ABSTRACT
Schistosomiasis (known as Bilharziasis) is the disease caused by a blood born fluke (trematode) of the genus Schistosoma. The intermediate hosts of all digenetic trematodes are snails and schistosomes are no exception. Adult schistosome worms live in a mammalian host, Schistosomiasis is the second most prevalent tropical disease in Africa after malaria and is of great public health and socio-economic importance in the developing world. This study is to clarify that Natural product extracts with non-toxic medicinal properties should be explored for possible intervention in schistosomiasis as a disease involving impairment of metabolism of infected subjects. These inspire more hope for reducing the intensity of schistosomal infection by reduction in worm burden, ova count, granuloma size and number leading to improvement in histopathological picture of liver, spleen and kidney as a result of reducing inflammatory and fibrotic reactions of schistosoma.
PDF Abstract XML References Citation
How to cite this article
URL: https://scialert.net/abstract/?doi=rjmp.2011.1.20
INTRODUCTION
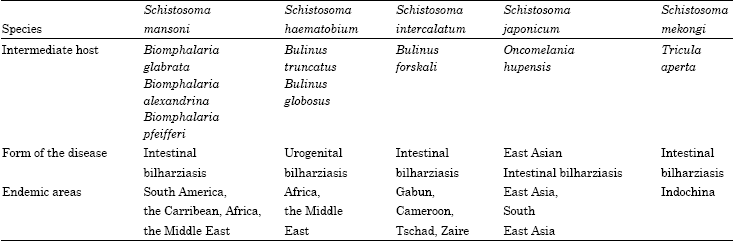
Schistosomiasis is the second most prevalent tropical disease in Africa after malaria and is of great public health and socio-economic importance in the developing world (Engles et al., 2002). Schistosoma mansoni-causative agent of intestinal bilharzia-originated in Africa but was carried to South America, with the slave trade; it is transmitted by snails of the genus Biomphalaria snails (link). Schistosoma haematobium which causes urinary bilharzia is transmitted by snails of the species Bulinus, which inhabit less permanent water bodies (Ross et al., 2002). The third major species of schistosome is S. japonicum, used to be widespread in Japan, China and the Far East and was the cause of widespread and gross morbidity and mortality. It affects not only man but also domestic and wild animals. S. japonicum is transmitted by an amphibious snail (species Oncomelania) which makes snail control relatively easy. There are two minor species of schistosomiais, First, S. intercalatum which is confined to West Africa and lives in the mesenteric vessels of man causing abdominal pain and bloody diarrhea. The second is S. mekongi, which is another form of intestinal schistosoma is found predominantly in Southeast Asia. The main reservoir for this species is dogs (Reich and Fenwick, 2001) as shown in Table 1.
LIFE CYCLE OF SCHISTOSOME
Schistosomiasis is a disease which is caused by various human-pathogenic trematodes belonging to the genus Schistosoma, more than 200 million people at present suffer from disease due to this parasitosis At the moment about 500-600 million people in 74 countries, that is approximately a tenth of the world population, are living with the risk of infection. Once in the human body, the cercariae develop further to schistosomula, schistosomes have separate sexes) (Urbani et al., 1997). Afterwards, the worms migrate upstream to their final destination within the venules of the bladder, rectum or colon.
| Table 1: | Illustrate the species of schistosomiasis; intermediate host; form of the disease and endemic area of each species |
 | |
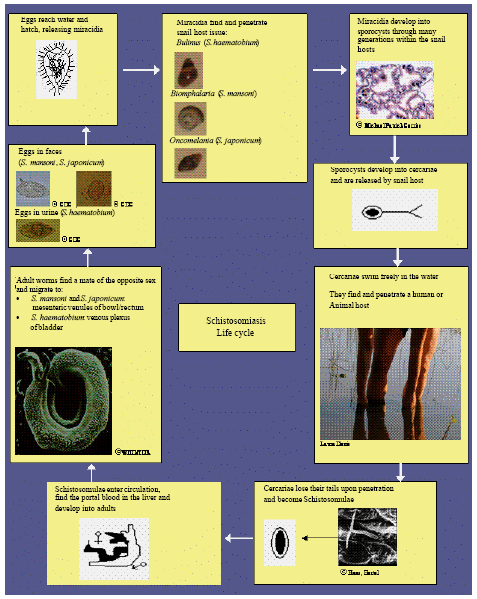 | |
| Fig. 1: | The Schistosomiasis life cycle |
Here, egg production starts (300 to 3000 day-1, depending on the species) (Bergquist, 2002) as shown in Fig. 1.
Inflammatory responses to the eggs lodged in the liver, bladder, or ureters rather than the worms themselves that cause the principal pathology associated with all forms of schistosomiasis (Davis et al., 1999). The shape and size of the eggs of the three major schistosome species are useful diagnostic features (Aka et al., 1999). Eggs reaching the liver are too large to reach the sinusoidal plexus and accumulate in presinusoidal venules within the portal triads, especially in the left lobe. There they induce granulomatous inflammation, fibrosis, venous obstruction, portal hypertension and splenomegaly (Stich et al., 1999). Liver enlargement initially tends to correspond in size to the intensity of concurrent or recent infections. Thus, hepatomegaly reflects granulomatous perioval inflammation rather than consequent fibrosis and occurs early in the evolution of chronic disease (Olds et al., 1996).
GEOGRAPHICAL DISTRIBUTION AND SPREAD OF THE DISEASE
Schistosomiasis is principally a disease of tropical and subtropical regions and is found in South and Central America, Africa, Asia and Southeast Asia. It is considered an endemic in 74 developing countries and more than 80% of infected people live in Africa (Anonymous, 2004). It has been estimated that 200 million people a year become infected (Keiser et al., 2002). A new focus of schistosomiasis infection is appearing in non-endemic areas as well as in areas where no autochthonous cases were reported for a long period of time. Examples are finding new foci of S.mansoni in Djibouti (Koeck et al., 1999) and reappearing of cases of S. haematobium in Jordan (Arbaji et al., 1998) present in Egypt, where control operation started many decades ago, land reclaimed along the Nile River has resulted in the introduction of schistosomiasis.
There are several factors that govern the outcome of the transmission process in areas where schistosomiasis is endemic such as the infection rates among snails and degree of human contact with water, which depends on social and cultural habits of the populations. Mammalian reservoirs of schistosomiasis In addition to human infection, Schistosoma species are also found in animals. They are responsible for transmission of the infection For S. japonicum group, The main animal reservoirs are cattle, buffalo, pig, dogs and rats. In the Philippines, animal reservoirs such as cow, water buffalo, dogs and pigs were responsible for 25% of environmental contamination. Animal infection with S. japonicum species were also reported from Indonesia and in Japan (WHO, 1990).
CONTROL OF SCHISTOSOMIASIS
Control through the intermediate host
Snail control: The control Schistosomiasis is through disrupting its life cycle. Snails, which are Schistosomiasis’s intermediate host, once destroyed, will prevent miracidiums from changing into cercariae (Yuan, 1989). To achieve this goal, the use of copper sulfate or copper carbonates material (Abel and Dessein, 1998) or Bromoacetamide (1c) to kill snails in swimming areas. For control of schistosomiasis, one strategy is based on the premise that snails resistant to parasitic infection could be used as biological competitors to replace existing susceptible snails in endemic areas (Yuan, 1989). This approach, however, requires a more thorough understanding of the genetics of the complex interrelationship between parasites and snails as shown Fig. 2. In which show the species of schistosomiasis and the different methods for controlling the schistosomiasis.
