases. It is believed that the toxicity is due to pyrrole esters of the type (22) which have potent alkylating capabilities. These esters are produced by pyrrolizidine alkaloids in the mammalian liver (Clarke and Clarke, 1975). Some amino acid metabolites such as cyanogenic glycosides act as feeding deterrents and potent allelopathic agents.
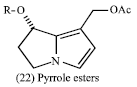 |
For insects, it is believed that alkaloids have an ecological role in two Species of butterflies since their larvae become distasteful to predators because of alkaloidal accumulations in their tissues (Mann, 1987).
In man, the alkaloid serotonin (23) is a natural nerve transmitter in the central nervous system, responsible in part for control of sleep patterns.
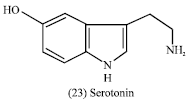 |
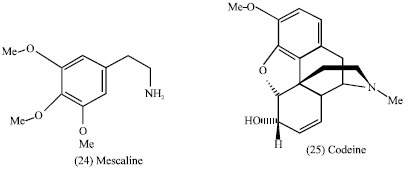
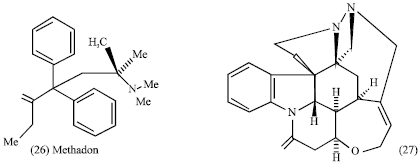
To man, alkaloids represent one of the largest and most interesting groups of plants metabolites. One reason for the growing attention concerning the natural compounds, is their pharmaceutical and therapeutical value (Garica et al., 2006). The pharmacologic action of alkaloids varies widely: many alkaloids such as mescaline (24), codeine (25), methadone (26) and morphine (2) have hallucinogenic properties and account for many analgesics today (Tyler et al., 1988). Whereas others strychnine brucine (dimethoxy strychinine) (27) are central stimulants.
 |
 |
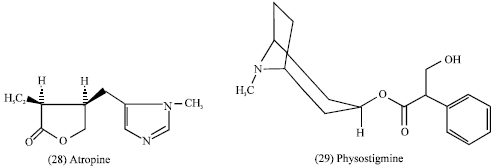
Some atropine (28) are mydriatics whereas others physostigmine (29), pilocarpene (30) are miotics.
 |
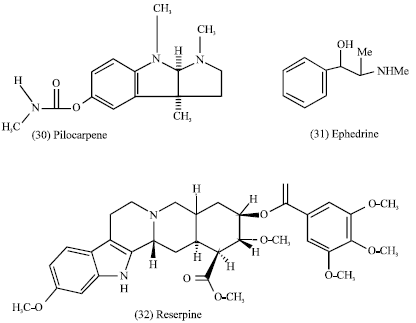
Some ephedrine (31) cause a rise in blood pressure, but others reserpine (32) produce a fall in excessive hypertension. In fact, the akaloids are capable of extensive physiologic activity (Tyler et al., 1988).
 |
The Solanaceous alkaloid atropine is a CNS stimulant used as an antidote in cases of poisoning caused by cholinesterase inhibitors such as physostigmine and organophosphate insecticides. Scopolamine has a depressant activity on the central nervous system and it is used to treat motion sickness. Cocaine (33) is used as a local anesthetic and the hydrochloride of the alkaloid cocaine is widely used to control severe pain associated with terminal cancer for it’s CNS stimulant properties (Tyler et al., 1988).
Ephedrine (31) is used in asthma therapy and to elevate blood pressure. The leaves of Catha edulis (Khat) family Celastraceae contain the alkaloid norpseudoephedrine (34) and are chewed in North Africa and Arabia for their narcotic effects (Clarke and Clarke, 1975).
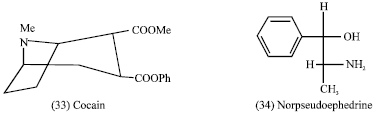 |
Emetine hydrochloride, the Isoquinoline alkaloid, is an antiamaebic and acts primarily in the intestinal wall and the liver. It inhibits polypeptide chain elongation, thereby blocking protein synthesis (Tyler et al., 1988).
