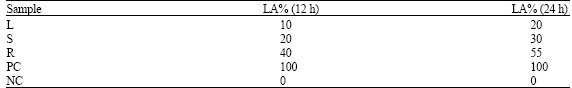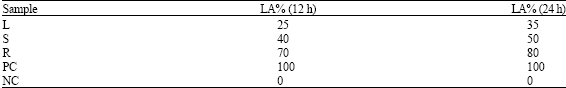Research Article
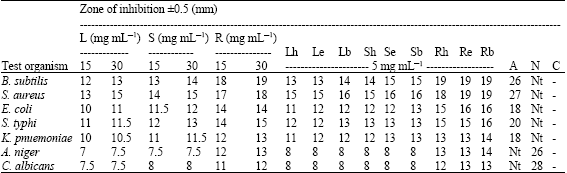
In vitro Biological Activities of Carica papaya
Department of Pharmaceutical and Medicinal Chemistry, University of Uyo, Uyo, Akwa Ibom State, Nigeria
Rene Nia
Department of Pharmacognosy and Natural Medicine, University of Uyo, Uyo, Akwa Ibom State, Nigeria
Kalu Ndukwe
Department of Pharmaceutics and Pharmaceutical Technology, Faculty of Pharmacy, University of Uyo, Uyo, Akwa Ibom State, Nigeria
Emmanuel Attih
Department of Pharmaceutical and Medicinal Chemistry, University of Uyo, Uyo, Akwa Ibom State, Nigeria