ABSTRACT
This study investigated the effect of sublethal concentration of malathion on the lipid peroxidation and various enzymes in mice liver, brain and serum. Mature mice were exposed to different doses of malathion and sublethal dose was calculated (80.6 mg kg-1 b.wt. day-1). Two groups of mice were made, first group is control and second experimental. Experimental group was again divided into five subgroups which were exposed to malathion for different time period (2, 4, 8, 15 and 30 days). On dissections, various biochemical estimations such as Aminotransferase (ALT), Aspartate aminotransferase (AST) Malondialdehyde (MDA) and Glutathione-S-transferase (GST) were performed for each exposure period in triplicate. It was found that malathion caused the oxidative stress in different tissues of mice. The maximum period of exposure was up to 30 days. The degree of lipid peroxidation increased in ascending order up to 30 days of exposure period. These findings indicates that the continuous and prolonged exposure to sublethal dose of malathion resulted in induction of lipid peroxidation in mice and alters the activity of enzymes like alanine ALT, AST and GST.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/rjet.2012.142.150
URL: https://scialert.net/abstract/?doi=rjet.2012.142.150
INTRODUCTION
Malathion is an organophosphorus insecticide (Maugh, 2010). Malathion exhibits low toxicity; but absorption or ingestion into the human body results in its metabolism to malaoxon which is more toxic (Edward, 2006). As malathion is widely used in agriculture and public health programs, it has caused significant environmental pollution and health hazards including severe acute and chronic human poisoning (Yarsan and Cakir, 2006). Pesticides and environmental chemicals produce free radicals which may cause toxicity (Abdollahi et al., 2004a). However, the insinuation of pesticide residues for human health should be broadly documented. Pesticides stimulate peroxidation of cellular membranes by different mechanisms; direct initiation by free radicals produced by metabolism of the chemicals, indirect initiation by the production of reactive forms of oxygen during their metabolism, inhibition of enzymatic systems of defence involved in the control of reactive oxidising entities and destruction of natural antioxidants (Dikshith, 1991; Comporti, 1993; Datta et al., 1994; Gupta et al., 1990; Parashar and Singh, 1987; Yarsan, 1998).
Various enzymes such as Aminotransferase (ALT), Aspartate aminotransferase (AST) and Glutathione-S-transferase (GST) are good indicators of oxidative stress. Malondialdehyde (MDA) is also a good biomarker for the evaluation of oxidative stress. Present study dealt with the effects of malathion at subacute and subchronic periods by estimating levels of AST, GST, ALT and MDA in different tissues like liver, brain and serum.
MATERIALS AND METHODS
Study design: Only the healthy pairs of mice were housed in the separate cages. The temperature of house was maintained in the range of 20-25°C. The animals were fed on commercially available pellet diet. Mature and healthy mice of either sex weighing between 25-30 g were used in this study. Mice were divided into two groups. Animals in each group were maintained on specific diet. The animals of group I and II were fed a stock diet used as a control. Animals from group II were given malathion orally (80.6 mg kg-1 b.wt. day-1) in a suspension made in distilled water while control group was given equal amount of water. Mice were selected for sublethal group exposed to only control diet and sacrificed at the end of the experimental period of thirty days (20 mice).
Group II- mice were exposed to sublethal concentration of malathion i.e., 1/3 of Lc 50/96 h.
Group II was further divided into five subgroups, each of 4 mice as under; depending on the malathion exposure period:
| • | SGI:2 days |
| • | SGII:4 days |
| • | SGIII:8 days |
| • | SGIV:15 days |
| • | SGV:30 days |
After the start of experiment, each subgroup at respective treatment period was sacrificed and was used for quantitative estimation of AST and ALT in liver and GST and MDA in liver, brain and serum in mice. Before this, lethal toxicity tests were carried out for four different concentrations of the Malathion.
Experimental design
Enzyme extraction: Animal was etherized. Pesticide treated mice were killed by cervical dislocation and were dissected. Blood was taken directly from the heart. Blood sample was collected in tube, allowed to clot and the serum was removed by centrifugation at 2000 xg for 10 min. All serum samples were sterile, haemolysis free. Sample was stored at -4°C (Meyer and Madias, 1994).
