Research Article
Antimicrobial Activities of Coula edulis
Department of Microbiology, Uyo, Akwa Ibom State, Nigeria
Kola K. Ajibesin
Department of Pharmacognosy and Natural Medicine, University of Uyo, Uyo, Akwa Ibom State, Nigeria
The monotype genus Coula, is a member of the family Olacaceae that comprises 250 species (Mabberley, 1997). Coula edulis Baill (African walnut) is a medium-sized, evergreen tree growing to a height of 25-38 cm with dense crown. The plant flowers between January and May (Alan, 1999). The can grow under plantation condition as a timber plant. Alan (1999) also described the fruit as a nut, ellipsoidal in shape, being about 3-4 cm long with flesh 5-6 mm thick surrounding the kernel.
Coula edulis has pinately compound leaves just like other members of Olacaceae. The leaflets are attached to a central stalk. The fleshy fruit is tasty but covered with a hard thick shell that makes the nut difficult to harvest. C. edulis is commonly known as African walnut or Gabon nut tree due to its edible seeds. It is referred to as ‘Ekom’ (Ibibio and Efik), Udo (Igbo), Ivianlegde (Edo) and Asala (Yoruba) in Nigeria, where the nut obtained from the fruits serves as a good source of nutrients to the local people (Ekop and Eddy, 2005). C. edulis enjoys wide acceptability as food because of its accessibility (i.e., it can be purchased in most places since it is hawked on the road, parks and sold at rural and urban markets). However, it has been listed amongst the endangered plants producing edible fruits and seeds in Southeastern part of Nigeria (Meregini, 2005). The nut has agreeable taste resembling hazelnut or chestnut. Its digestibility property has been linked to its flavonoids (Waterman et al., 1980). Nearly half of the weight of the fruit is oil. Fatty acids containing a large proportion of oleic acid and triacylglycerides were found in the oil (Tchiegang et al., 1998). The fresh seeds were reported to contain fat, starch, flour, potassium and phosphorus (Ekissi et al., 2005). The dried bark of C. edulis was also reported to contain acetylenes known to exhibit anticancer activity (Dembitsky, 2006). Minquartynoic acid has been identified from the plant (Fort et al., 2000). The stomachic bark decoction is used to treat dysentery in Liberia, while the bark powder is used for dressing sores in Equatorial Africa and as decoction, to stimulate appetite and counteract anaemia (Duke, 2001). However, its root has been implicated as toxic through intraperitoneal route to rat (Ajibesin et al., 2002). Other walnuts like English walnut (Juglans regia) and black walnut (Juglans nigra) elicit antimicrobial properties (Alkhawajah, 1997). However, so far there has been no attempt to study the potential of C. edulis antimicrobial activity against a range of pathogenic species isolated from patients. The present study was conducted to investigate the antimicrobial property of ethanolic extracts of C. edulis leaves, stem bark, roots and fruits against five pathogenic species and to determine the classes of chemical constituents of the extracts.
Plant Material
The mature leaves (1 kg), stem bark (1 kg), roots (1 kg) and fruits (1 kg) of C. edulis Baill were collected in June, 2006, at Ikot Idaha in Ikono Local Government Area of Akwa Ibom State. The plant was first identified locally by its local name, Ekom and a taxonomist in the Department of Botany Herbarium, University of Uyo, authenticated it. The plant materials were then separately air-dried at room temperature. Each dried plant material was ground into a fine powder.
Preparation of the Extract
The dried powder of each plant sample (500 g) was macerated in 70% ethanol for 72 h. The extract were filtered and concentrated to dryness in vacuo at 40°C.