Codeine and its salts, the most widely used opium alkaloid, are narcotic analgesics and antitussives; they are used as sedatives especially in allying coughs whereas papaverine hydrochloride is a smooth muscle relaxant (Tyler et al., 1988).
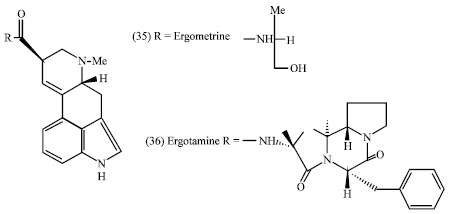
Some vinca alkaloids such as vicristine (5) (R = CHO) and vinblastine (6) (R = Me) are anti-tumor agents and are used in cancer chemotherapy. Some ergot alkaloids have pharmacological properties such as the amides of lysergic acid; ergometrine (35) and ergotamine (36) which are used as uterine contracting agents (Mann, 1987).
 |
Terpenes: Terpenes are secondary metabolites essential for the maintenance of life and many of them have physiological effects (Tedder et al., 1972). Groups of regular and irregular monoterpenes are used as natural insecticides (Evans, 1989).
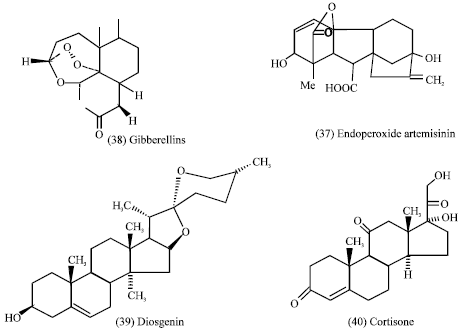
Over 500 compounds of the sesquiterpenes are known; they are particularly characteristic of the Compositae but do occur sporadically in other families. Many possess antitumor, anti-leukaemic, cytotoxic and antimicrobial activities. They can be responsible for skin allergies in humans and they act as insect feeding deterrents (Evans, 1989). Sesquiterpene lactones possess potent antitumor properties. The marked biological activity of these lactones of α –β- unsaturated lactones derived from them is thought to be due to their properties as Michael acceptors, that is, as potent alkylating agents. Most of the alkylating agents are cytotoxic (Hegnauer, 1977). They display a wide variety of biological effects such as antiviral, anti-inflammatory and catatonic activity (Quintero et al., 1999). In previous experiments, some derivatives of sesquiterpene lactones were prepared to be tested as antiviral and/ or catatonic agents. These experiments suggested that some derivatives should inhibit DNA replication in cancer cell lines (Quintero et al., 1999). Other several series of analogues of lactones and sesquiterpenoid lactones were synthesized and tested. They are α, β-unsaturated lactones, aroylacrylic acids and esters; the common molecular basis is the presence of α, β-unsaturated carbonyl moiety. The biological functioning of such compounds has been related to the reactivity of the activated double bond towards nucleophilic groups of essential enzymes. Studies of Q.S.A.R (Quantitative Structure-Activity Relationship) were performed, correlating biological activity with their lipophilic character, enzymatic inhibition and reaction rate with model nuleophiles. The in vitro cytotoxic activity of the sesquiterpene lactone the endoperoxide artemisinin (37) and some chemically prepared derivatives, which have been found to display cytotoxicity to murine and human tumor cells. In artemisinin, the endoperoxide group appeared to play a role in cytotoxicity. It was clearly more cytotoxic than deoxy artemisinin, which lacks the Endoperoxide Bridge (Beekman et al., 1998).
