Research Article
Assessment of Pesticide Residues in Vegetables at the Farm Gate: Cabbage (Brassica oleracea) Cultivation in Cape Coast, Ghana
Department of Environmental Science, School of Biological Sciences, University of Cape Coast, Ghana
Since 1950, the use of pesticides has increased 50 folds and 2.5 million tons of industrial pesticides are now used annually (Farag et al., 2011). This is to be expected as food security issues particularly in developing countries are very high on the international agenda. Pests contribute significantly to food losses and the control of pests is very central to the attainment of food security at all spatial scales (Iya and Kwaghe, 2007; Al-Eed et al., 2006). Pesticides are extensively used in agricultural production to check or control pests, diseases weeds and other plant pathogens in an effort to reduce or eliminate yield losses and preserve high product quality (Eskenazi et al., 2008; Sanborn et al., 2004). Although pesticides are manufactured under very strict regulation processes to function with logical certainty and minimal impact on human health and the environment, serious concerns have been raised about health risks resulting from residues in food (Damalas and Eleftherohorinos, 2011; Eskenazi et al., 2008). By their very nature, most pesticides show a high degree of toxicity because they are intended to kill certain organisms and thus create some risk of harm (Abdelgadirand and Adam, 2011; Zidan, 2009). Within this framework, pesticide use has evoked grave concerns not only of potential effects on human health but also about impacts on wildlife and sensitive ecosystems (Power, 2010; Damalas and Eleftherohorinos, 2011).
In developing countries such as Ghana, farmers face immense risks of exposure owing to the use of toxic chemicals that are banned or restricted in other countries (Nasr et al., 2007; Al-Eed et al., 2006; Adhikari, 2010). Wrong application techniques, badly maintained or totally unsuitable spraying equipment and inadequate storage practices exacerbate these risks (Al-Wabel et al., 2011). Often the reuse of old pesticide containers for food and water storage also contributes to the risk of exposure (Ecobichon, 2001; Damalas and Eleftherohorinos, 2011). Pesticide residues in/on plants may be unavoidable even when pesticides are used in accordance with good agriculture practices (Uysal-Paha and Bilisli, 2006). Research conducted for the past decade in Ghana and internationally point to the presence of pesticide residues in a number of food items including strawberries, onions, cucumber, lettuce, cabbage, okra, pepper, tomatoes, beans, oranges and lemons (Hanson et al. 2007; Hussain et al., 2002; El-Nahhal, 2004). Pesticide residues do not only constitute a possible danger to soil microfauna and microflora (Pal et al., 2006). In fact, their toxic effects on humans are manifest when bioaccumulation occurs along the food chain after initial plant uptake (Hanson et al., 2007; Pal et al., 2006; Al-Wabel et al., 2011).
Vegetable production is a key component of Ghana’s food security strategy (Parker et al., 2010). One such vegetable is cabbage and a wide array of pests attacks it. Consequently, pesticides such as Attak, Desbin, PAWA-karate, Dursban 4 E, Fura 3G, Bossmate 2.5 E and Pyrinex among others, is used in Ghana to deal with these pests. Most vegetable farmers in Ghana (87%) use synthetic chemical pesticides to control pests on vegetables including a number of highly persistent organochlorine pesticides (Essumang et al., 2008). Lax regulations have allowed inappropriate application practices to develop, such as mixing of two or more pesticides (Farag et al., 2011). Lack of knowledge of the types, use and the effects (additive, synergistic, independent and antagonistic) of these pesticides among small and large scale farmers has resulted in their misuse and consequently their accumulation in various foods and feed items (Essumang et al., 2008; Farag et al., 2011). According to Farag et al. (2011) reliable pesticide residue analysis may be of immense significance as indicators of the potential risks of pesticide exposure on human health.
Pesticides residues on crops are monitored with reference to maximum residue limits and are based on analysis of quantity of a given residue remaining on food product samples. The Maximum Residue Limit (MRL) is not a health-based exposure limit and thus exposure to residue in excess of an MRL does not necessarily imply a risk to health (Boobis et al., 2008). This is because the use of a pesticide would not be allowed if the proposed MRL resulted in long-term and short-term exposure of pesticide residue and the human diet above safety limit. The units of the pyrethroid and organophosphate residues are given in mg kg-1 and μg g-1 , respectively. The aim of the study was to assess whether pesticide residues in cabbage grown under pesticide application comply with the allowed MRLs set by the European Union. Specifically, this study therefore set out to:
| • | Determine the types of pesticides used by vegetable farmers in Cape Coast |
| • | Assess and compare the levels of pesticide residues in cabbage from two main vegetable-growing hubs in Cape Coast |
| • | Compare the levels of pesticide residues to the EU allowed Maximum Residue Limit (MRL) values for each active ingredient identified |
Study area: Forty cabbage samples was collected from a cabbage farm close to the industrial area (Fig. 1) and the University of Cape Coast Technology farm in the Cape Coast Metropolis (5° 14" 0' N to 5° 5" 30' N and 1° 14" 30' W to 1° 22" 30 W) (Parker et al., 2010). Sampling was carried out from December 2010 to April 2011.The original samples were then condensed into 10 composite samples. The metropolis covers about 122 km2. The vegetation is mainly coastal savanna grassland. The area has a bi-modal rainy season from May to June and August to October with an annual rainfall ranging between 750 and 1000 mm (Parker et al., 2010). The soils at the research site are Acrisols (Parker et al., 2010).