Phytochemical Screening
The dry ethanolic extracts of C. edulis leaves (50 g), stem bark (28 g), root (33 g) and fruits (20 g) were screened for their classes of bioactive compounds using standard procedures (Trease and Evans, 2002; Sofowora, 1993; Culei, 1982). The samples were tested qualitatively for the presence of chemical constituents such as tannins, terpenes, saponins, flavonoids, cardiac glycosides, alkaloids and anthraquinones (Harbone, 1984). For flavonoids and tannins, a part each of the dry extracts was dissolved in distilled water, filtered and ferric chloride solution added to the filtrate. Blue-black coloration indicated their presence. To a part of the filtrate were added magnesium strips followed by concentrated HCL (Shinoda’s test). An orange effervescence also showed the presence of flavonoids. A part of each dry extract was shaken with benzene, filtered and 10% NH3 solution added to the filtrate (Borntrager’s test). A rose pink colour in the ammonia layer indicated the presence of antraquinones. For, alkaloids, each part of the dry extract was warmed with 5% HCl, filtered when cool and Dragendorff’s reagent added to the filtrate. Orange precipitate formed showed the presence of alkaloids. To determine cardiac glycosides and terpenes, test such as Salkowski’s and Lieberman’s tests were applied. The dry extract was dissolved in chloroform and concentrated H2SO4 was introduced down the side of the test tube. A reddish-brown ring indicated the presence of steroid, an aglycone part of the cardiac glycoside (Salkowski’s test). Another part of the extract was dissolved in acetic anhydride and cooled well in ice and concentrated H2SO4 was carefully added. A colour change from blue to green indicated the presence of terpenes (Lieberman’s test). Saponins were determined through Frothing test. The extract was vigorously shaken with distilled water in a test tube. Frothing which persisted on warming for about 15 min indicated the presence of saponins.
Microorganisms
The test microorganisms used for the antimicrobial activity screening were Escherichia coli, Staphylococcus aureus, Salmonella typhi, Pseudomonas aeruginosa and Candida albicans. These microorganisms were clinical isolates obtained from the medical diagnostic laboratory, University of Uyo Health Centre and Microbiology Laboratory of St. Luke General Hospital, Uyo, Akwa Ibom State. They were stored at 4°C before use.
Antimicrobial Susceptibility Test
Antimicrobial properties of the plant extracts were tested by the agar diffusion technique using spreading method of Cruickshank et al. (1980). A loop full of the broth culture of each microorganism was uniformly spread over the surface of a sterile nutrient agar plate with a bent sterile glass rod. Equally spaced holes (5 mm in diameter each) were punched on the surface of the inoculated agar plate, using sterile cork borer. The holes were completely fixed with prepared extracts. Four holes on the nutrient agar plates were filled with four different concentrations of ethanolic extracts, the fifth and the sixth holes were filled with methanol-water 1:1 and Chloramphenicol, a standard drug, respectively which served as controls. However, the fungal plates bore Nystatin as standard drug in the sixth hole. The diameter of the zones of inhibition was measured. Clear zones of inhibition indicated susceptibility of the organisms while absence of such zones indicated resistance.
The Minimum Inhibitory Concentration (MIC) of the extracts was determined by standard two-fold dilution technique (Washington, 1985). Various amounts of each extract solution were introduced into sets of test tubes containing the culture media. Fifty microliter of the standard test bacterial and fungal broth cultures were added into each of the test tubes. The set of test tubes containing a mixture of bacteria and the extracts were incubated at 37°C for 24 h, while those containing the fungus were incubated at 25°C for 7 days. A positive control tube containing only the growth medium and each of the organisms were also set up. The minimum inhibitory concentration was regarded as the lowest concentration of the extracts that did not permit any visible growth when compared with that of the control tubes (Table 1).
| Table 1: | Minimum inhibitory concentration of the ethanol extracts of the plant parts of Coula edulis |
 | |
| -: No inhibition | |
The ethanolic extracts of the leaves, stem bark, roots and fruits of Coula edulis Baill. showed different classes of bioactive constituents such as alkaloids, flavonoids, tannins, saponins, terpenes and cardiac glycosides (Table 2). The extracts of the various parts of C. edulis showed variable degrees of antimicrobial activities against one or more of the tested organisms (Table 3). Ethanolic extract of the fruits showed the highest inhibitory activity against S. aureus at a concentration of 100 mg mL-1 with a zone of inhibition of 36 mm, while the same extract did not show any effect against E. coli, S. typhi, Ps. aeruginosa and C. albicans, since zone of inhibition of 10 mm or greater was considered as good antimicrobial activity. Ethanolic extract of stem bark of the plant provided the highest activity against Ps. aeruginosa and S. aureus at concentration of 100 mg mL-1 with a zone of inhibition of 40 and 34 mm, respectively. The stem bark and the leaves exhibited reasonable activity against C. albicans at all concentration levels.