Liver and brain were taken out, washed with cold 0.9% NaCl and homogenized in 5 mM phosphate buffer. Homogenates was centrifuged at 10,000 xg for 20 min at 4°C. Then supernatant was collected and stored at -80°C. Supernatant was used for the determination of enzyme activity (Yusuf et al., 2009).
Biochemical estimation:
| • | MDA assay: MDA content in liver, serum and brain homogenate was determined spectrophotometrically by the presence of thiobarbituric acid reactive substance. (Ohkawa et al., 1979) |
| • | Glutathione (GSH) assay: GSH content in liver, serum, brain homogenate was determined spectrophotometrically (Beutler et al., 1963) |
| • | AST assay: AST content of the liver homogenate was determined spectrophotometrically (Bergmeyer and Bernt, 1974) |
| • | ALT assay: ALT content of liver homogenate was determined spectrophotometrically (Bergmeyer and Bernt, 1974) |
| • | Enzymatic estimation: Protein estimation was performed by Lowery’s method (Lowry et al., 1951) |
Statistical analysis: Values are expressed in Mean±Standard Error (SE) with p-value<0.05. Data was analyzed using SPSS software.
RESULTS
In the present study, estimation of AST and ALT in liver and GST and MDA in liver, brain and serum of mice exposed to sublethal concentration were determined. Following results were obtained.
Liver aspartate aminotransferase (AST) assay: The result showed that sublethal dose of Malathion increases the level of aspartate aminotransferase in mice liver from day two to day thirty. Increase in level of aspartate aminotransferase was exposure period dependent. It was found that the increase in aspartate aminotransferase level in liver is significant at 8th day and highly significant at 15th and 30th day of exposure period. The most significant value of aspartate aminotransferase in liver of mice was 0.128±0.009 mg mL-1 which was observed on 30th day of exposure period On 2nd and 4th day the increase in level of Aspartate aminotransferase was not significant. The effect of malathion on Aspartate aminotransferase in liver is shown in Table 1.
Liver alanine aminotransferase (ALT) assay: It was observed that the sublethal dose of malathion increases the level of alanine aminotransferase in mice liver from day two to day thirty. Increase in level of alanine aminotransferase was also exposure period dependent. It was found that the increase in alanine aminotransferase level in liver is significant at 8th day and highly significant at 15th and 30th day of exposure period. The most significant value of alanine aminotransferase in liver of mice was 0.128±0.009 mg mL-1 which was observed on 30th day of exposure period. On 2nd and 4th day of exposure period the increase in Alanine aminotransferase level is not significant. The effect of malathion on alanine aminotransferase in liver is shown in Table 2.
| Table 1: | Effect of malathion on aspartate aminotransferase in liver of mice |
 | |
| Values are Mean±SE (n = 4), *, **Significant difference between treatment and control at p<0.05 and p<0.01, respectively | |
| Table 2: | Effect of malathion on alanine aminotransferase in liver of mice |
 | |
| Values are Mean±SE (n = 4), *,**Significant difference between treatment and control at p<0.05 and p<0.01, respectively | |
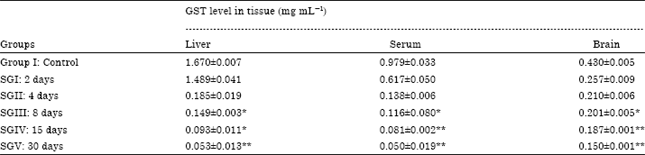
| Table 3: | Effect of malathion on glutathione-S-transferase in different tissues of mice |
 | |
| Values are Mean±SE (n = 4), *,**Significant difference between treatment and control at p<0.05 and p<0.01, respectively | |
| Table 4: | Effect of malathion on Malondialdehyde in different tissues of mice |
 | |
| Values are Mean±SE (n = 4), *,**Significant difference between treatment and control at p<0.05 and p<0.01, respectively | |
Glutathione-S-transferase (GST) assay: It was observed that sublethal dose of malathion decreases the level of glutathione-S-transferase in mice from day two to day thirty. Decrease in the level of glutathione-S-transferase was exposure period dependent. It was found that the decrease in glutathione-S-transferase level in all the tissues is significant at 15th and 30th day of exposure period. The most significant values of glutathione-S-transferase in liver was 0.053±0.013 mg mL-1, in serum was 0.050±0.019 mg mL-1 and in brain it was 0.150±0.001 mg mL-1, all these values were observed on 30th day of exposure period. On 2nd and 4th day of exposure period the change in the level of glutathione-S-transferase is not significant. The effect of malathion on glutathione-S-transferase in different tissues are shown in Table 3.