Environmental control: Schistosomiasis can only be transmitted by water contacts mostly for domestic and recreational purposes and also occupational (Aka et al., 1999). Therefore, providing safe water supply and sanitary facilities safe including safe drinking-water, washing facilities, cattle watering facilities and bathing, not only reduce the risk of infection with schistosomiasis, but also reduce the source of infection with other parasite and bacterial infections (Arene et al., 1998).
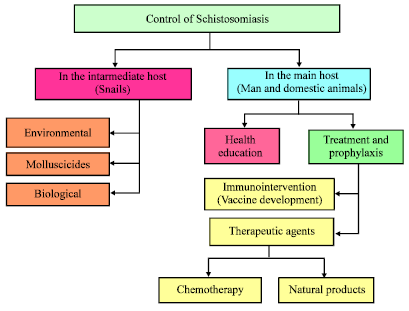 | |
| Fig. 2: | Diagrammatic feature of different methods for control schistosomiasis in the intermediate host and the main host |
Control by using molluscicides: The use of molluscicides for the control of the disease is also important as, unlike the use of synthetic drugs, it prevents the reinfection of people after treatment. Metallic salts, such as copper sulfate, were among the first agents used and were most effective when applied to standing bodies of water. Copper sulfate was introduced by dragging burlap sacks filled with large CuSO4 crystals behind slow moving boats. This compound worked well enough, but it also limited algal growth, that in turn affected growth patterns of fish that served as primary sources of protein (WHO, 1993). Newer molluscicides, such as nicotinanilide, organotin, dibromo-nitraozo-benzene, sodium pentachlorophenate, tritylmorpholine, sodium dichloro-bromopheno, niclosamide and acetamide analogs replaced copper sulfate, as these were deemed safer to the environment (Zhang et al., 1996).
Niclosamide used be a drug of choice for schistosomiaisis, but too many suffered from the same side effect of depletion of glycogen stores. This led in some cases to coma, an unacceptable outcome of treatment. Its use is limited by cost, as well. Plant-derived molluscicides have proven too variable in their effectiveness and are difficult to manufacture (Andrews et al., 1983).
Biological control: It was shown that Microsporidium sp. can interfere with the development of the sporocyst stage of S. mansoni. More of these kinds of associations most likely exist in nature and discovering them may result in the development of a useful adjunct to current control strategies (Dunne et al., 1995). A number of predator/competitor snail species are receiving more and more attention as potential control agents, as well. In well-controlled situations, such as small, artificial ponds. In Grenada, Martinique, Guadelupe, Puerto Rico and St. Lucia showed that Ampullariidae (Pomacea glauca and Marisa cornuarietis) and Thiaridae (Tarebia granifera and Melanoides tuberculata) snails out-competed Biomphalaria sp. for space and resources. Competitor snail species were also used successfully as a follow up measure after molluscicide use in some rivers of central Venezuela (Katz, 1998). Temperature can determine whether or not snails can reproduce. Below 100°C, which occurs usually in early spring in sub-tropical environments, reproduction is severely inhibited. Both adults and eggs succumb at temperatures that exceed 300°C (Zhang et al., 1990).
Control through the main host
Health education: Many countries lack the funds, or do not realize the inherent need to implement health education programs that inform the populace of ways to protect themselves from schistosomiasis (Zhen, 1993). Currently, in countries that have been able to implement such programs, they tend to emphasize the control of human behavior as they relate to the spread of schistosomiasis. Earlier versions of these programs stressed the need for snail control and the attendant reduction of transmission. Educated, informed people are able to adopt control strategies at the personal level, even if it requires reducing their contact with contaminated water sources, or making water safe to drink.
Treatment and prophylaxis
Vaccine development: Vaccine, ultimately, is anticipated to be the most effective form of Schistosomiasis treatment and control (Kojima, 2004). If proved to be effective and inexpensive enough for worldwide distribution, it would eliminate the need for snail and reservoir host control. Thus far, the vaccine based on irradiated cercariae offers almost complete protection in experimental animals (Doenhoff et al., 1988). The first generation vaccines were directed against infection and/or worm fecundity. Currently there is a natural balance, tempering anti-schistosomal responses by stimuli down-regulating the granulomatous reaction against eggs in the tissue (Capron et al., 1995). Present study promises to improve the understanding of cytokine interaction in the development of pathology and immunity, looking for a way to induce maximum levels of immunity without enhancing egg-associated reactions (Doenhoff, 1998). Fasciola and Schistosoma worm antigens mixed with or without saponin as well as saponin alone succeeded to protect mice against S. mansoni infection with more potent effect of the separately saponin and Fasciola antigens. This protection is achieved by reduction in total, male and female worms as well as the levels of toxins elaborated by them. This proved the role of these antigens in eliminating the product of oxidative stress and assistance in immune-mediated destruction of eggs that ameliorate the histopathological picture of the liver cells and preserve its function (Maghraby et al., 2010a). Schistosoma mansoni egg granuloma size reduction in liver section after vaccination with Fassiola or Schistosoma egg, in addition to Fassiola or Schistosoma egg with saponin antigens (Maghraby et al., 2010b).
As recombinant DNA techniques cannot yet be utilized to produce carbohydrate antigens it is more useful to focus on antigens which are predominantly of protein nature. In addition, carbohydrate antigens often cross-react with egg antigens and augment the risk of activating granulomatous reactions. The comparison between the relatively small granulomas of chronic schistosomiasis and those associated with early infection (Von Lichtenberg, 1987) and the fact that most anti-carbohydrate antibodies eventually become down-regulated (Omer-Ali et al., 1989), also emphasizes the usefulness of protein antigens. The list of published schistosome antigens, now numbering in excess of one hundred, comes from various parasite stages with the schistosomulum surface membrane being the preferential target (McKerrow et al., 1985).
Therapeutic agents
Chemotherapy: This is the most effective method for reducing the infection rates of schistosomiasis. Access to very effective drug in the last three decades has resulted in reducing the prevalence and morbidity of the disease in many areas of the world and reducing the public health importance of Schistosomisis in some countries (Doenhoff, 1998). Treatment of infected cases provides the most effective short-term results in control of schistosomiasis (Castro et al., 2002). It also reduces morbidity and rate of the transmission. Treatment of cases as a control method require proper planning such as defining objectives of treatment, using the most reliable case finding method, selection of appropriate drug, section of the population to be treated, correct dosage schedule and having adequate information on the drug and its side-effects (Cioli et al., 1995). Because case detection is the first approach for planning chemotherapy and selection of most effective chemotherapy, diagnostic techniques available will be described first. Although, several drugs were used for treatment of Schistosomiasis in the past, now only a few drugs are used. Introduction of praziquantel has transformed the treatment of schistosomiasis (Colley et al., 2001). This drug is effective, generally in a single dose, against all species of the parasite. Other drugs include metrifonate, which is active against S. haematobium and oxamniquine, which is effective against S. mansoni.
Synthetic antischistosomal drugs
Praziquantel: Praziquantel is a heterocyclic pyrazine-isoquinoline and is highly active against a wide range of trematodes, including all species of schistosome pathogenic to humans Praziquantel with a single dose of 40 mg kg-1 is effective. In the treatment of all forms of schistosomiasis in both adults and children. The cure rate is usually 60-90% with egg reductions of 90-95% in those not cured (Andrews, 1981) Praziquantel although generally well olerated, it may induce abdominal discomfort, bloody diarrhoea, nausea, headache, dizziness, urticaria and rectal bleeding in patients with heavy worm loads (Beck et al., 2001).
