Research Article
Effects of Multiple-source Pollution on Spatial Distribution of Polychaetes in Saudi Arabia
Department of Sciences, College of Teachers, King Saud University, Riyadh, Kingdom of Saudi Arabia
The Arabian Gulf which extends along the eastern coast of Saudi Arabia, is a semi-enclosed basin situated in the subtropical zone (Nasser, 2011). This basin is characterized by low precipitation, low hydrodynamism and high evaporation which causes accumulation of contaminants in sea water and sediment (Alyahya et al., 2011; El-Gendy et al., 2012). In fact, the Gulf coastal waters of Saudi Arabia are subjected to considerable pressure from oil spill (from oil tankers and oil-related industries), sewage and industrial wastes which are responsible for the contamination of the waters with consequent loss in biodiversity.
Benthic communities are particularly sensitive to environmental pollutants (Holland et al., 1987; Khoja, 2000; Ahmed et al., 2007; Amar et al., 2007; Mani et al., 2008; Omoigberale and Ogbeibu, 2010; Shokat et al., 2010). The abundance, diversity, biomass and species composition of benthic invertebrates can be used as indicators of changing environmental conditions (Pearson and Rosenberg, 1978). Among benthos, polychaetes are ideal indicator organisms of marine pollution (Tomassetti and Porrello, 2005; Tabatabaie et al., 2009; Surugiu et al., 2010; Musale and Desai, 2011; El-Gendy et al., 2012) since they constitute well over half of the total number of organisms in and on the bottom and thus give a good indication of benthic conditions.
Relationship between environmental factors and spatial variation of benthic polychaetes has been discussed by Kristensen (1988), Miron and Kristensen (1993), Pardal et al. (1993), Guerra-Garcia and Garcia-Gomez (2001), Moreira et al. (2006), Musale and Desai (2011) and El-Gendy et al. (2012).
The aim of this contribution is (1) to document how can different kinds of pollutants affect the spatial patterns of polychaete assemblages from the Arabian Gulf coast (Eastern Province) of Saudi Arabia and (2) to evaluate the influence of environmental pollution on density, frequency, diversity and evenness of polychaetes of the studied area.
MATERIAL AND METHODS
Study area: The study area is located at the Saudi Arabian Gulf coast between Half Moon Bay (GPS Coordinates: 50.0389E, 26.1653N) and Manifa (GPS Coordinates: 48.95417E, 27.61306N). This area belongs to the Eastern Province which is the largest province in Saudi Arabia. Indeed, it receives a large pollution load from different sources. The major threats to the marine environment of the province are related to land-based activities and oil spill (from oil transportation by tankers and pipelines and exploration). Land-based activities include urbanization and coastal development (for example, dredge and fill operations), industries including power and desalination plants and refineries, recreation and tourism, waste water treatment facilities, power plants, coastal mining and quarrying activities, oil bunkering and habitat modification such as the filling and conversion of wetlands. Five stations have been chosen in the Eastern province on the Arabian Gulf coast of Saudi Arabia (Fig. 1). Sampling has been performed in the Arabian Gulf during August and September 2010.