| Table 2: | Phytochemical composition of plant parts Coula edulis studied |
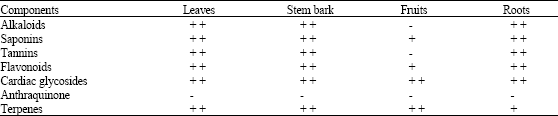 | |
| + +: Present, +: Trace, -: Absent | |
| Table 3: | Antimicrobial effects of ethanol extracts of the plant parts of Coula edulis |
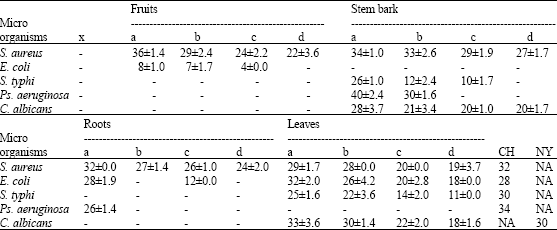 | |
| x: Distilled water, a: 100 mg mL-1, b: 50 mg mL-1, 25 mg mL-1, d: 12.5 mg mL-1, -: No inhibition, CH: Chloramphenicol (4 μg mL-1), NY: Nystatin (4 μg mL-1), NA: Not Applicable | |
The stem bark extract did not exhibit any antimicrobial effect against E. coli. The root extract provided the highest activity against S. aureus and E. coli at concentration of 100 mg mL-1 with inhibition zones of 32 and 28 mm, respectively. However, it did not show any antimicrobial activity against C. albicans and S. typhi. Amongst the test isolates, Ps. aeruginosa did not show any sensitivity to the leaf extract, but the extract provided the highest inhibition against C. albicans at a concentration of 100 mg mL-1 with an inhibition zone of 33 mm.
The result of this study showed that Coula edulis Baill exhibited antimicrobial effects against the microorganisms tested. The result obtained from this study also revealed that the plant contained bioactive compounds known to contribute to the antimicrobial potential of plants (alkaloids, saponins, tannins and flavonoids). The bioactive agents responsible for the inhibitory effects of the extracts of the leaves, stem bark, roots and fruits of C. edulis are known to be pharmacologically active principles mentioned above (Sofowora, 1993). Some of the pharmacologically active ingredients encountered in this study have also been reported as antimicrobial agents (Sofowora, 1993). For instance, berberine, an alkaloid isolated from Berberis vulgaris and aesculetin, a coumarin identified in Fraxinus rhynchophylla have been reported to cure dysentery (Fabricant and Farnsworth, 2001). Tannins isolated from Vaccinium vitis-idaea and saponins from Hedera taurica and Allium minutifolium have also been reported to exhibit antimicrobial activities (Ho et al., 2001; Tyshkevich and Krivorutchenko, 2003; Barile et al., 2007). The result of the antimicrobial assay has revealed that all the plant extracts studied produced zones of inhibition of growth of the various pathogenic organisms tested. The stem bark and leaf extracts gave the broadest spectrum of activities. Furthermore, these extracts alone showed antifungal activity against the only the only fungus tested. However, the stem bark extract showed the highest activity against Ps. aeruginosa, while the fruit extract gave the least against E. coli. Following similar trend, the fruit exhibited the least activity amongst all the parts tested. This may be due to the fact that the bioactive constituents such as alkaloids, tannins and flavonoids responsible for the antimicrobial activity of the fruit either occurred in trace or were lacking. Similar pattern of activities has also been reported in Triumfetta cordifolia, where the seeds of the plant showed the least antimicrobial effect amongst other parts (Ekpo et al., 2007). Thus, the bioactive agents of C. edulis detected through phytochemical screening may be responsible for the inhibitory effects of the various parts of the plant. This result also suggests that the concentration level of the chemical constituents determines the degree of potency of the extracts as antimicrobial agents.
The test plant although common, especially in Nigeria, is not known for its antimicrobial properties and as such is not used in the treatment of such ailments. From this study, C. edulis is recommended for use in the treatment of infections caused by these microorganisms. However, this study does not justify the use of the fruit in the treatment of such infectious diseases.
From the result obtained, ethanolic extracts of Coula edulis leaves, stem bark and roots showed good antimicrobial activities against a range of pathogenic microorganisms from patients. The presence of saponins, tannins, flavonoids and alkaloids in these extracts may explain their antimicrobial activity. This is the first report of antimicrobial activity of the ethanolic extracts of the leaves, stem bark, roots and fruits of C. edulis. Further research should be carried out on the fractionation of the extracts, leading to the isolation of pure compounds and elucidation of their structures in order to enhance and better evaluate their antimicrobial activity.