Research Article
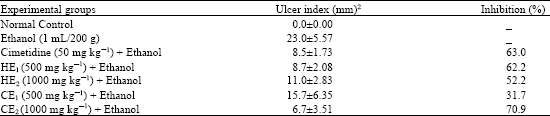
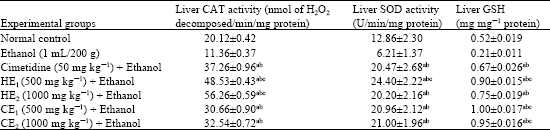
Anti-Ulcerogenic Activity of Two Extracts of Parquetina nigrescens and their Effects on Mucosal Antioxidants Defence System on Ethanol-Induced Ulcer in Rats
Biochemistry Unit, Department of Chemical Sciences, College of Natural and Applied Sciences, Bells University of Technology, Ota, Ogun State, Nigeria
O.T. Kayode
Department of Biochemistry, Faculty of Basic Medical Sciences, College of Medicine, University of Ibadan, Ibadan, Nigeria
A.A. Odetola
Department of Biochemistry, Faculty of Basic Medical Sciences, College of Medicine, University of Ibadan, Ibadan, Nigeria