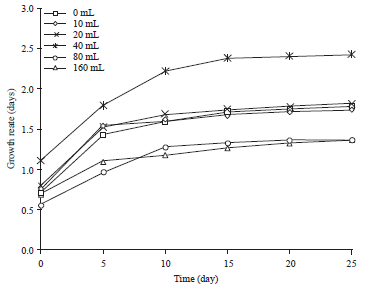
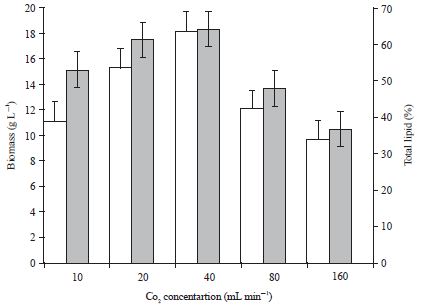
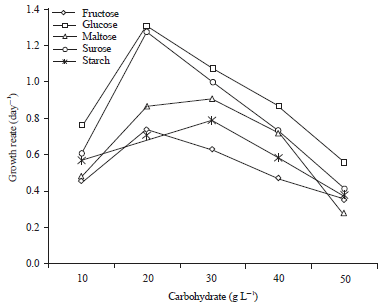
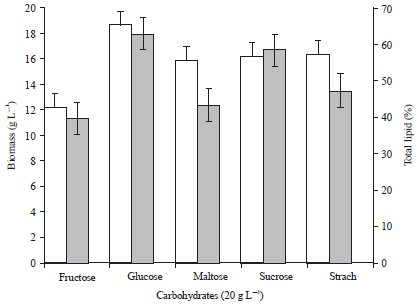
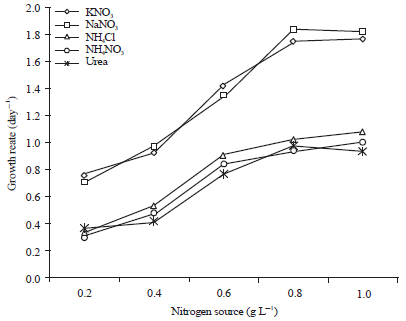
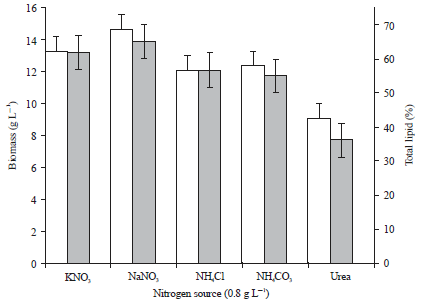
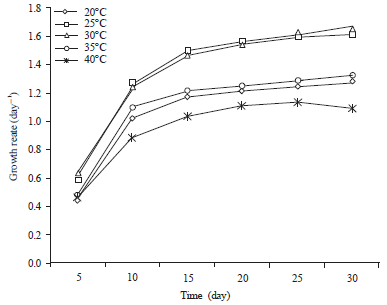
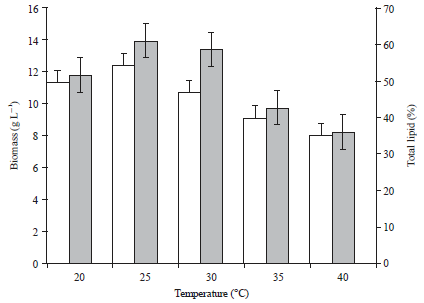
Research Article
Cultural Conditions and Nutrient Composition as Effective Inducers for Biomass and Lipid Production in Fresh Water Microalgae
Department of Biotechnology, Indian Academy Degree College, Centre for Research and Post Graduate Studies, Bengaluru, 560043, Karnataka, India