Resistance to praziquantel in the treatment of schistosomiasis has been reported from Egypt where the drug has been used aggressively for more that 10 years (Doenhoff et al., 2000; El-Banhawey et al., 2007; El-Ansary et al., 2007). There are some reports of the possibility of resistance of some strains of schistosomiasis to praziquantel (Fallon et al., 1995; Mandour et al., 1990) some believe this resistance does not exist (Ismail et al., 1999).
Metrifonate: Metrifonate is an organophosphorus compound originally used as an insecticide. It is well tolerated and is very effective in the treatment of S. haematobium mostly in mass chemotherapy programs (Danso-Appiah and De-Vlas, 2002). Because of the lower coast of metrifonate and the low side-effects, this drug is recommended for the mass-treatment of school children. Even when viable worms remain, egg counts after one year is reduced to less than 20% of pretreatment levels.
The effective dose in adults and children is 7.5 mg kg-1 on given three times at intervals of two weeks (Feldmeier and Chitsulo, 1999). Because of the side effect of this drug, metrifonate should not be used for mass chemotherapy in communities recently exposed to insecticides or other agricultural chemicals with an anticholinesterase action. After administration, of metrifonate abdominal pain, nausea, vomiting, diarrhoea, headache and vertigo may occur.
Oxamniquine: Oxamniquine, a tetrahydroquinoline derivative is used for the treatment of schistosomiasis due to S. mansoni both in the acute stage and in patients with hepatosplenic involvement. The effective dose varies between 15 and 60 mg kg-1 given over two to three days (Botros et al., 1989). Since oxamniquine has been known to cause seizures. Epileptic patients should remain under observation for several hours following treatment. It is used extensively in control programs in South America. Some strains of S. mansoni are resistant to this drug. Side effects of oxamniquine are dizziness, drowsiness, headache and raise of the levels of serum transaminases in some patients (Fallon and Doenhoff, 1994).
Artemisinin: There is some concern that large-scale use of an artemisinin derivative against Schistosomiasis might select for resistance in malaria parasites. Although, this risk appears to be low, mainly because of the very short elimination half-lives of artemisinins (Genovese et al., 2000; N'Goran et al., 1997, 2003), at present, use of these combinations should not be recommended in areas where both schistosome and malaria parasites coexist (Hastings et al., 2002; Xiao et al., 2002).
Virulence mechanisms contributing to the disease process: In infection by S. mansoni, the major pathologic changes are not caused by the adult worm itself but by eggs which do not reach the intestinal lumen, but instead, become trapped in other body tissues. At these sites, areas of local inflammation are produced, cumulating in the formation of granulomas around eggs (Giboda and Smith, 1994). The eggs of Schistosomoes are lethal to a human’s body system. When the body detects these eggs, an inflammatory reaction is triggered. Instead of killing the eggs, the eggs gain a protein capsule from the inflammatory cells, which in turn regulate its ovulation. The encapsulated eggs cause liver damage. Eggs undetected by the body’s defense system also harm the body. The embryos in the un-capsulated eggs release a toxin that damages the liver (Reynolds et al., 2002). The formation of granuloma around schistosome eggs in the liver and the intestine is the major cause of pathology in schistosome infections. Granuloma and the subsequent fibrosis in the liver appear to be primarily responsible for mortality and morbidity by this highly endemic parasitic disease (El-Banhawey et al., 2007). Ultrasound is used for detection of complications of infection in animals resulted in a marked decrease in liver glycogen (Akpinar and Metin, 1999). Visible to the human eye are the enlargement of the liver and spleen. Once symptoms have been noticed treatment can occur. In vivo microscopy revealed in addition to these lesions, dilatation and sacculation of sinusoids. These lesions were associated with varying degrees of reduction of blood flow due to schistosomules (El-Banhawy et al., 2007; Aly and Hamed, 2006). Enlargement of the spleen and especially liver will continue resulting development of the arteriovenous shunts in the lever. Ascites of the liver and esophageal varices may proceed. Among sever cases hepatomegaly and enlargement of the spleen are found in high number of cases (Talaat and Miller, 1998). Other clinical manifestations of chronic schistosomiasis include core pulmonale due to egg deposition in the lungs and subsequent development of pulmonary hypertension. Pulmonary complications of S. mansoni infection manifest with bronchopulmonary symptoms (Akpinar and Metin, 1999).
Total protein was reduced in bilharzail infection. This could be attributed to cellular damage caused by parasite toxins. The main fraction of total protein content is albumins and the reduction in total protein may be due to reduction in albumin fraction level that in turn may be result from decrease anabolism or increase catabolism; hence, malnutrition and/or malabsorption may contribute to decrease biosyntheses of albumin (El-Fakahani et al., 1993; Rizk et al., 2000; El-Ansary et al., 2007). The significant decrease in total protein is mainly due to increase in messenger RNA degradation which is the possible cause for the hypoalbuminemia of murine schistosomiasis (Metwally et al., 1990). Perioval granulomas in the liver lead to fibrosis.
The enlargement of the spleen may be attributed to the direct deposition of the eggs in that organ or due to inflammatory and fibrotic reactions in the splenic host that are the main factors responsible for obstruction to portal venous flow which its major consequence is splenomegaly, in histopathological examination, congestion was evident in sinusoids of red pulp and lymphoid follicles (White pulp) were enlarged. This marked congestion in red pulp showed evidence of haemorrhages (Aly and Hamed, 2006).
Natural product: The recent approach on the development of new drugs from natural products for treatment of human diseases especially in developing countries which still rely on traditional medicinal for their primary health care based largely from various species of plants (WHO, 2002).
There is still intensive search for effective anti-schistosomal drugs with minimal side effects (El-Banhawey et al., 2007). Plants are the basis of traditional medicine systems that have been in existence for thousands of years and continue into modern times (Jellin et al., 2000). The antibiotic penicillin being the most well known. In addition to plants and microbes there has been growing interest in the role of animals as sources of medicines including products derived from frogs and from marine snails (Duke, 2002).
Natural products also have considerable value as insecticides, contributing to human health both through improved agricultural (and hence food) productivity and in the control of insect-borne diseases. Several other herbs are traditionally used for treatment of parasites, including male fern (Dryopteris filix mas) root, tansy (Tanacetum vulgare) leaf, wormwood, sweet annie, black walnut (Juglans nigra) fruit and cloves (Syzygium aromaticum) (Bruneton, 1999).
Allium sativum: The antiparasitic nature of Allium sativum (garlic) is demonstrated in the uses to which it has been applied in folk medicines around the world. For example, it has been traditionally used to treat parasitic worms in such diverse cultures as East Asia, India, Italy, North America, Peru, Saudi Arabia, Tunisia and the West Indies. Traditional practitioners in Greece have long used garlic extracts to protect against amoebic infections (Mirelman et al., 1987).
Garlic has been demonstrated to kill parasites, including amoeba (Mirelman et al., 1987) and hookworm (Bastidas, 1969) in test tubes and in animals. Older studies in humans support the use of garlic to treat roundworm, pinworm and hookworm (Koch and Lawson, 1996). However, due to a lack of clinical trials, the amount of garlic needed to treat intestinal parasites in humans is not known.