Malondialdehyde (MDA) assay: The results indicated that the sublethal dose of malathion increases the level of malondialdehyde in mice from day two to day thirty. Increase in level of Malondialdehyde was also exposure period dependent. It was found that the increase in Malondialdehyde level in serum is significant at 4th, 8th day, 15th and 30th day of exposure period. The most significant values of malondialdehyde in liver was 2.021±0.050 mg mL-1, in serum was 0.256±0.050 mg mL-1 and in brain it was 2.016±0.005 mg mL-1, all these values were observed on 30th day of exposure period. On 2nd day of exposure period the increase in malondialdehyde level in brain was not significant. The effect of malathion on malondialdehyde in different tissues of mice is shown in Table 4.
DISCUSSION
Effects of insecticides on lipid peroxidation have been evaluated in several studies. Malathion is the most widely used organophosphate insecticide all over the world. Malathion has many structural similarities with naturally occurring compounds and their primary target of action in insects is the nervous system; it also inhibit the release of the acetylcholinesterase at the synaptic junction (Cabello et al., 2001). Yarsan and Cakir (2006) studied the subacute and subchronic effect of dichlorvos on lipid peroxidation in mice. In this study subacute and subchronic periods were compared with each other and it was found that Superoxide Dismutase (SOD) and Catalase (CAT) activities increased at subchronic period (Yarsan and Cakir, 2006). Wankhade et al. (2008) observed that Malathion inhibited Liver AChE activity in Mice.
Ranjbar et al. (2002) studied the status of oxidative stress and AChE activity in blood samples obtained from 45 OP formulatory pesticide workers with a minimum history of 1 year in the age range of 23-55. The results showed that there was marked inhibition of AChE activity and increased TBA reactive substances indicating lipid peroxidation. El-Gohary et al. (1999), studied the effect of deltamethrin to testicular apoptosis in rats and the protective effect of nitric oxide (NO) synthase inhibitor was investigated.
Effect of subchronic exposure to malathion in the production of oxidative stress was evaluated in male Wistar rats by Akhgari et al. (2003). Administration of malathion (100, 316, 1000 and 1500 ppm) for 4 weeks increased CAT, SOD activities, as well as MDA concentration in red blood cells and liver. Azmi et al. (1999) studies the effects of tetranortriterpenoids (Neem product SDS) and deltamethrin (pyrethroid) on phosphomonoesterase activity in Cyprinus (common carp) and reported enzyme inhibition under the effect of these pesticide. The result demonstrated that the magnitude of AChE inhibition in peripheral tissues does not accurately reflect the central inhibitory effects of malathion on AChE activity in specific brain regions (Banasik et al., 2003). Burgees et al. (1999) observed that a organophosphate insecticide reduced cholinesterase activity in birds. Shadnia et al. (2005) studied, genotoxicity and oxidative stress in workers who formulate OP-pesticide and observed that chronic exposure to OP pesticides was associated with increased activities of CAT, SOD and GSP-Qx in erythrocytes. Study of effects of malathion to oxidative stress and AChE activity in saliva and plasma in rats following subchronic exposure was studied by Abdollahi et al. (2004a) and inferred that malathion at doses of 100, 500 and 1500 ppm increased plasma thiobarbituric acid reactive substances by 61, 69 and 63%, respectively.