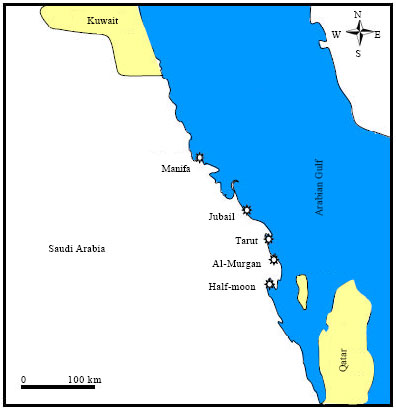 | |
| Fig. 1: | Location map of the studied area showing sampling sites |
| • | Site 1: Half Moon: A beach for swimming during the summer which receives disposal effluents of domestic sewage water as well as waste disposal through boats and ships and is affected by several activities of the King Abdul-Aziz marine port. The stability of Half Moon has been disturbed by the effects of domestic sewage on sediment (Baker and Hosny, 2005). altering the salinity and organic matter values and consequently the faunal composition |
| • | Site 2: Al-Murgan: A remote area, far removed from obvious sources of metal pollution, with no appreciable human activities. (This location was chosen as a reference site to provide information on natural metal levels in non-contaminated sediments) |
| • | Site 3: Tarut Bay: Adjacent to an agricultural drainage area and receives many heavy metals such as Cu, Cd, Zn and Pb (Alyahya et al., 2011) discharged by different industries. The bottom of the bay is characterized by being muddy and fertile in nature. The inadequacy of the command-and-control approach to pollution is reflected in the continuing coastal pollution in the vicinity of industrial complexes, such as Al-Khobar, Tarut Bay including Ras Tanura and Jubail of Saudi Arabia (UNEP, 1999) |
| • | Site 4: Jubail: Consists of the Old Town of Al Jubail which was originally a small fishing village, up to 1975 and the new industrial area. In 1975, Jubail was designated as a site for a new industrial city by the Saudi government and has seen rapid expansion and industrialization since. It is the largest industrial complex of its kind in the world and consists of petrochemical plants, fertilizer plants, steel works, industrial port and huge number support industries (Abdul Azis et al., 2000) |
| • | Site 5: Manifa: Located in the northern part of the Saudi Arabian coast, close to Kuwait shores. This site is effected by oil spill related to the well-known oil field, the Manifa oil field which is situated just offshore of the Eastern Province of Saudi Arabia in the shallow coastal waters of the western Arabian Gulf that generally have depths of less than 5 m (Al-Dakhil and Close, 2008) |
Sampling and analytical procedures: Sediment samples were collected in triplicate (n = 3) by operating van Veen grab (0.04 m2 area) in a depth range of 25-30 m. In order to minimise the loss of organisms during their transportation ashore, the samples were placed in situ in polyethylene bags. Samples were immediately fixed in 10% buffered formalin and rinsed in the laboratory with freshwater. After that, the sediment was sieved through a 0.5 mm mesh screen and animals retained were preserved in 5% buffered formaldehyde. The retained polychaetes were sorted under the stereomicroscope, identified to the species level and counted. Sorted and identified animals were preserved in 80% ethyl alcohol. For additional details of the methodology (Rizzo and Amaral, 2000).
Some physical and chemical variables of the water column were also measured at each sampling site. Dissolved Oxygen (DO) and temperature were measured using a YSI 95 m (Model 95/25 Ft) and salinity was measured using a WTW meter (LF 320) with a Tetra-Con 325 probe (both meters were calibrated following manufacturer instructions). Salinity measurements have been reported according to the Practical Salinity Scale of 1978 (PSS 78) as dimensionless values. A Sensorex combination pH electrode (45°C) with a Rex PH meter (Model pHb-4) was used for all pH measurements (in the field and in the lab) and was calibrated against standardized pH 4 and 7 NIST buffers before use.
The concentration of nitrates, nitrites, ammonia and phosphates was determined spectrophotometrically with a Merck RQFlex Plus device which uses special Reflectoquant® strips.
Statistical analysis: Polychaete structure was calculated in terms of species richness (S), total abundance (N), Evenness (D), frequency (F) and diversity. To measure species diversity, two indices; the Shannon-Wiener diversity (H') (Shannon index) and the Margalef's index (d) were used. Calculations were done using package PAST version 1.42. When indices were congruent, the most significant value has been reported.
Marine pollution is considered an important environmental threat throughout the eastern and western coast of Saudi Arabia (El-Sayed and Niaz, 1999; Al-Farawati, 2010; Al-Farraj et al., 2011; Alyahya et al., 2011; Karami et al., 2011; El-Gendy et al., 2012).
Polychaetes, the dominant benthic fauna in the marine environment, play an important role in bio-monitoring the marine environmental quality being monitors for toxic materials and pollution indicators (Remani et al., 1983; Warwick and Ruswahyuni, 1987; Guerra-Garcia and Garcia-Gomez, 2004; Jayaraj et al., 2007; Dean, 2008; Santi and Tavares, 2009; Shen et al., 2010).
The presence or absence of specific polychaetes in marine sediments provides an excellent indication of the condition or health of the benthic environment (Pocklington and Wells, 1992). On the other hand, polychaetes are among many other benthic marine macrobenthos groups clearly reflected the diversity and abundance patterns of the benthic communities (Olsgard and Somerfield, 2000). In this context, both biotic (species richness, abundance, diversity, etc.) and abiotic (salinity, temperature, oxygen concentration, etc.) variables are responsible for polychaete distribution (Kristensen, 1988; Musale and Desai, 2011; Guerra-Garcia and Garcia-Gomez, 2001, Moreira et al., 2006; El-Gendy et al., 2012).