Diterpenes (gibberellins) seem to behave as plant hormones, responsible for increased growth and induction of flowering. Gibberellic acid (GA3) is the most familiar gibberellin (38) (Harborne, 1998). Diosgenin (39) has a variety of clinical uses for the production of the anti-inflammatory agent, cortisone (40) used to suppress rheumatoid arthritis (Mann, 1987). Carotenoids produce a number of important metabolites such as vitamin A by degradation.
 |
Cardiac glycosides: Cardiac glycosides are a class of natural products that are used to increase cardiac contractile force in patients with congestive heart failure and cardiac arrhythmias (Hauptman et al., 1999). The most familiar are digoxin (4), digitoxin (3), which are derived from the plant genera Digitalis and Strophanthus gratus, respectively. Their mechanisms of action in the heart are well known and involve inhibition of the plasma membrane Na+/K+-ATPase (Rose and Valdes, 1994) leading to increased intracellular Na+ and Ca+ and decreased intracellular K+ (Kasner and Ganz, 1992). The increased intracellular Ca2+ promotes muscle contraction and cardiac contractile force (Rose and Valdes, 1994). Digitoxin (3) and digoxin (4) also induce vomiting and affect the central nervous system and their combined effects are unpleasant to grazing animals.
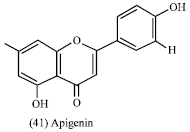
Phenolic compounds: The wider distribution and more abundant presence of flavonoids in the plant kingdom, together with reported research results using individual flavonoids, suggest that flavonoids may contribute to the preventive effect of a plant-based diet against chronic disease. Consumption of a diet rich in fruits and vegetables has been associated with reduced rates of many forms of cancer, including colon cancer (Wang et al., 2000). Certain compounds have been identified in fruits and vegetables that may account for this cancer prevention, e.g. apigenin (41) was found to be cancer chemo preventive agent (Wang et al., 2000). It has been shown to be growth inhibitory in many cancer cell lines such as human breast cancer cells (Hirano et al., 1989), leukemia cells (Lee et al., 1995) and solid malignant tumor cells (Fotsis et al., 1997). It was reported that apigenin is a strong inhibitor of ornthine decarboxylase activity, a marker of tumor promotion, induced by 12-O-tetradecanoylpharbol-13-acetate (TPA) in the murine epidermis (Wei et al.,1990).
 |
Furthermore, apigenin has been shown to increase gap junctions in rat liver epithelial cells, a property that may contribute to its chemo preventive ability by maintaining cell-cell communication (Chaumontet et al., 1994). Most recently, it was reported that apigenin induces a reversible G2/M arrest in both epidermal and fibroblast cells by inhibition of P34cdc2 kinase activity. The ability of apigenin to induce cell-cycle arrest may be a critical component of the biologic mechanism by which apigenin inhibits skin carcinogenesis (Lepley and Pelling, 1997).
Some phenolic compounds from Duchesnea chrysantha as Gallic acid (42), methyl caffeate (43), protocatechuic (44) acid inhibited some human cancer cell (Lee and Yang, 1994). Several phenolic acids-caffeic (45) and Gallic acid (42) derivatives - were screened for their potential antiprolifrative and cytotoxic properties in different human cancer cell lines.
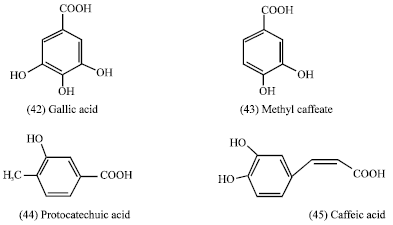 |
Distinct effects were found for different cell lines, which points that the trihydroxylated derivatives yielded better results than the dihydroxylated ones (Gomes et al., 2003). These phenolic compounds show a wide and contradictory behavior, involving antitumor and pro-and antimutagenic activity (Skaper et al., 1997). Their antitumor activity is highly dependant upon their conformational characteristics, which, in turn, determine their antioxidant/pro-oxidant properties (Gomes et al., 2003).
The presence of the double bond in the side chains was found to lead to an increase of those activities (Gomes et al., 2003). Mechanism of polyphenol cytotoxicity may be related to their pro-oxidant properties, since the same polyphenols could behave as both antioxidant and pro-oxidant, depending on the concentration and free radical source (Sergediene et al., 1999). Tannins, the polyphenols, show astringent action when applied to living tissues. This action form the basis for therapeutic application of tannins. Tannin-bearing drugs and tannic acid and their derivatives are employed in medicine as astringent for it forms precipitate with the protein as the protective coat that forms in the treatment of burns (Tyler et al., 1988).
