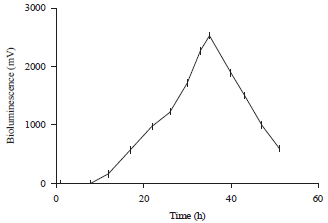
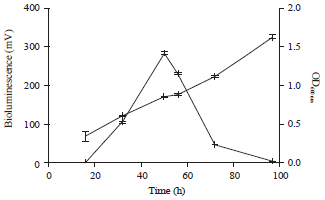
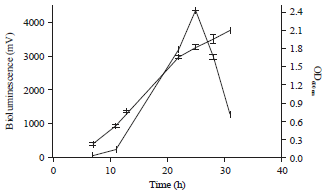
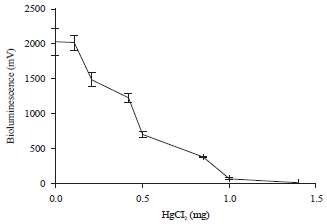
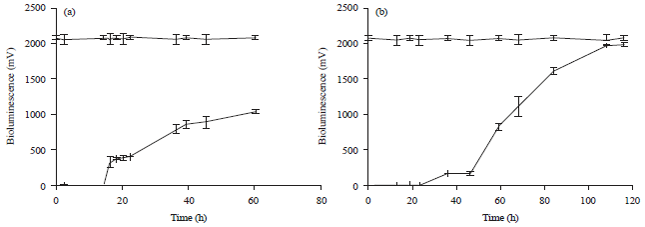
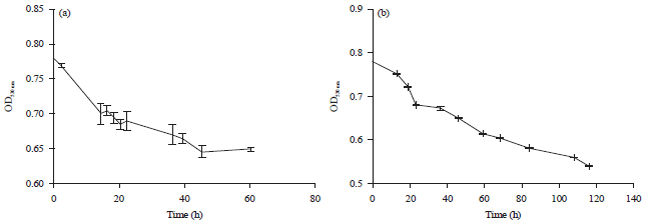
Research Article
Detoxification Assessment of Inorganic Mercury by Bioluminescence of Vibrio fischeri
Laboratory of Microbiology, Pharmacology, Biotechnology and Environment, Faculty of Sciences, A�n Chock, University Hassan II, Km 8 route d`ElJadida, B.P. 5366 Maarif, Casablanca, Morocco
LiveDNA: 212.20070
Ali Daher
Polz Laboratory for Microbial Ecology and Evolution, Massachusetts Institute of Technology, 15 Vassar Street, Bldg. 48-417 Cambridge, Massachusetts, USA
Khalid Belghmi
Laboratory of Microbiology, Pharmacology, Biotechnology and Environment, Faculty of Sciences, A�n Chock, University Hassan II, Km 8 route d`ElJadida, B.P. 5366 Maarif, Casablanca, Morocco
Djolo Yigerta Dah Dossounon
Laboratory of Microbiology, Pharmacology, Biotechnology and Environment, Faculty of Sciences, A�n Chock, University Hassan II, Km 8 route d`ElJadida, B.P. 5366 Maarif, Casablanca, Morocco
Mohamed Blaghen
Laboratory of Microbiology, Pharmacology, Biotechnology and Environment, Faculty of Sciences, A�n Chock, University Hassan II, Km 8 route d`ElJadida, B.P. 5366 Maarif, Casablanca, Morocco