In the study, bottom water temperatures were generally in the range 16-36°C. Level of dissolved oxygen concentration and pH value were much higher in Al-Murgan (control site) than in other sites (Fig. 2). In addition, the mean salinity level in control site was higher (44 psu) than other sites.
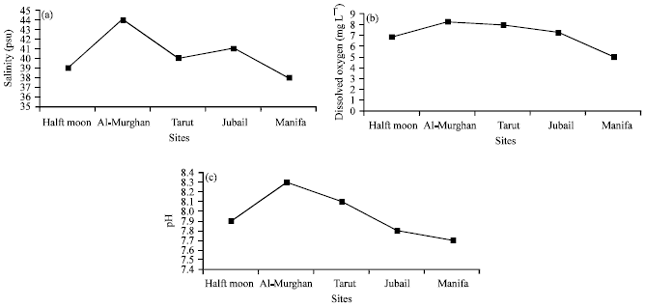 | |
| Fig. 2(a-c): | Mean of environmental variables in each sampling station, (a) Salinity, (b) Dissolved oxygen and (c) pH |
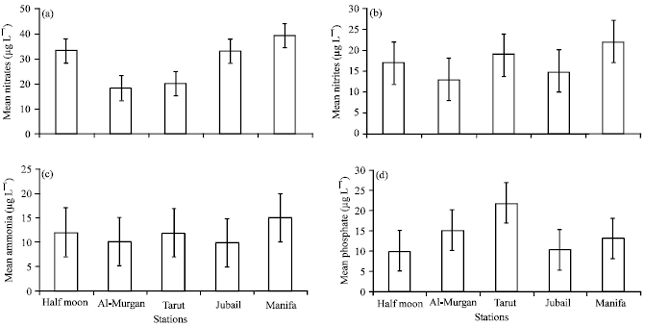 | |
| Fig. 3(a-d): | Mean of nutrient concentrations in different sites, (a) Nitrates, (b) Nitrites, (c) Ammonia and (d) Phosphate |
Generally, species numbers of polychaetes decrease along with the decrease in salinity (Surugiu et al., 2010). The decline in dissolved oxygen concentrations can also promote the formation of reduced compounds, such as hydrogen sulphide (H2S), resulting in higher adverse effects on aquatic animals (Diaz and Rosenberg, 1995; Wetzel, 2001; Breitburg, 2002).
The nutrient concentrations (nitrates, nitrites, ammonia and phosphates) were higher at the polluted stations (Tarut, Half Moon, Jubail and Manifa) than in Al-Murgan (Fig. 3). Differences in nutrient levels between control site and other stations were highly significant (p<0.01).
Eutrophication (the accelerated production of organic matter, particularly algae, in a water body) is usually caused by an increase in the amount of nutrients being discharged to the water body. UNEP (1999) reported that eutrophication is common in some industrialized areas of the Gulf, where dense mats of filamentous green algae are observed and indicate high nutrient levels. Polychaete is adapted to live in waters with extremely low algal concentrations (Riisgard and Ivarsson, 1990).
Increased nutrient loads in polluted sites produce frequent algal blooms which lead to hypoxia and occasional anoxia with subsequent mass mortality of bottom and near bottom organisms (Zaitsev, 1991). Pearson and Rosenberg (1978) have noticed that there is an increase in polychaete diversity with increasing organic nutrients up to an intermediate level and then a decline in diversity with even greater organic inputs.