Wormseed: Wormseed (Chenopodium ambrosioides) is a traditional remedy for infections with worms. However, a study in Mexico found that the powdered herb was not effective at eradicating hookworm, roundworm, or whipworm (Kliks, 1985).
Pumpkin seeds: Pumpkin seeds (Cucurbita pepo) have purported effects against tapeworms. Pumpkins and other squashes are native to North and Central America, but have since been cultivated around the world (Oliver et al., 2003). The seeds are primarily used in herbal medicine; the yellow blossoms of pumpkins are also used as medicine in some native. Active constituents: Pumpkin seeds contain several major groups of active constituents: essential fatty acids, amino acids, phytosterols (e.g., beta-sitosterol) minerals and vitamins. Other major constituents include mucilaginous carbohydrates and minerals (Sheir et al., 2001). Curcurbitin is a constituent in pumpkin seeds that has shown anti-parasitic activity. In China, pumpkin seeds have been shown to effectively treat acute Schistosomiasis, a severe parasitic disease occurring primarily in Asia and Africa that is transmitted by snails (Weiss, 1985).
Anise: Anise may have modest antiparasitic actions and has been recommended by some practitioners as a treatment for mild intestinal parasite infections 74 (Weiss, 1985).
Olive leaf: Olive leaf has been used in traditional medicine to reduce fever, blood sugar, blood pressure and as a diuretic (Privitera, 1996). In 1854, the Pharmaceutical Journal contained an article outlining its use to counter cases of fever and malaria (Bruneton, 1999). Active constituents: Olive leaf has a wide number of constituents, including oleuropein and several types of flavonoids (e.g., rutin, apigenin, luteolin. Olive leaf is listed in Duke's Handbook of Medicinal Herbs as antibacterial, antioxidant and a hypoglycemic, with indications against such diverse conditions as malaria, lymphtic disorders and Schistosomiasis (Duke, 2002).
Vernonia amygdalina leaf: The curative and prophylactic effects of petroleum ether and ethanolic leaf extracts of Vernonia amygdalina Del (family compositae) have potential curative effects on kidney, liver and spleen experimental Schistosomiasis in mice (Ogboli et al., 2000).
Lapachol: D’Arco (1996-2003) and its constituents have demonstrated antiviral properties against various viruses including Herpes I and II, influenza, poliovirus and vesicular stomatitis virus. Its anti-parasitic actions against various parasites including malaria, Schistosoma and Trypansoma have been clinically validated (Gilbert et al., 1970; Cheever, 1997). Lapachol, a chief constituent of the wood and bark of the pau d'arco tree, has anti-inflammatory, antimalarial, antibacterial, antifungal, antiparasitic and immunomodulatory activity (Austin, 1979; Jellin et al., 2000), many of which have been backed up by results from animal and other laboratory studies (Foster and Tyler, 1999).
Echinacea: Echinacea is used for a range of benefits, including as an antiviral, an immune stimulant and to relieve urinary tract infections and yeast-related disorders. Extracts from Echinacea purpurea add to the body's resistance to bacterial and viral infection (Jellin et al., 2000; Bruneton, 1999) and have shown indirect antiviral activity (Privitera, 1996).
Peppermint: Curled mint (Mentha crispa) leaf, a close relative of peppermint, has been shown in a preliminary trial to help relieve the symptoms of giardia and amoeba infections in children and adults, as well as to eliminate these parasites in many cases (Santana et al., 1992). This study used a tincture of curled mint in the amount of 2 mL three times per day for five days, or 1 mL three times per day for five days for children. Given their close relationship, peppermint could probably be substituted for curled mint when curled mint is unavailable.
Myrrh: Myrrh is an oleo-gum resin from the stem of the plant Commiphora mol. A new trend for treatment of liver disorders as a result of S. mansoni infection is the use of natural plant extract of Commiphora (Mirazid) (Sheir et al., 2001). Commiphora extract (Mirazid) has been proved to be safe antifasciolicidal drug without any side effect (Hassan et al., 2003). Moreover, it's very effective in treatment of Schistosoma haematobium (El-Baz et al., 2003). Also, Massoud et al. (2004) reported that Mirazid caused disruption of S. mansni worms' tegument and collapse of tubercles causing eradication in worm burden. Natural Purified Commiphora plants extract caused significant increase in ATP, TA, Pi, glucose, glycogen, adenosine deaminase and protein, after reduction caused by infection, this enhancing ability of the extract may be related to antioxidantive activities (Aly and Aly, 2006). In various reports concerning S. mansoni, Sheweita et al. (1998) pointed out that levels of reduced glutathione and glutathione reductase increased, while the activity of glutathione-S-transferase decreased in human and mice infected with S. mansoni. In this respect S. mansoni infection alters and consumes the hepatic levels of glutathione, superoxide dismutase, catalase and glutathione metabolizing enzymes (antioxidant system) and these alterations may affect the capacity of the liver to detoxify or neutralize the effect of toxic endogenous and exogenous compounds (Van Waarde et al., 1990).
Citrus reticulata: Citrus has been reported to have anti-leukemia (Mak et al., 1999), inhibited human cancer cell proliferation (Tian et al., 2001) antibacterial activity (Jayaprakasha et al., 2000), antioxidants activity (Tanizawa et al., 1992). Antibiotic properties (Tkachenko et al., 1999), antimicrobial and antibacterial activities (Jayaprakasha et al., 2000), respectively. Thus the significant improvement of Citrus plants on all the previous mentioned parameters in infected mice may be attributed to citrus fruits contain high concentrations of several polymethoxylated flavones (Demirci et al., 2000). The latter group of compounds occurs without glycosidic classes of phenols, including numerous hydroxycinnamates, flavonoid glycosides and linkages and has been shown to inhibit the proliferation of number of cancer and protect protein against six oxidative damage (Manthey and Guthrie, 2002). The anti-inflammatory activities of citrus flavonoids a rise from the antioxidant properties of these compounds (Manthey et al., 2001). Antibiotic, free radical scavenger, anti-leukemia and antibacterial activities (Akira et al., 2000), respectively. In addition, the citrus seeds and its flours were rich in oil and protein and proved to be a good source for minerals K, Ca, P, Na, Fe and Mg (Sabah et al., 1986). The essential oil of citrus plants was shown to possess also antimicrobial activities (Demirci et al., 2000). Eradication of the number of egg count and worm burden is shown in infected mice treated with Mirazid more than citrus plants extracts which give an additional support for the curative effects of both extracts (Aly and Aly, 2006).
Ailanthus altissima: The chloroform extract of Ailanthus altissima stem bark wide range of biological activities showed a pronounced improving effect against organs (liver-kidney-spleen) damage caused by parasitic infection (Aly and Hamed, 2006). A. altissima possesses antituberculosic activity antiplasmodial activity (Okunade et al., 2003) and antitumor activity (Tamura et al., 2003).
Zizyphus spina-christi: The ethanolic extract of Zizyphus spina-christi root shows antidiabetic activity, antimicrobial activity (Shahat et al., 2001) and antidiarrheal activity (Adzu et al., 2003) antischistosomal activity (Aly et al., 2006; El-Rigal et al., 2006). Caused reduction in the number of worm burden, ova count granuloma size and count as well as improvement in the histopathological picture of liver, kidney and spleen of infected mice (Aly and Hamed, 2006).
