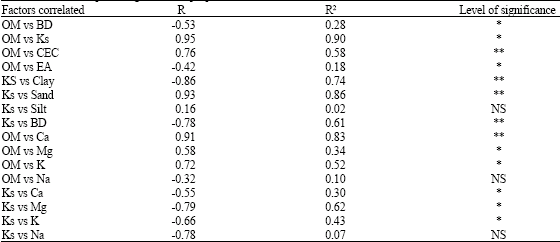
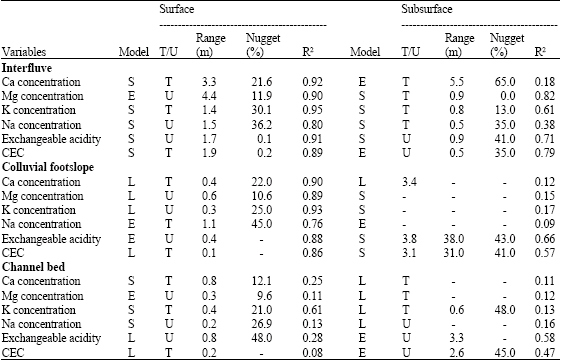
Research Article
Spatial Changes in the Distribution of Exchangeable Cations in Soils of a Forested Hilly Landscape
Department of Soil Science and Technology, Federal University of Technology, P.M.B. I 526, Owerri, Nigeria
F.O.R. Akamigbo
Department of Soil Science, University of Nigeria, Nsukka, Nigeria