The effect of exposure to chlorfenvinphos on lipid metabolism and apoptotic and necrotic cells death in the brain of rats was studied by Roszczenko et al. (2012) and concluded that low repeated exposure to OP pesticides may influence the nervous system through disrupting the lipid profile of the nervous tissue and decreasing the number of the nervous cells.
Effects of Aldicarb and Malathion on Lipid Peroxidation were investigated by Yarsan et al. (1999). High doses of aldicarb and malathion stimulated lipid peroxidation on subacute, subchronic and chronic periods. Therefore, especially MDA levels in plasma and GSH-Px and Cu-Zn SOD activities in erythrocytes were significantly changed. These findings show that pesticide exposure can produce hazardous effects such as stimulation of lipid peroxidation in a mammalian test species and may be of concern to humans (Yarsan et al., 1999). Seth et al. (2000) studied the effects of propoxur on lipid peroxidation in which they found that propoxur increased MDA levels and altered the Glutathione (GSH) levels.
Oxidative stress and lipid peroxidation were estimated by measuring the MDA concentration, the end product of lipid peroxidation and assessing total antioxidant power (Abdollahi et al., 2004b; Ranjbar et al., 2002).
In the present study, MDA levels in tissues like liver, blood and brain were found to increase significantly at high exposure period (30 days). These findings were compatible with other study results. In vitro study of Dowla et al. (1996), the effects of acephate, cadmium, methamidophos, maleic hydrazide and nicotine on the erythrocyte enzyme activities such as delta-aminolevulinic acid dehydratase, SOD and Plasma cholinesterase were investigated. They observed that methamidophos and other chemicals inhibited the plasma SOD activities. Pedrajas et al. (1995) studied the effects of xenobiotics, paraquat, CuCl2, dieldrin and malathion on oxidative process. In that study, malathion enhanced significantly the microsomal soluble Thiobarbituric Acid Reactive Substances (TBARS) while microsomal glutathione transferase activity decreased. Result of the present study is compatible with this study. Altuntas et al. (2002) studied the effects of methidathion on lipid peroxidation and antioxidant enzymes and ameliorating effects of a combination of vitamin E and C against the toxicity. In that study results showed that treating rats with methidathion increased the lipid peroxidation and decreased antioxidant enzymes in erythrocytes. Also lipid peroxidation level, CAT, SOD and GSH-Px activities in erythrocytes were evaluated as biomarkers of oxidative stress. Ruckmani et al. (2011) observed that sub chronic malathion exposure causes progressive hyperglycemia in Wistar rat.
Daily exposure to concentrations of 50 mg kg-1 carbaryl and/or malathion for 21 days produced a 20% decrease in hepatic GSH content. Histopathology revealed no necrosis after three weeks of treatment with these pesticides individually or in combination (Abdel-Rahman et al., 1985). The role of glutathione (GSH) in the detoxification of malathion and in the potentiating action of impurities on the toxicity of malathion was investigated in freshly isolated rat hepatocytes. Malathion and four of its major impurities at concentrations of 0.01 to 1 mm caused a marked depletion of the hepatocellular GSH content within 60 min (Malik and Summer, 1982). Thus, present study is compatible with the study of Malik and Summer (1982).
Oral administration of malathion (MTH) in sublethal (100 mg kg-1) or minimal lethal (125 mg kg-1) doses in buffalo calves produced toxicity with an onset within 15-20 min and peak effects including severe tremors and convulsions within 40-60 min (Gupta, 1984). O,O,S-Trimethyl phosphorothioate (OOS-TMP) is an impurity present in widely used organophosphorus insecticides such as malathion. Oral treatment of rats with the compound produces prominent bronchiolar epithelial necrosis. Following the administration of [3H]OOS-TMP to rats, substantial amounts of radiolabeled material were covalently bound to lung with a concomitant depletion of glutathione (GSH) (Imamura and Hasegawa, 1984). Srikanth and Seth (1990) also observed alterations in xenobiotic metabolizing enzymes in brain and liver of rats exposed to endosulfan and malathion.
