Research Article
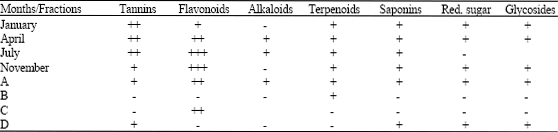
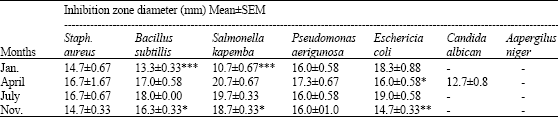
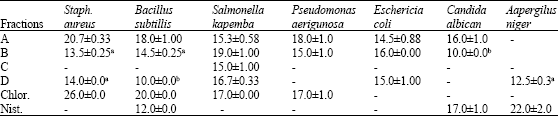
A Study of the Seasonal Variation in the Antimicrobial Constituents of the Leaves of Loranthus micranthus Sourced from Percia americana
Department of Pharmaceutical and Medicinal Chemistry, Faculty of Pharmaceutical Sciences, University of Nigeria, Nsukka, Enugu State, Nigeria
C.A. Dieke
Department of Pharmaceutical and Medicinal Chemistry, Faculty of Pharmaceutical Sciences, University of Nigeria, Nsukka, Enugu State, Nigeria
F.B.C. Okoye
Department of Pharmaceutical and Medicinal Chemistry, Faculty of Pharmaceutical Sciences, University of Nigeria, Nsukka, Enugu State, Nigeria