Berberine: Berberine is derived from several plants, including barberry, Oregon grape, goldenseal and goldthread (Coptis chinensis). Preliminary trials have shown that berberine can be used successfully to treat giardia infections (Choudhry et al., 1972; Gupte, 1975). In addition, test tube studies show that berberine kills amoebae, although it is not known whether this effect occurs in humans (Kaneda et al., 1991; Miyares et al., 1988). The amount required is approximately 200 mg 3 times per day for an adult-a level high enough to potentially cause side effects. Therefore, berberine should not be used without consulting a doctor.
Ipecac: Emetine and other alkaloids in ipecac kill several types of parasites; including amoeba, pinworms and tapeworms (Oelkers, 1962 ; Wright and Phillipson, 1990). Generally, the amounts of ipecac needed to produce these effects in people are high and can lead to severe side effects. Emetine or its somewhat safer form, dihydroemetine, are reserved for rare cases of people infected with amoebae who are not cured by using anti-amoeba drugs (Schmeller and Wink, 1998) because of the danger involved, ipecac and emetine should never be use.
Wormwood: Wormwood general strengthening herb that benefits the whole body, while stimulating and invigorating the whole digestive process. Helps the body deal with infections helps kill both the egg and adult stages of over 100 parasites including giardia (affecting more and more people), amoebas, many worms and liver flukes (Kaneda et al., 1991). Other traditional applications include regulating menstruation and reducing fever (Schmeller and Wink, 1998). Duke's handbook of Medicinal Herbs lists antibacterial and antifungal properties for wormwood (Bastidas, 1969).
Rhizoma rhei: Rhizoma rhei effective against Schistosomiasis (parasitic disease from fresh water snails infecting over 200 million people in 74 countries) (Duke, 2002). Mild natural laxative which evacuates parasites, cryptosporidium (protozoa) and toxins.
Sarsaparilla: Sarsaparilla strengthens the bones and muscles. This herb is used to treat urinary tract infections, rheumatoid arthritis, boils, abscesses and to neutralize mercury toxicity (Colley et al., 2001).
Rhubarb root-rheum officinale: Rhubarb root purges the body of bile, parasites and a stagnating food by stimulating the gall duct to expel toxic waste matter. It has been shown to alleviate chronic liver problems by cleansing the liver (Wright and Phillipson, 1990).
Thyme: Traditionally it is the thyme leaf and flowering tops that have been used therapeutically. In folk medicine thyme is used to stimulate the appetite, suppress coughing and relieve digestive disorders such as chronic gastritis, diarrhoea in children and flatulence. It is also used to expel parasitic worms (Miyares et al., 1988; Gupte, 1975; Oelkers, 1962), particularly in children.
Black walnut: Black walnut has been used in folk medicine as an astringent, laxative and a vermifuge. It is used to expel tapeworms and other internal and external parasites (Choudhry et al., 1972). The American Medical Ethnobotany Reference Dictionary claims that the juice from black walnut hull is effective against ringworm, but some warnings have been issued regarding the topical use of this herb. Black walnut is traditionally regarded as being antiparasitic and a vermifuge (kills worms) (Schmeller and Wink, 1998; Sheir et al., 2001).
Pulicaria crispa: The ethanolic extract of natural medicinal plant Pulicaria crispa have an immunostimulatory effect against schistosomiasis before and after S. mansoni infection. Al-Yahya et al. (1988) added that it has a chemopreventive activity against cancer diseases. In addition, the plant extract of Pulicaria incisa given orally to normal rats showed transient hypoglycemic effects only 1 h after administration (Shabana et al., 1990). Moreover, ethanolic extracts of pulicaria orientalis and butanol extracts of aerial part of Plucaria gnaphaloides (PG) showed antibacterial activity against both Gram-positive and Gram-negative bacteria as well as antifungal activities (Ali et al., 2001; Mahasneh, 2002). Pulicaria crispa plants extract give an additional support for the protection role against schistosomiasis. Mice pre-treated with Pulicaria crispa extract showed no side effects of most parameters compared to the normal healthy control group (Hamed et al., 2004).
Eitharexlum quadrangular Jacq.: Ethanolic extract of Citharexylum quadrangular Jacq receded also immuonmodulatory effect against schistosomiasis before and after S. mansoni infection (Shalaby and Bahgat, 2003). The same outers added that, this plant extract showed also antimicrobial effect against E. coli, C. albicans and Batrytis alli. It can be applied clinically as a prophylactic treatment against schistosomiasis together with the ideal anti-schistosomal drug praziquantel. Significant amelioration was noticed in the levels of liver function enzyme activities in S. mansoni infected mice as a result of prophylactic treatment with. Citharexylum quadrangular Jacq with significant reduction of worm burden and ova count. Hence, both plant extracts can be applied clinically as a prophylactic treatment against schistosomiasis (Hamed et al., 2004).
Curcuma longa: Curcuma longa extract as a plant with recently reported many medicinal properties (Olajide, 1999). It was tested as antibilharzial drug. The obtained data proved that C. longa extract was efficient in the repletion of the depleted glycogen reserves and induced a significant elevation of glucose concentration in control and infected C. longa-treated animals (Rizk et al., 2000). The potential activity of this plant extract in inducing glycogen and glucose levels could be easily correlated to the previous reports of El-Ansary and Farouk (2001). They reported that C. longa extract was effective in restoring normal Adenylate Energy Charge (AEC), through the activation of the oxidative phosphorylation pathway. Stimulation of oxidative phosphorylation as the main ATP-generating pathway could explain the glycogen. Higher glycogen reserves in C. longa-treated control animals could acertain the mode of action of this extract (El-Banhawey et al., 2007).
Curcumin, obtained from powdered rhizomes of plant Curcuma longa Linn., is commonly used as coloring agent in food, drugs and cosmetics (Chuang et al., 2000). Curcuma longa extract as a remarkable non-toxic plant with many medicinal properties should be explored for possible intervention in schistosomiasis as a disease involves impairment of metabolism of infected subjects (El-Ansary et al., 2007).
Curcumin inhibits cancer at initiation, promotion and progression stages of tumor development (Radhakrishna et al., 2004; Mohanty et al., 2004). Research in Germany and India shows that curcumin can also help prevent gallbladder disease (Olajide, 1999; Chuang et al., 2000).
Capparis spinosa and Acacia arabica: Molluscicides may induce various pathological manifestations to the snails which could render the molluscan hosts less suitable to parasitic infection (El-Ansary et al., 2001). The powdered activity of Capparis spinosa and Acacia arabica showed molluscicidal activity against Biomphalaria alexandrina specific intermediate hosts to Schistosoma mansoni. C. spinosa has antimicrobial activity against some bacterial and some fungal species (Mahasneh, 2002). Lyophilized extract of C. spinosa showed a significant antioxidant effect (Bonina et al., 2002; Germano et al., 2002) predicted through measuring some glycoclytic related parameters such as lactate/pyruvate ratio, glycogen and adenine nucleotides, that glycolysis as an emergency pathway for generating ATP is of critical importance for trematode-infected, B. alexandrina (Aly et al., 2004).
Endod: Endod berries, thesource of the molluscicidal saponins. Although, we now know that preparation and application of the endod berries is a safe procedure and that the molluscicidal saponins are decomposed to water and carbon dioxide in the environment, some outstanding questions are still to be solved. One of the future projects should deal with ecotoxicology in respect of the biological impact of the Endod berries on the environment. Toxicity testing of endod, a natural plant extract, as a prerequisite for its safe use as a molluscicide (Lemma, 1965, 1970). The berries may be less detrimental to the ecosphere than are the synthetic molluscicides; however, they still are harmful to life in the water (Lemma et al., 1983). Also snails that are not host snails for schistosomes and to fish. The dead snails and fish are not poisonous to their predators or scavengers, but the lack of prey for some time may influence at least part of the river system (Amusan et al., 1995).
