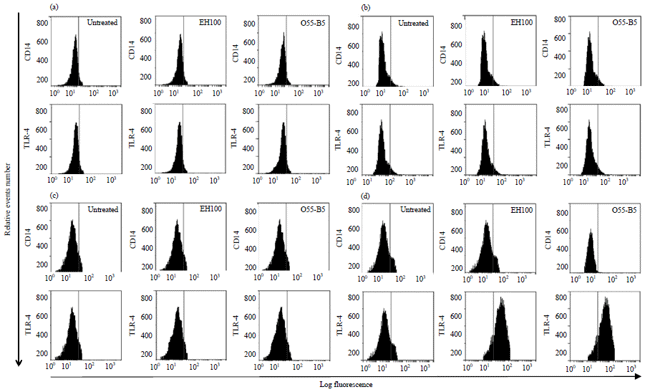
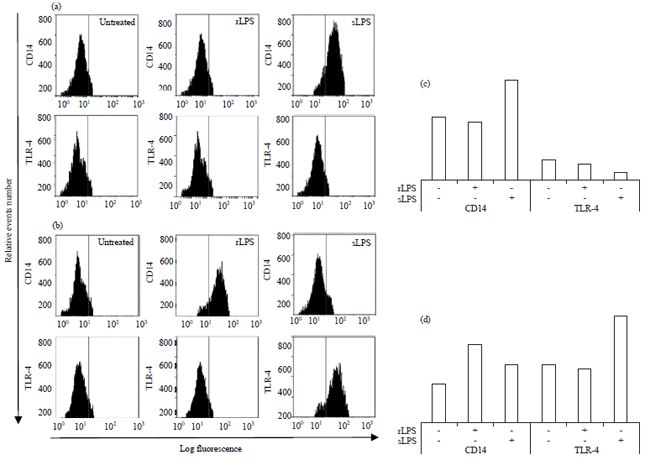
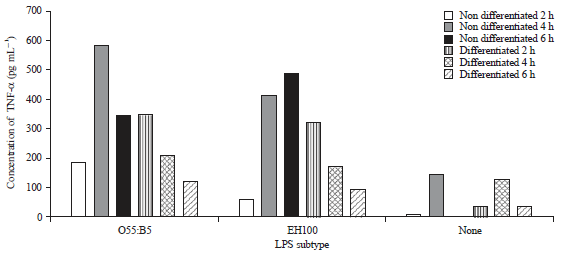
Research Article
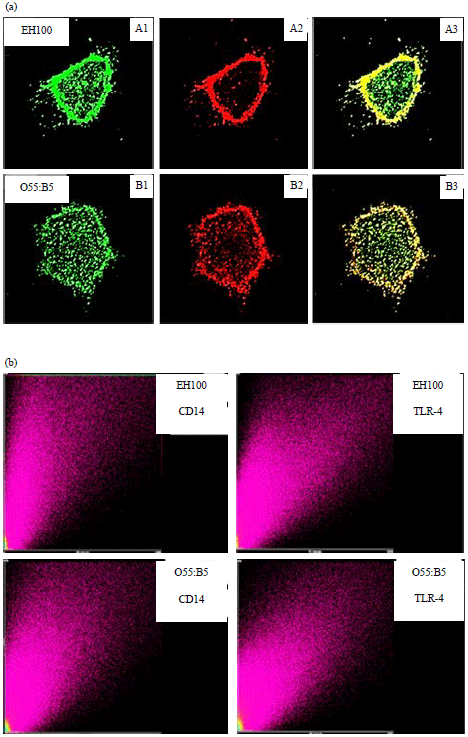
LPS Subtypes Activate Inflammatory Signaling Through CD-14 and TLR-4 in Human Monocytic Cells
Department of Pathology, College of Medicine, Qassim University, P.O. Box 6655, 51452 Buraidah, Saudi Arabia
LiveDNA: 966.23477
Zafar Rasheed
Department of Medical Biochemistry, College of Medicine, Qassim University, Buraidah, Saudi Arabia
Abdullah S.M. Aljohani
Department of Veterinary Medicine, College of Agriculture and Veterinary Medicine, Qassim University, Buraidah, Saudi Arabia
Mohsen Almakrami
School of Biological sciences, University of Essex, Colchester, United Kingdom
Hussain Al Ssadh
School of Biological sciences, University of Essex, Colchester, United Kingdom
LiveDNA: 966.29413
Nelson Fernandez
School of Biological sciences, University of Essex, Colchester, United Kingdom