CONCLUSION
After analysis of Alanine aminotransferase (ALT), Aspartate aminotransferase (AST) in liver and Malondialdehyde (MDA) levels and Glutathione-S-transferase (GST) level in different tissues like blood, liver and brain, it was observed that there is a significant effect of sublethal dose of malathion on these enzymes and MDA level. There is increase in activity of Aspartate aminotransferase and Alanine aminotransferase in mice when mice is exposed to malathion for a long period (30 days),while activity of glutathione-S-transferase was decreased. Sublethal dose of malathion increases the level of Malondialdehyde in mice from day two to day thirty. Thus sublethal dose of malathion caused oxidative stress in mice. Thus it can be concluded that sublethal dose of malathion affects activity of various enzymes like ALT, AST and GST in different tissues like blood, liver and brain and also increases lipid peroxidation.
ACKNOWLEDGMENT
I acknowledge my guide Dr. K.M. Kulkarni for his support and guidance.
REFERENCES
- Abdel-Rahman, M.S., D.W. Lechner and K.M. Klein, 1985. Combination effect of carbaryl and malathion in rats. Arch. Environ. Contam. Toxicol., 14: 459-464.
CrossRef - Abdollahi, M., A. Ranjbar, S. Shadnia, S. Nikfar and A. Rezaie, 2004. Pesticides and oxidative stress: A review. Med. Sci. Monit., 10: RA141-RA147.
PubMedDirect Link - Akhgari, M., M. Abdollahi, A. Kebryaeezadeh, R. Hosseini and O. Sabzevari, 2003. Biochemical evidence for free radicalinduced lipid peroxidation as a mechanism for Subchronic toxicity of malathion in blood and liver of rats. Hum. Exp. Toxicol., 22: 205-211.
CrossRefDirect Link - Altuntas, I., N. Delibas and R. Sutcu, 2002. The effects of organophosphate insecticide methidathion on lipid peroxidation and anti-oxidant enzymes in rat erythrocytes: Role of vitamins E and C. Hum. Exp. Toxicol., 21: 681-685.
CrossRefDirect Link - Banasik, M., T. Stedeford, A.S. Persad, K. Ueda, S. Tanaka, C. Muro-Cacho and R.D. Harbison, 2003. Selective inhibition of acetylcholinesterase in the cerebellum and hippocampus of mice following an acute treatment with malathion. J. Enzym. Inhib. Med. Chem., 8: 551-555.
PubMed - Beutler, E., O. Duron and B.M. Kelly, 1963. Improved method for the determination of blood glutathione. J. Lab. Clin. Med., 61: 882-888.
PubMedDirect Link - Cabello, G., M. Valenzuela, A. Vilaxa, V. Duran, I. Rudolph, N. Hrepic and G. Calaf, 2001. A rat mammary tumor model induced by the organophosphorous pesticides parathion and malathion possibly through acetylcholinesterase inhibition. Environ. Health Perspect, 109: 471-479.
PubMed - Datta, C., J. Gupta and D. Sengupta, 1994. Interaction of organophosphorus insecticides phosphamidon and malathion on lipid profile and acetylcholinesterase activity in human RBC membrane. Indian J. Med. Res., 100: 87-89.
PubMed - Dowla, H.A., M. Panemangalore and M.E. Byers, 1996. Comparative inhibition of enzymes of human erythrocytes and plasma in vitro by agricultural chemicals. Arch. Environ. Contam. Toxicol., 31: 107-114.
PubMed - Gupta J., C. Datta, A. Sarkar and D. Sengupta, 1990. Effect of malathion on antioxidant defence system in human foetus an in vitro study. Indian J. Exp. Biol., 30: 352-354.
PubMed - Gupta, R.C., 1984. Acute malathion toxicosis and related enzymatic alterations in bubalus bubalis: Antidotal treatment with atropine, 2-PAM and diazepam. J. Toxicol. Environ. Health, 14: 291-303.