CONCLUSION
In conclusion, the author proposed that natural product extracts with non-toxic medicinal properties should be explored for possible intervention in schistosomiasis as a disease involving impairment of metabolism of infected subjects. These inspire more hope for reducing the intensity of schistosomal infection by reduction in worm burden, ova count, granuloma size and number leading to improvement in histopathological picture of liver, spleen and kidney as a result of reducing inflammatory and fibrotic reactions of schistosoma.
REFERENCES
- Abel, L. and A.J. Dessein, 1998. Genetic epidemiology of infectious diseases in humans: Design of population-based studies. Emerg. Infect. Dis., 4: 1-13.
PubMed - Aka, N.A., A.C. Allabi, G. Dreyfuss, D. Kinde-Gazard and L. Tawo et al., 1999. Epidemiological observations on the first case of human paragonimiasis and potential intermediate hosts of Paragonimus sp. in 32-Benin. Bull. Soc. Pathol. Exot., 92: 191-194.
PubMed - Akpinar, M.A. and K. Metin, 1999. The amount of glycogen in the liver and muscle tissues of starved and fed oncorhy-nchus mykiss. Turk. J. Biol., 23: 107-113.
Direct Link - Awadh Ali, N.A., W.D. Julich, C. Kusnick and U. Lindequist, 2001. Screening of Yemeni medicinal plants for antibacterial and cytotoxic activities. J. Ethnopharmacol., 74: 173-179.
CrossRefPubMedDirect Link - Aly, H.F. and S.A. Aly, 2006. Essential role of Citrus reticulata and Mirazid in treatment of Schistosoma mansoni infected mice: Biochemical and parasitological studies. Pol. J. Food Nutr. Sci., 15: 461-467.
Direct Link - Ali, S.A. and M.A. Hamed, 2006. Effect of Ailanthus altissima and Zizyphus spina-christi on bilharzial infestation in mice: Histological and histopathological studies. J. Applied Sci., 6: 1437-1446.
CrossRefDirect Link - Aly, S.A., H.F. Aly, N. Saba-el-Rigal and E.M. Sammour, 2004. Induced changes in biochemical parameters of the molluscan tissues non-infected using two potent plants molluscicides. J. Egypt Soc. Parasitol., 34: 527-542.
PubMed - Aly, S.A., N.S. El-Rigal and M.Z. Rizk, 2006. Nutritional supplementation with Ailanthus altissima and Ziziphus spina christi to compensate for some metabolic disorders in Schistosoma mansoni infected mice. Pak. J. Biol. Sci., 9: 1700-1706.
CrossRefDirect Link - Al-Yahya, M.A., A.M. El-Sayed, J.S. Mossa, J.F. Kozlowski and M.D. Antoun et al., 1988. Potential cancer chemopreventive and cytotoxic agents from Pulicaria crispa. J. Nat. Prod., 51: 621-624.
PubMed - Anonymous, 2004. Who`s next in line? Succession management planning and leadership development go hand in hand. Strategic Direction, 20: 30-32.
CrossRef - Arbaji, A., Z.S. Amr, A.A. Abbas, R. Al-Oran, S. Al-Kharabsheh and W.N. Al-Melhim, 1998. New sites of Bulinus Truncatus and indigenous cases of urinary schistosomiasis in Jordan. Parasite, 5: 379-382.
PubMed - Arene, F.O.I., E.I. Ibanga and J.E. Asor, 1998. Epidemiology of paragonimiasis in Cross River basin, Nigeria: Prevalence and intensity of infection due to Paragonimus uterobilateralis in Yakurr local government area. Public Health, 112: 119-122.
CrossRef - Austin, F.R., 1979. Schistosoma mansoni chemoprophylaxis with dietary lapachol. Am. J. Trop. Med. Hyg., 23: 412-419.
PubMed - Adzu, B., S. Amos, M.B. Amizan and K. Gamaniel, 2003. Evaluation of the antidiarrhoeal effects of Zizyphus spina christi stem bark in rats. Acta Trop., 87: 245-250.
CrossRef - Bastidas, C.J., 1969. Effect of ingested garlic on Necator americanus and Ancylostoma caninum. Am. J. Trop. Med. Hyg., 13: 920-923.
PubMed - Beck, L., T.C. Favre, O.S. Pieri, L.C. Zani, G.G. Domas and C.S. Barbosa, 2001. Replacing oxamniquine by praziquantel against Schistosoma mansoni infection in a rural community from the sugar-cane zone of northeast Brazil: An epidemiological follow-up. Mem. Inst. Oswaldo Cruz., 96: 165-167.
PubMed - Bergquist, N.R., 2002. Schistosomiasis: From risk assessment to control. Trends Parasitol., 18: 309-314.
CrossRef - Bonina, F., C. Puglia, D. Ventura, R. Aquino and S. Tortora et al., 2002. In vitro antioxidant and in vivo photo protective effects of a lyophilized extract of Capparis spinosa L. buds. J. Cosmet. Sci., 53: 321-335.
PubMed - Botros, S.A., N. Soliman, M. El-Gawhary, M. Selim and N. Guirguis, 1989. Effect of combined low dose praziquantel and oxamniquine on different stages of schistosome maturity. Trans. R. Soc. Trop. Med. Hyg., 83: 86-89.
CrossRef - Capron, A., G. Riveau, J.M. Grzych, D. Boulanger, M. Capro and R. Pierce, 1995. Development of a vaccine strategy against human and bovine schistosomiasia. Background and update. Mem Inst Oswaldo Cruz, 90: 235-240.
PubMed - Castro, N., H. Jung, R. Medina, D. Gonzalez-Esquivel, M. Lopez and J. Sotelo, 2002. Interaction between grapefruit juice and praziquantel in humans. Antimicrob. Agents Chemother., 46: 1614-1616.
PubMed - Cheever, A.W., 1997. Differential regulation of granuloma size and hepatic fibrosis in schistosome infections. Mem. Inst. Oswaldo Cruz, 92: 689-692.
CrossRef - Choudhry, V.P., M. Sabir and V.N. Bhide, 1972. Berberine in giardiasis. Indian Pediatr., 9: 143-146.
Direct Link - Chuang, S., A. Cheng, J. Lin and M. Kuo, 2000. Inhibition by curcumin of diethylnitrosamine-induced hepatic hyperplasia, inflammation, cellular gene products and cell-cycle-related proteins in rats. Food Chem. Toxicol., 38: 991-995.
CrossRefPubMedDirect Link - Cioli, D., L. Pica-Mattoccia and S. Archer, 1995. Antischistosomal drugs: Past, present and future? Parmacol. Ther., 68: 35-85.
CrossRef - Colley, D.G., P.T. Lo-Verde and L. Savioli, 2001. Medical helminthology in the 21st century. Science, 293: 1437-1438.
PubMed - Danso-Appiah, A. and S.J. de Vlas, 2002. Interpreting low praziquantel cure rates of Schistosoma mansoni infections in Senegal. Trends Parasitol., 18: 125-129.
CrossRefPubMedDirect Link - Davis, G.M., T. Wilke, Y. Zhang, X. Xu and Q. Chi-Ping Spolsky, 1999. Snail-Schistosoma, Paragonimus interactions in China: Population ecology, genetic diversity, coevolution and emerging diseases. Malacologia, 41: 355-377.