SOME SELECTED SUDANESE MEDICINAL PLANTS
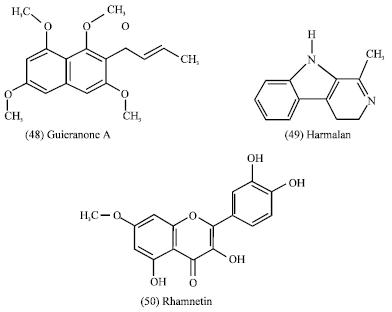
Guiera senegalensis: Guiera senegalensis J.F. Gmel (Combretaceae) is a shrub of savannah region of West and central Africa. Its leaves (Fig. 2) are commonly used in traditional medicine in gastrointestinal disorders, respiratory infections, rheumatism and as antimalarial agent (Fiot et al., 2006). Previous studies indicated the presence in leaves of two alkaloids (harman, tetrahydroharman or eleagnine), flavonoids, naphthopyrans (5-methyldihydroflavasperone (46) and 5-methylflavasperone (47), tannins and a naphthyl butenone (guieranone A) (48).
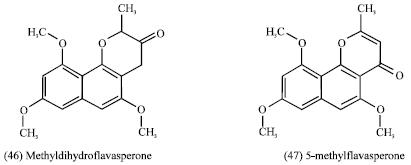 |
In roots, only tannins were obtained and the beta-carboline alkaloids harmalan (49) (Mahmoud and Khalid, 1997; Ancolio et al., 2002; Silva and Gomes, 2003). Rhamnetin (quercetin 7-methyl ether) (50) was isolated from the leaves of the plant which showed a strong inhibitory activity on 5-lipoxygenase from porcine leucocytes (Bucar et al., 1998). Further pharmacological activities which showed a strong inhibitory activity on 5-lipoxygenase from porcine leucocytes (Bucar et al., 1998).
 | |
| Fig. 2: | Guiera senegalensis (Leaves) Family: Combretaceae Local name: Gubaich |
Further pharmacological activities (antitussive or antimicrobial properties) of leaves of Guiera senegalensis have already been shown (Kudi et al., 1999). Total alkaloids from leaves and roots of Guiera senegalensis, in vitro study of antiplasmodial activity and cytotoxicity presented approximately the same antiplasmodial activity and a low cytotoxicity toward human monocytes (THP1 cells) (Fiot et al., 2006).
 |
Similar effects of roots and leaves explain that leaves are the most used in traditional medicine to treat malaria, because of the difficulty to obtain roots. Harman and tetrahydroharman could be responsible of antiplasmodial activity of the whole plant (Fiot et al., 2006) while guieranone A presented a high toxicity toward human monocytes (THP1 cells), two cancer cell lines (human cervix and human colon carcinoma) and against normal skin fibroblasts too (Fiot et al., 2006). The leaves ethanolic extract of G. senegalensis, total cardiac glycosides and the total sesquiterpene lactones isolated from the plant showed cytotoxicity to the brine shrimps (Dahawi, 2009).
Croton zambezicus: Croton zambezicus Muell. Arg. (Euphorbiaceae) is a shrub or small tree reaching 10 m in height. It's a Guineo-Congolese species widespread in Tropical Africa (Adjanohoun et al., 1989). Some members of Euphorbiaceae family contain anthraquinones, triterpenoids, fatty acid epoxides, unsaturated fatty acids and antitumor agents. Alkaloids, when present, are usually of the aporphine, pyridine, indole, quinoline or tropane types (Evans, 1989).