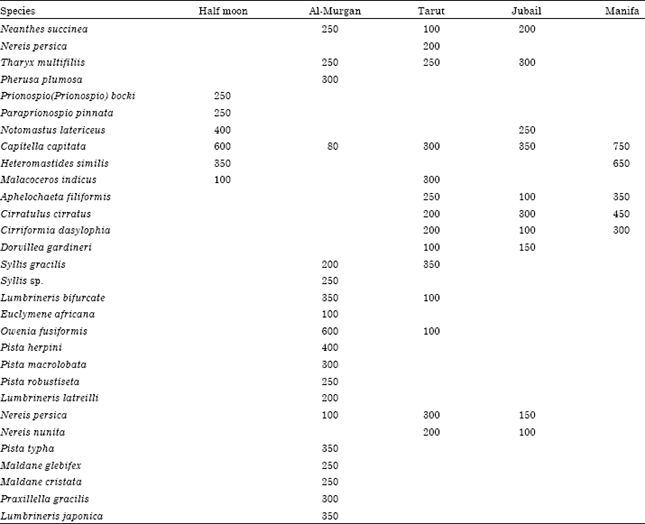
The fauna: A total of 30 polychaete species have been reported during this study (Table 1). The most common families in polluted sites (1, 3, 4 and 5) were Cirratulidae, Capitellidae and Spionidae both in term of abundance and species richness. On the other hand, Lumbrineridae, Terebellidae and Maldanidae are common in control site (#2). The three polychaete species, Capitella capitata, Cirratulus cirratus and Cirriformia dasylophia represent the highest abundance in the study. Amongst, Capitella capitata was the most prevalent species in all stations (Fig. 4).
| Table 1: | Polychaete species identified from the studied locations |
 | |
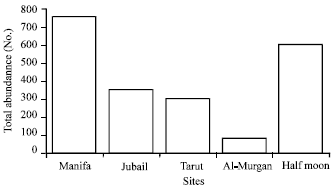 | |
| Fig. 4: | Total abundance of the most common Capitella capitata in different sites |
Community pattern: In the study, species richness and abundance of polychaetes taxa were found to vary among different sites (Fig. 5). These differences could be probably related to the pollution level in each area although other factors as salinity and nutrient cent rations could also be related.
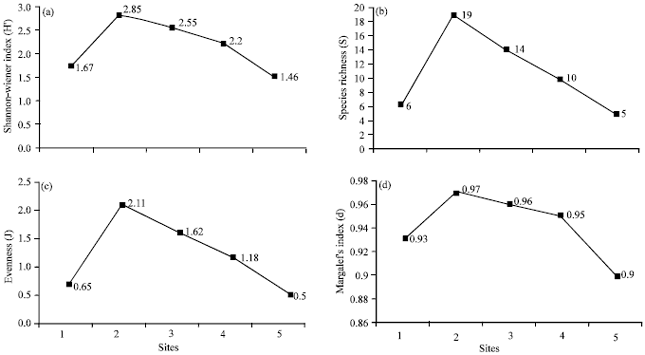 | |
| Fig. 5(a-d): | Variation in univariate measures of the polychaete communities |
It is obvious from the statistical analysis that species richness is significantly higher in Al-Murgan site (n = 19), than in Tarut, Jubail, Half Moon and Manifa, respectively (Fig. 5). Polychaete species richness is usually low in areas that affected by pollution (Kennish, 1997; Guerra-Garcia and Garcia-Gomez, 2001, 2004; Moreira et al., 2006; Musale and Desai, 2011; El-Gendy et al., 2012).
In addition, the lowest population density at the polluted location (1, 3, 4 and 5) is due to the low level of dissolved oxygen, to the decrease of salinity and to the high concentration of toxic compounds resulting from the decomposition of organic matter. Nutrient enrichment significantly affects the composition of marine communities (Larsson et al., 1985; Kotta and Kotta, 1997; Kotta et al., 2000). Domestic (as in Half Moon), industrial sewage (as in Jubail and Tarut) and drainage water from agricultural land (as in Tarut) contain organic matter and nutrients (NH4, NO2, NO3 and PO4). By discharging these wastes into the sea, a local increase in the quantity of the particulate and dissolved organic matter has occurred. This organic matter serves as food for many benthic surface or sub-surface deposit-feeders. If the quantity of domestic waste is large the bottom-dwelling organisms cannot assimilate this increase. Then the bacterial decomposition of unassimilated organic matter increases and hence the concentration of dissolved oxygen diminishes to critical levels. Because oxygen deficiency is the main ecological factor causing severe stress to the macrobenthos, the number of species and the number of individuals will decrease until they disappear altogether (Surugiu et al., 2010).
In Tarut and Jubail, the main source of pollution was heavy metals from agricultural and industrial wastes (UNEP, 1999; Alyahya et al., 2011). Nevertheless, species richness, abundance, diversity and evenness are moderate to relatively high following the control site of Al-Murgan (Fig. 5). Ward and Hutchings (1996) found Capitella at the most contaminated site at a density of 322 worms per m2, indicating that the species is self-sustaining and can tolerant very high concentration of metals. Similarly, sediments with extremely high levels of heavy metals are almost exclusively colonized by polychaetes (Beesley et al., 2000).