Experimental procedure
Extraction process: The method of extraction used was the USEPA method 3510 for extracting multi-residue pesticides in non fatty crops. The extraction was done with ethyl acetate as the solvent. Sodium hydrogen carbonate (NaHCO3) was used to neutralise any acid that may be present. The sample was washed thoroughly with distilled water. The sample was placed in mortor and anhydrous sodium sulphate (Na2SO4) was used to remove water from the sample matrix. Twenty grams of the fresh cabbage was weighed using the Libror-3200 series weighing balance. After weighing, the sample was washed thoroughly with distilled water. The sample was placed in a mortar and with the help of a pestle it was ground into a paste. The paste was transferred into a conical flask with the help of a spatula. Ethyl acetate (40 mL) was added and the mixture was shaken thoroughly. A 5 g portion of sodium hydrogen carbonate (NaHCO3) was added to the mixture followed by 20 g of anhydrous sodium sulphate (Na2SO4) and the entire mixture was shaken vigorously for four to five hours.
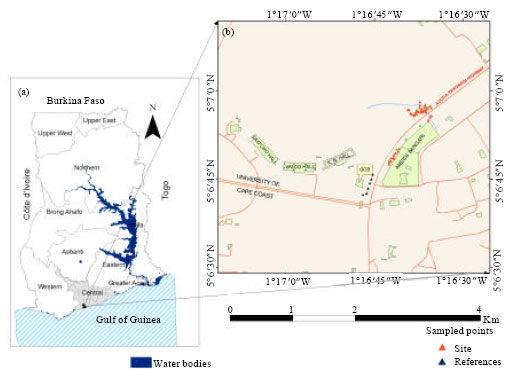 | |
| Fig. 1: | Map of study area showing sampling points; (a) Ghana-central region and (b) Research area (Cape coast) |
This was to ensure that enough of the pesticide residue dissolved in the ethyl acetate. The procedure was repeated for the samples from each location. The mixture was then filtered into a labeled container. The mixture was centrifuged at a speed of 1800 rpm for 5 min and the organic layer was decanted into a container. A 1:1 mixture of 5 mL ethyl acetate and cyclohexane was then added.
Procedure for clean-up: A 10 mm chromatographic column was filled with 3 g activated silica gel and topped up with 2 to 3 g of anhydrous sodium sulphate. Next, 5 mL of n-hexane was added to the column by allowing the tap to run. The residue in 2 mL n-hexane was then transferred onto the column and the extract rinsed thrice with 2 mL hexane. The procedure was repeated for all the samples. The samples from the UCC Technology village (School farm) were labeled S.F 1-S.F 5 and the samples from the farm behind Ameen Sangari industries were labeled A.M 1-A.M 5. The samples were sealed and placed in the refrigerator at the laboratory below room temperature to prevent evaporation of the ethyl acetate. The crude extract was filtered and taken to the Ghana Standards Board Analytical Chemistry Laboratory for Gas Chromatography analysis. Spectroscopic data on pesticide residues detected in the samples is shown in appendix 1.
Gas chromatography: The instrument used in the GC analysis was Varian CP-3800 GC. It is easily configured to perform online analysis for monitoring critical gas and liquid process streams. A 25 μL glass Hamilton syringe was used to inject the GC samples. Only 2-4 μL of the sample was injected onto the column. The syringe was examined carefully before it was filled. A small amount of the liquid was slowly drawn by raising the plunger and then pressed to expel the liquid back into the liquid. This served to “rinse” the syringe with the sample, ensuring that what was measured in the GC run was the composition of the mixture. The rinsing process was repeated twice. Then the plunger was slowly drawn up again while the needle was in the liquid and the syringe was carefully filled with the liquid. Small air bubbles in the syringe did not affect the GC run. The sample was injected into the injector port. Two things were done sequentially and quickly. The needle of the syringe was pushed through the injector pot and immediately the plunger was pressed to inject the sample. Then immediately the start button on the recorder was pressed. A bit of resistance was felt from the rubber septum in the injector port. The recorder was observed for some time. Within several minutes, it recorded several peaks and the GC run was ended. The recorder printed out the peaks, the retention times and the areas under the peaks.