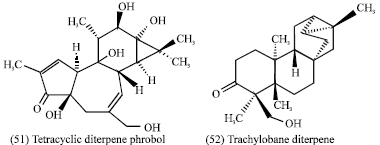
The Croton zambezicus leaf decoction is used in Benin as anti-hypertensive, anti-microbial (urinary infections) and to treat fever associated with malaria (Adjanohoun et al., 1989). The genus Croton is well known for its diterpenoid content and a lot of different types of diterpenes (phorbol esters, clerodane, labdane, kaurane, trachylobane, pimarane, etc.) have been isolated from this genus. There is very little literature concerning the phytochemical study of Croton zambezicus although if this plant is widely used in African traditional medicine (Block et al., 2004). There is no literature concerning Croton zambezicus seeds (Fig. 3) which has pharmacological uses in folk medicine in Sudan. Croton tiglium seeds contain about 50% of fixed oil which contains croton resin; also 'crotin', a mixture of croton-globulin and croton-albumin comparable with ricin. The oil also contains diesters of the tetracyclic diterpene phorbol (51) (Evans, 1989). Labdane, clerodane and trachylobane diterpenes have been identified in the stem bark of C. zambezicus (Ngadjui et al., 2002). A new cytotoxic trachylobane diterpene (52) from the leaves of C. zambezicus has been identified (Block et al., 2002).
 | |
| Fig. 3: | Croton zambezicus (seeds) Family: Euphorbiaceae Local name: Umgleigla |
The flavonoid of the plant seeds showed high toxicity to the brine shrimps (Dahawi, 2009).
 |
Hyphaene thebaica: Hyphaene thebaica family (Palmae) contains saponins, tannins, catechins, flavonoids, terpenoids and ketones in addition to fixed oil, carbohydrates and leaf wax (Evans, 1989).
A previous pharmacological and phytochemical study was carried out in the different organs of Hyphaene thebaica reported the presence of high concentration of tannins and various concentrations of flavonoids, terpenoids, steroids and alkaloids. The Doum seed extract was shown to have some biological activities in vitro (El-Siddig, 1996). There is no literature concerning the doum hair (Fig. 4) that has been one of the plants of the study.
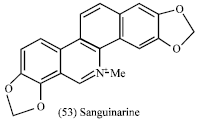
Argemone mexicana (seeds, roots and leaves): Argemone nexicana belong to Papaveraceae family which is characterize by the presence of isoquinoline and benzophenanthridine-type alkaloids. Sanguinarine (53) is a quaternary benzo[C] phenanthridine alkaloid widely distributed in the plants of the Papaveraceae, Fumariaceae and Rutaceae families (Garcia et al., 2006).
 |
 | |
| Fig. 4: | Hyphaene thebaica (hair) Family: Palmae local name: Doum |
These biologically active alkaloids were reported to have a wide range of potentially useful medicinal properties, such as antimicrobial (Facchini, 2001; Simanek, 1985), anti-inflammatory (Lenfeld et al., 1981) and antitumoral activities (Adhami et al., 2004). Sanguinarine has a strong bactericidal effect on gram-positive bacteria. These medicinal properties are due to the interaction of sanguinarine with DNA (Bajaj et al., 1990; Maiti et al., 1982). On the other hand, adverse effects (genotoxicity and hepatotoxicity) have been reported. In previous study some isolated benzophenanthridine-type alkaloids from the chloroform extract from the aerial parts of Argemone mexicana were evaluated for their cytotoxicity to human nasopharyngeal carcinoma (HONE-1) and human gastric cancer (NUGC) cell lines. Chelerythrine that is present in the root extract was found to exhibit significant activity against NUGC, while angoline inhibited both types (Duke, 1992b). (+)-Argenaxine showed moderate activity against the NUGC cell line (Chang et al., 2003). Also in the brine shrimp test, high toxicity of ethanolic extracts of Argemone mexicana seeds, roots and leaves are reported (Dahawi, 2009).