Many studies have reported that polychaetes display great variability in their sensitivities to heavy metals both between species and with regards to a particular heavy metal (Athalye and Gokhale, 1991; Reish and Gerlinger, 1997; Sun and Zhou, 2007). Unfortunately, the results of these laboratory studies often cannot be applied to the field situation as these metals are adsorbed onto the sediments segregating them from the benthic organisms (Dean, 2008).
On the other hand, most polychaetes may take up appreciable amounts of metals in their food. They often have capabilities to detoxify these metals and store them in a less toxic form. Metallothionein is considered as a detoxification protein for heavy metals in polychaetes (Won et al., 2008).
Many polychaetes take up high levels of heavy metals associated with their food but these metals are converted to less toxic forms in the gut and are less hazardous than absorption through the body wall (Selck et al., 1998; Berthet et al., 2003). For example, Koechlin and Grasset (1988) found that silver (Ag) which is highly toxic, could be stored in the sabellid Sabella pavonina, in a biologically inactive form allowing for high body concentration of the metal with no apparent ill effects.
In the studied area, Manifa was the most polluted station. It had the lowest values of species richness and diversity. This station is strongly affected by oil spills form nearby oil well (oil transportation by tankers and pipelines, offshore oil exploration and development and recreation) (Fowler, 1993; Abu-Zinada, 2008). The polychaete community in this station was significantly adversely affected by the oil pollutants. Capitella capitata, Heteromastides similis and Cirratulus cirratus are the dominant polychaete species in Manifa. Morgan et al. (1974) stated that if there had been a buildup of hydrocarbons or other pollutants, then one would expect a reduced benthic fauna and the appearance of pollution-tolerant species, such as Capitella capitata, in large numbers at the affected localities. Dense populations Capitella build up rapidly in oil-contaminated sediments (Grassle and Grassle, 1974; Mendez et al., 2001).
Referring to values of species richness, abundance, diversity and evenness, the Half Moon station is the second polluted station in the studied area after Manifa station (Fig. 5). The polychaete community in this station was apparently adversely affected by the increased sewage pollution. Capitella capitata, Notomastus latericeus and Heteromastides similis were the most common polychaete species in Half Moon. Among them, Capitella capitata represents the greatest number of occurrence. The opportunistic polychaete family Capitellidae becomes very abundant close to the sewage outfall and is an indicator for organic pollution (Pocklington and Wells, 1992; Kress et al., 2004; Al-Farraj et al., 2012).
Al-Murgan station (Control site) is characterized by the highest species richness, abundance and diversity (Fig. 5). Owenia fusiformis, Pista herpini and Lumbrineris japonica were the most polychaete species recorded in control site. Owenia fusiformis is not known to have any strong opportunistic properties (Flaten et al., 2007). They occur together with other species (e.g., Syllis gracilis and Maldane cristata) that characterize regions not exposed to environmental stress, e.g., pollution.
| • | The western coast of the Arabian Gulf, Eastern Province of Saudi Arabia, is affected by various sources of pollution (sewage, industrial, agricultural and oil pollutants) that cause significant variations in composition, distribution and abundance of polychaete community |
| • | Al-Murgan, the control station, had the highest number of species, diversity and evenness with the dominant polychaete species Owenia fusiformis, Pista herpini and Lumbrineris japonica |
| • | Manifa had the lowest values of abundance, species richness and diversity with the omnipresent polychaete species Capitella capitata, Heteromastides similis and Cirratulus cirratus |
| • | Species richness, abundance, diversity and evenness were moderate to relatively high in Jubail and Tarut stations that are affected by heavy metal pollution. This may be due to their tolerance to high levels of heavy metals or their capabilities to detoxify these metals and store them in a less toxic form |
| • | The polychaete species Capitella capitata, Notomastus latericeus and Heteromastides similis were dominant at Half Moon station that is influenced by organic (sewage) pollution |
| • | Results suggest the distribution and abundance of polychaetes in the studied area were highly dependent on salinity, nutrient contents and dissolved oxygen |
The author is grateful for the Scientific Committee of King Saud University and the Deanship of Scientific Research, King Saud University, Kingdom of Saudi Arabia for supporting this study. Special thank to the Research Center, College of Teachers, King Saud University for financial assistance.