Direct Link - Doenhoff, M.J., 1998. Is schistosomicidal chemotherapy sub-curative? Implications for drug resistance. Parasitol. Today, 14: 434-435.
CrossRef - Doenhoff, M.J., J. Modha an J.R. Lambertucci, 1988. Anti-schistosome chemotherapy enhanced by antibodies specific for a parasite esterase. Immunology, 65: 507-510.
PubMedDirect Link - Dunne, D.W., P. Hagan and F.G. Abath, 1995. Prospects for immunological control of schistosomiasis. Lancet, 345: 1488-1491.
PubMed - El-Ansary, K.A., S.A. Ahmed and S.A. Aly, 2007. Antischistosomal and liver protective effects of Curcuma longa extract in Schistosoma mansoni infect mice. Indian J. Exp. Biol., 45: 791-801.
PubMed - El-Rigal, N., S. Aly, M.Z. Rizk and A. Said, 2006. Effect of Ailanthus altissima and Ziziphus spina christi extracts on some hepatic marker enzymes and antioxidants in Schistosoma mansoni infected mice. Polish J. Food Nutr. Sci., 15: 199-202.
Direct Link - El-Banhawey, M.A., M.A. Ashry, A.K. EL-Ansary and S.A. Aly, 2007. Effect of Curcuma longa or parziquantel on Schistosoma mansoni infected mice liver-histological and histochemical studies. Indian J. Exp. Biol., 45: 877-889.
PubMed - El-Baz, M.A., T.A. Morsy, M.M. El-Bandary and S.M. Motawea, 2003. Clinical and parasitologicals studies on the efficacy of Mirazid treatment of Schistosomiasis haematobium in Tatoon, Etsa Center, El Fayoum Governorate. J. Egypt. Soc. Parasitol., 33: 761-776.
PubMed - El-Fakahani, A.F.M., K.F. Abdella, H.M. El-Hadi, S.M. Abd-El Aziz and L.M. Afifi, 1993. The effect of praziquantel treatment on the liver functions, worm burden and granuloma size using two drug regimens in murine Schistosoma mansoni infection. J. Egypt. Soc. Parasitol., 23: 877-886.
PubMed - Fallon, P.G., R.F. Sturrock, A.C. Niang and M.J. Doenhoff, 1995. Short report: Diminished susceptibility to praziquantel in a Senegal isolate of Schistosoma mansoni. Am. J. Trop. Med. Hyg., 53: 61-62.
PubMedDirect Link - Fallon, P.G. and M.J. Doenhoff, 1994. Drug-resistant schistosomiasis: Resistance to praziquantel and oxamniquine induced in Schistosoma mansoni in mice is drug specific. Am. J. Trop. Med. Hyg., 51: 83-88.
PubMed - Feldmeier, H. and L. Chitsulo, 1999. Therapeutic and operational profiles of metrifonate and praziquantel in Schistosoma haematobium infection. Arzneimittelforschung, 49: 557-565.
PubMed - Demirci, F., G. Işcan, K. Guven, N. Kirimer, B. Demirci and K.H. Başer, 2000. Antimicrobial activities ferulago essential oils. Z. Naturforsch. C, 55: 886-889.
PubMed - Genovese, R.F., D.B. Newman and T.G. Brewer, 2000. Behavioral and neural toxicity of the artemisinin antimalarial, arteether, but not artesunate and artelinate, in rats. Pharmacol. Biochem. Behav., 67: 37-44.
CrossRefPubMedDirect Link - Germano, M.P., R. De-Pasquale, V. D`Angelo, S. Catania, V. Silvari and C. Costa, 2002. Evaluation of extract and isolated fraction from Capparis spinosa L. buds as an antioxidant source. J. Agric. Food. Chem., 50: 1168-1171.
CrossRef - Giboda, M. and J.M. Smith, 1994. Schistosoma mansoni eggs as a target for praziquantel: Efficacy of oral application in mice. J. Trop. Med. Hyg., 97: 98-102.
PubMed - Jayaprakasha, G.K., P.S. Negi, S. Sikder, L.J. Rao and K.K. Sakariah, 2000. Antibacterial activity of citrus reticulata peel extracts. J. Biosci., 55: 1030-1034.
PubMed - Gupte, S., 1975. Use of berberine in treatment of giardiasis. Am. J. Dis. Child., 129: 866-866.
PubMed - Hastings, I.M., W.M. Watkins and N.J. White, 2002. The evolution of drug-resistant malaria: The role of drug elimination half-life. Philos. Trans. R. Soc. London Ser. B Biol. Sci., 357: 505-519.
CrossRef - Hassan, M., M. El-Motaiem, H. Afify, B. Abaza, M. El-Shafei and A. Massoud, 2003. In vitro effect of Mirazid on Schistosoma mansoni in worms. J. Egypt. Soc. Parasitol., 33: 999-1008.
PubMed - Mohanty, I., D.S. Arya, A. Dinda, S. Joshi, K.K. Talwar and S.K. Gupta, 2004. Protective effects of Curcuma longa on ischemia-reperfusion induced myocardial injuries and their mechanisms. Life Sci., 75: 1701-1711.
PubMed - Ismail, M., S. Botros, A. Metwally, S. William and A. Farghally et al., 1999. Resistance to praziquantel: Direct evidence from Schistosoma mansoni isolated from Egyptian villagers. Am. J. Trop. Med. Hyg., 60: 932-935.
PubMed - Kaneda, Y., M. Torii, T. Tanaka and M. Aikawa, 1991. In vitro effects of berberine sulphate on the growth and structure of Entamoeba histolytica, Giardia lamblia and Trichomonas vaginalis. Ann. Trop. Med. Parasitol., 85: 417-425.
PubMed - Keiser, J.E.K., B.H. N'Goran, C. Singer, M. Lengeler, Tanner and J. Utzinger, 2002. Association between Schistosoma mansoni and hookworm infections among schoolchildren in Cote d'Ivoire. Acta Trop., 84: 31-41.
CrossRef - Kliks, M.M., 1985. Studies on the traditional herbal anthelmintic Chenopodium ambrosioides L.: Ethnopharmacological evaluation and clinical field trials. Soc. Sci. Med., 21: 879-886.
PubMed - Kojima, S., 2004. Overview: from the horse experimentation by Prof. Akira Fujinami to paramyosin. Parasitol. Int., 53: 151-162.
CrossRef - Koeck, J.L., C. Modica, F. Tual, E. Czarnecki and R. Fabre et al., 1999. Discovery of a focus of intestinal bilharziasis in te republic of Djibouti Med. Trop., 59: 35-38.
PubMed - Lemma, A., 1970. A laboratory and field evaluation of the molluscicidal property of P. dodecandra. Bull. World Health Org., 42: 597-612.
Direct Link - Mahasneh, A.M., 2002. Screening of some indigenous Qatari medicinal plants for antimicrobial activity. Physiother. Res., 16: 751-753.
CrossRefDirect Link - Manthey, J.A. and N. Guthrie, 2002. Anti proliferative activities of Citrus flavonoids againt six human cancer cell lines. Agric. Food Chem., 50: 5837-5843.
PubMed - Manthey, J.A., N. Guthrie and K. Grohmann, 2001. Biological properties of citrus flavonoids pertaining to cancer and inflammation. Curr. Med. Chem., 8: 135-153.