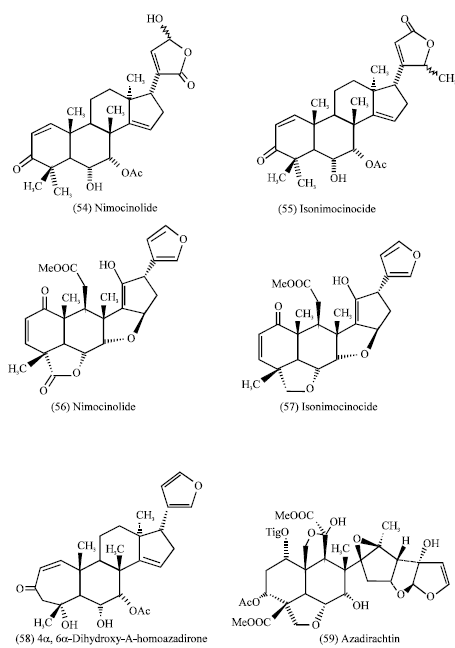
Azadirachta indica (young and mature leaves): Siddiqui et al. (1986) reported that, Nimocinolide (54) and Isonimocinocide (55) were isolated from green winter leaves of Azadracta indica (Meliaceae). They were reported to possess larvicidal properties when applied to mosquito larvae Aedes aegypti (Naqvi, 1987) so the higher activity of the young leaves may be due to the presence of these compounds. it was observed that the ethanolic extract of the young leaves of Azadracta indica showed high cytotoxicity compared to the mature one in the brine shrimp test (Dahawi, 2009).
Other minor components belonging to the nimbin group isolated from the plant leaves, Nimbolide (56) and 28-Deoxonimbolide (57) are cytotoxic towards human tumor cell lines (Kigodi et al., 1989).
Also 4α, 6α-Dihydroxy-A-homoazadirone (58) was isolated from neem leaf extracts (Bruhn et al., 1984) and was found to be an antifeedant of moderate activity. It is obvious that Azadirachtin (59) is still the most potent ingredient of neem, which exhibits only very weak antifeedant activity and no toxicity (Serit et al., 1992).
 |
it was observed that the ethanolic extract of the young leaves of Azadracta indica (Meliaceae) showed high cytotoxicity (LC50 = 28 μg mL-1) compared to the mature one.
Balanites aegyptiaca (stem bark): The plant ethanolic extract exhibited high toxicity to the brine shrimps (Dahawi, 2009).
Cymbopogon schoenanthus (aerial parts): Cymbopogon schoenanthus (L.) Spreng is an aromatic plant very often used in traditional pharmacopoeia for intestinal as well as external treatments (Yentema et al., 2007). In the review, the most important plant essential oil is the monoterpene (53.2%) and sesquiterpene (12%). The monoterpenes Piperitone (60) and δ-2-Carene are the principal components in the plant essential oil (Yentema et al., 2007). Essential oils have a significant role in fields such as medicine, pharmacy, cosmetics, chemical and food-processing industries (Yentema et al., 2007).
 |
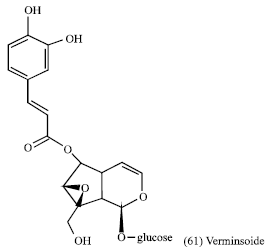
Kigelia africana (fruit and seeds): Kigelia Africana (Kigelia pinnata), belonging to the family Bignoniacea is widespread across Africa and is found in wet savannah and riverine areas. The plant is used in Africa for anti-inflammatory, anti-microbial and anti-skin-aging effects. Chemical analysis of a polar extract of fruit from K. africana in previous researches indicated the presence of Verminoside (61), an iridoid, as a major constituents and a series of polyphenols such as verbascoside. In vitro assays showed that verminoside had significant anti-inflammatory effects (Picerno et al., 2005).
 |
Citrullus colocynthis (fruit, seeds and leaves): Colocynth is the dried pulp of the fruit Citrullus colocynthis (Fam. Cucurbitaceae). The activity of colocynth is contained in an ether-chloroform soluble resin. Among the several components of the pulp is Cucurbitacin E or α-Elatrin (62) in the form of glycoside. Cucurbitacins have been shown to have tumor-necrosing activity. Choline and two alkaloids have also been reported (Evans, 1989).