CrossRef - Imamura, T. and L. Hasegawa, 1984. Role of metabolic activation, covalent binding and glutathione depletion in pulmonary toxicity produced by an impurity of malathion. Toxicol. Applied Pharmacol., 72: 476-483.
PubMed - Malik, J.K. and K.H. Summer, 1982. Toxicity and metabolism of malathion and its impurities in isolated rat hepatocytes: Role of glutathione. Toxicol. Applied Pharmacol., 66: 69-76.
CrossRef - Meyer, K.B. and N.E. Madias, 1994. Cisplatin nephrotoxicity. Mineral Electrolyte Metab., 20: 201-213.
PubMedDirect Link - Lowry, O.H., N.J. Rosebrough, A.L. Farr and R.J. Randall, 1951. Protein measurement with the folin phenol reagent. J. Biol. Chem., 193: 265-275.
CrossRefPubMedDirect Link - Ohkawa, H., N. Ohishi and K. Yagi, 1979. Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal. Biochem., 95: 351-358.
CrossRefPubMedDirect Link - Pedrajas, J.R., J. Peinado and J. Lopez-Barea, 1995. Oxidative stress in fish exposed to model xenobiotics. Oxidatively modified forms of Cu, Zn-superoxide dismutase as potential biomarkers. Chem. Biol. Interact., 98: 267-282.
CrossRef - Ranjbar, A., P. Pasalar and M. Abdollahi, 2002. Induction of oxidative stress and acetylcholinesterase inhibition in organophosphorous pesticide manufacturing workers. Hum. Exp. Toxicol., 21: 179-182.
CrossRefDirect Link - Roszczenko, A., J. Rogalska, J. Moniuszko-Jakoniuk and M.M. Brzoska, 2012. The effect of exposure to chlorfenvinphos on lipid metabolism and apoptotic and necrotic cells death in the brain of rats. Exp. Toxicol. Pathol., (In Press).
CrossRefDirect Link - Ruckmani, A., P.G. Nayar, V.G.R. Konda, N. Madhusudhanan and E. Madhavi et al., 2011. Effects of inhalational exposure of malathion on blood glucose and antioxidants level in wistar albino rats. Res. J. Environ. Toxicol., 5: 309-315.
CrossRefDirect Link - Srikanth, N.S. and P.K. Seth, 1990. Alterations in xenobiotic metabolizing enzymes in brain and liver of rats coexposed to endosulfan and malathion. J. Applied Toxicol., 10: 157-160.
CrossRef - Yarsan, E., M. Tanyuksel, S. Celik and A. Aydin, 1999. Effects of aldicarb and malathion on lipid peroxidation. Bull. Environ. Contam. Toxicol., 63: 575-581.
CrossRef - Yarsan, E. and O. Cakir, 2006. Effects of dichlorvos on lipid peroxidation in mice on subacute and subchronic periods. Pest. Biochem. Physiol., 86: 106-109.
CrossRefDirect Link - Yusuf, A.A., R.C. Abaidoo, E.N.O. Iwuafor, O.O. Olufajo and N. Sanginga, 2009. Rotation effects of grain legumes and fallow on maize yield, microbial biomass and chemical properties of an alfisol in the Nigerian savanna. Agric. Ecosyst. Environ., 129: 325-331.
CrossRef - Abdollahi, M., S. Mostafalou, S. Pournourmohammadi and S. Shadnia, 2004. Oxidative stress andcholinesterase inhibition in saliva and plasma of rats following subchronic exposure to malathion. Comparative Biochem. Physiol. Part C: Toxicol. Pharmacol., 137: 29-34.
CrossRef - El-Gohary, M., W.M. Awara, S. Nassar and S. Hawas, 1999. Deltamethrin-induced testicular apoptosis in rats: The protective effect of nitric oxide synthase inhibitor. Toxicology, 132: 1-8.
CrossRef - Shadnia, S., E. Azizi, R. Hosseini, S. Khoei and S. Fouladdel et al., 2005. Evaluation of oxidative stress and genotoxicity in organophosphorus insecticide formulators. Hum. Exp. Toxicol., 24: 439-445.
CrossRef