CrossRefPubMedDirect Link - Massoud, A.M., N.M. El-Kholy, F.A. El-Shennawy and R.E. Farag, 2004. Study of some immune aspects in patients with fascioliasis before and after Chommiphora molmol (Mirazid) treatment. J. Egypt. Soc. Parasitol., 34: 315-332.
PubMedDirect Link - Mandour, M.E., H. El-Turabi, M.M.A. Homeida, T. El-Sadig and H.M. Ali et al., 1990. Pharmacokinetics of praziquantel in healthy volunteers and patients with schistosomiasis. Trans. R. Soc. Trop. Med. Hyg., 84: 389-393.
CrossRef - McKerrow, J.H., P. Jones. H. Sage and S. Pino-Heiss, 1985. Purification and characterization of an elastinolytic proteinase secreted by cercariae of Schistosoma mansoni. J. Biol. Chem., 260: 3703-3704.
PubMed - Mirelman, D., D. Monheit and S. Varon, 1987. Inhibition of growth of Entamoeba histolytica by allicin, the active principle of garlic extract (Allium sativum). J. Infect. Dis., 156: 243-244.
PubMed - Miyares, C., I. Hollands and C. Castaneda, 1988. Clinical trial with a preparation based on propolis propolisina in human giardiasis. Acta Gastroenterol. Latinoam., 18: 195-201.
PubMed - N'Goran, E.K., S. Diabate, J. Utzinger and B. Sellin, 1997. Changes in human schistosomiasis level after the construction of two large hydroelectric dams in central Cote d'Ivoire. Bull. World Health Org., 75: 541-545.
PubMed - N′Goran, E.K., J. Utzinger, H.N. Gnaka, A. Yapi and N.A. N′Guessan et al., 2003. Randomized, double-blind, placebo-controlled trial of oral artemether for the prevention of patent Schistosoma haematobium infections. Am. J. Trop. Med. Hyg., 68: 24-32.
PubMed - Ogboli, A.U., I.H. Nock, E.M. Obdurahman and N.D.G. Ibrahim, 2000. Medicinal application of vernonia amygdalina del leaf extracts in the treatment of schistosomiasis in mice. Nig. J. Nat. Prod. Med., 4: 73-75.
Direct Link - Okunade, A.L., R.E. Bikoff, S.J. Casper, A. Oksman, D.E. Goldberg and W.H. Lewis, 2003. Antiplasmodial activity of extracts and quassinoids isolated from seedlings of ailanthus altissima (simaroubaceae). Phytother. Res., 17: 675-677.
Direct Link - Olajide, O.A., 1999. Investigation of the effects of selected medicinal plants on experimental thrombosis. Phytother. Res., 13: 231-232.
PubMedDirect Link - Olds, G.R., R. Olveda, G. Wu, P. Wiest and S. Mc-Garvey et al., 1996. Immunity and morbidity in schistosomiasis japonica infection. Am. J. Trop. Med. Hyg., 55: 121-126.
PubMed - Pillai, G.R., A.S. Srivastava, T.I. Hassanein, D.P. Chauhan and E. Carrier, 2004. Induction of apoptosis in human lung cancer cells by curcumin. Cancer Lett., 208: 163-170.
PubMed - Reynolds, S.R., C.B. Shoemaker and D.A. Harn, 2002. T and B cell epitope mapping of Sm23, an integral membrane protein S. mansoni. J. Immunol., 149: 3995-4001.
PubMed - Shabana, M.M., Y.W. Mirhom, A.A. Genenah, E.A. Aboutabl and H.A. Amer,1990 1990. Study into wild Egyptian plants of potential medicinal activity Ninth communication: Hypoglycaemic activity of some selected plants in normal fasting and alloxanised rats. Arch. Exp. Veterinarmed., 44: 389-394.
PubMed - Shahat, A.A., L. Pieters, S. Apers, N.M. Nazeif, N.S. Abdel-Azim, D.V. Berghe and A.J. Vlietinck, 2001. Chemical and biological investigations on Zizyphus spina-christi L. Phytother. Res., 15: 593-597.
CrossRefPubMedDirect Link - Sheir, Z., A.A. Nasr. A. Massoud, O. Salama and G.A. Badra et al., 2001. A safe, effective, herbal anti-schistosomal therapy derived from myrrh. Am. J. Trop. Med. Hyg., 65: 700-704.
PubMed - Sheweita, S.A., S.A. Mngoura and A.G. El-Shemi, 1998. Schistosomiasis induced chang in glutathione levels and glutathione reductase/glutathione-s-transferase activities in human. Liver. Helminthol., 72: 71-71.
Direct Link - Stich, A.H., S. Biays, P. Odermatt, C. Men and C. Saem et al., 1999. Foci of Schistosomiasis mekongi, Northern Cambodia: II. Distribution of infection and morbidity. Trop. Med. Int. Health, 4: 674-685.
PubMed - Tanizawa, H., Y. Ohkawa, Y. Takino, A. Ueno, T. Kageyama and S. Hara, 1992. Studies on natural antioxidants in Citrus species. I. Determination of antioxidant activity of citrus fruits. Chem. Pham. Bull., 40: 1940-1942.
PubMedDirect Link - Tamura, S., N. Fukamiya, M. Okano, J. Koyoma and K. Koike et al., 2003. Three new quassinoids, ailantinol, E, F and G from Ailanthus altissima. Chem. Pharm. Bull., 51: 385-389.
Direct Link - Urbani, C., A. Toure, A.O. Hamed, M. Albonico and I. Kane et al., 1997. Intestinal parasitic infections and schistosomiasis in the valley of the Senegal river in the Islamic Republic of Mauritania. Med. Trop., 57: 157-160.
PubMed - Van Waarde, A., G. van den Thillart, C. Erkelens, A. Addink and J. Lugtenburg, 1990. Functional coupling of glycolysis and phosphocreatine utilization in anoxic fish muscle. J. Biol. Chem., 265: 914-923.
Direct Link - Wright, C.W. and J.D. Phillipson, 1990. Natural products and the development of selective antiprotozoal drugs. Phytother. Res., 4: 127-139.
CrossRefDirect Link - Xiao, S.H., M. Tanner, E.K. N′Goran, J. Utzinger and J. Chollet et al., 2002. Recent investigations of artemether, a novel agent for the prevention of Schistosomiasis japonica, mansoni and haematobia. Acta Trop., 82: 175-181.
PubMed - Zhang, S., Z. Liu, G. Li, J. Zhong and Y. Cheng, 1990. Snail distribution and susceptible zones of schistosomiasis in endemic areas around Poyang Lake. Zhongguo Ji Sheng Chong Xue Yu Ji Sheng Chong Bing Za Zhi, 8: 8-12.
PubMed - Zhen, J., 1993. A brief introduction to nation-wide sampling survey on schistosomiasis. Chin. Med. J., 106: 569-575.
PubMed - Omer-Ali, P., M. Mansour, J.N. Woody, S.R. Smithers and A.J.G. Simpson, 1989. Antibody to carbohydrate and polypeptide epitopes on the surface of Schistosoma mansoni in Egyptian patients with acute and chronic schistosomiasis. Parasitology, 98: 417-424.
PubMed - Engels, D., L. Chitsulo, A. Montresor and L. Savioli, 2002. The global epidemiological situation of schistosomiasis and new approaches to control and research. Acta Tropica, 82: 139-146.
CrossRef