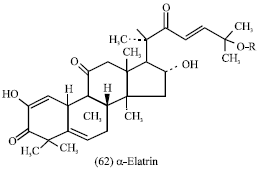 |
Punica granatum (fruit pericarp): The dried pericarp of the fruit of Punica granatum L. (Punicaceae) is known as Pomegranate rind. The ethanolic extract of the fruit pericarp was found to be toxic to the brine shrimps in BST (Dahawi, 2009). Pomegranate rind is very astringent and contains about 28% of tannin and coloring matter (Evans, 1989). Also Duke (1992a) reported the presence of active compounds as Gallic acid, mannitol, pectin, punicalagin punicalin and tannins. All these reported compounds and groups observed experimentally were known as active and toxic and most of the compounds were reported as anticancer and used as pesticides (Duke, 1992b).
Sonchus oleraceus (leaves, roots, flowers and seeds): In previous studies it was reported that this plant contains the biologically active Luteolin- 7- glucoside (Duke, 1992a) that showed antiviral and pesticidal activity (Duke, 1992b) and the Cytotoxic, antitumor, fungicide and herbicide scopoletin (Duke, 1992a, b). In addition, the studies reported the presence of sesquiterpene glycosides (El-Zalabani et al., 1999; Mercadante and Rodriguez-Amaya, 1990).
Cola nitida (seeds): Cola or kola nuts are the dried cotyledon of Cola nitida (Cola vera) (Sterculiaceae). In the tropical countries where it grows, the fresh nut is chewed as a stimulant. C. nitida is a large tree indigenous to West Africa. Kola seeds possess the central stimulating action of Caffeine (1-2.5%) and a little Theobromine. It also contains about 5-10% of tannoids, particularly Catechol and Epicatechol. In the dried nuts Caffeine and Theobromine, are found free (Tyler et al., 1988; Evans, 1989).
Catharanthus roseus (leaves): Catharanthus roseus (Apocynaceae) is an example of a drug plant, which has been introduced into medicine in cancer therapy. After production of leukopenic actions of the leaves extracts during 1955-1960, about 90 alkaloids have been isolated from the plant. Of particular interest is a group of about 20 dimeric alkaloids which contain those having antineoplastic activity, including Leurocristine (Vincristine) (5) and Vincaleukoblastine (Vinblastine) (6).
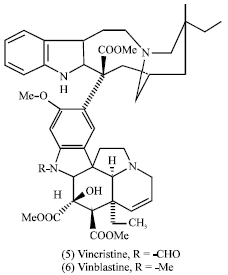 |
Vinblastine is therefore composed of the indole alkaloid Catharanthine (63) and the dihydroindole alkaloid Vindoline (64), both of which occur free in the plant (Evans, 1989).
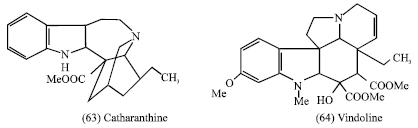 |
Lantana camara (leaves): Lantana camara L. (Verbenaceae) it is a large evergreen strong- smelling herb (Anonymous, 1989). Its leaves are used as, antitumoral, antibacterial and antihypertensive agent (Taoubi et al., 1997). Several tri- terpenoids, naphaquinones, flavonoids, alkaloids and glycosides isolated from this plant are known to exert diverse biological activities including cytotoxic and anticancer properties (Ghisallberti, 2000). In previous study, it was reported that the leaves methanolic extract showed moderate activity against Dalton’s lymphoma Ascites (DLA) cells (Raghu et al., 2007). the presence of toxic lantanoids e.g. Lantadene A (65) and Lantadene B (66) was reported.
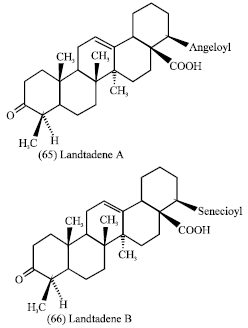 |