Calculation of pesticide residue concentration: The pesticide residue concentration was deduced from the following equation:
Where:
| C | = | Concentration of pesticide residue (ppm) |
| a | = | The concentration of the identified analyte in the sample solution from GC determination step (ppm) |
| b | = | The sample equivalent in the extraction step (ppm) |
| d | = | The dilution factor of GPC cleanup step |
| f | = | The dilution factor of Silica gel column cleanup step |
Multivariate statistics: Descriptive statistics of pesticide residues were performed using MS-Excel and SPSS version 16. Elements of descriptive statistics of samples (distribution, dispersion, central tendency) generated included mean, range, minimum, maximum, skewness, kurtosis, variance, median, mode, standard deviation and percentiles. Cluster analysis and Principal Component Analysis (PCA) were also performed using SPSS version 16. PCA produces several important outputs of which two namely Eigen values: the variances accounted for by the component; and eigenvectors: that specify the directions of the PCA axes were considered in the analysis.
Concentrations of pesticide residues: Twenty-one pesticide residues were detected in the cabbage samples of which 9 (allenthrin, bifenthrin, lambda-cyhalothrin, fenvalerate 2, cyfluthrin 3, cypermethrin, cypermethrin 2, permethrin and deltamethrin) are pyrethroids and 12 (diazinon, fenitrothion, ethoprophos, chlorpyrifos, phorate, fonofos, pirimidophos-m, profenofos, malathion, dimethoate, chlorfenvinp and parathion-et) are organophosphates. The level of Allethrin detected in A.M.1 was higher than the Maximum Residual Limit (MRL) by a factor of 20. The level detected in A.M.1 is not legally permitted or acceptable in food under the laws of the EU. Levels of allethrin residues detected in all other samples fall below the MRL for cabbage. Eu maximum residual limits of permitted in cabbage are shown in Table 1 and levels of pesticide residues in samples from Ameen Sangari is shown in Table 2.
The levels of allethrin residues detected in cabbage from four S.F. locations were slightly above the allowed MRLs (Table 3). There was no detected residue of allethrin in S.F3. The average allethrin residue in cabbage from A.M locations was relatively higher than in the S.F samples.
The average value for phorate residue in the cabbages from A.M samples was 1.475 μg g-1 (Table 4). This value is 30 times above the MRL. This is quite high and may be due to over application of pesticides on the cabbage from this location i.e. (vegetable farm behind Ameen Sangari industries). The average peak value for phorate residues in the cabbage from S.F. samples was 0.024 μg g-1. This value is below the MRL. The levels of ethoprophos detected in the cabbage from the vegetable farm behind Ameen Sangari industries were above the maximum residue limit (Table 5).
| Table 1: | EU Maximum Residue Limits (MRLs) of Permitted in Cabbage |
 | |
| Table 2: | Types and levels of pesticide residues in samples from Ameen Sangari area |
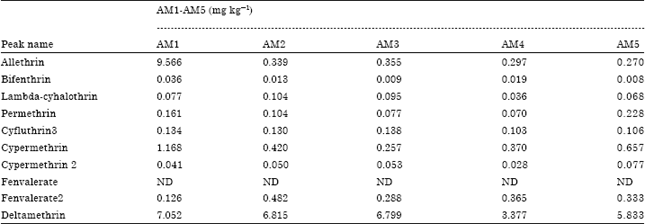 | |
| ND: Not detected; AM1-AM5 represents the five sites at the farm near Ameen Sangari industries | |
| Table 3: | Types and levels of pesticide residues in samples from UCC technology farm |
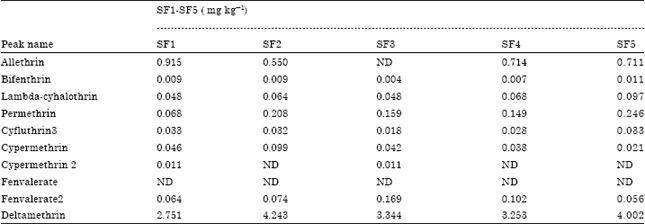 | |
| ND: Not detected; SF1-SF5 represents the five sites at the University of Cape Coast Technology village | |
| Table 4: | Types and levels of pesticide residues in samples from Ameen Sangari area |
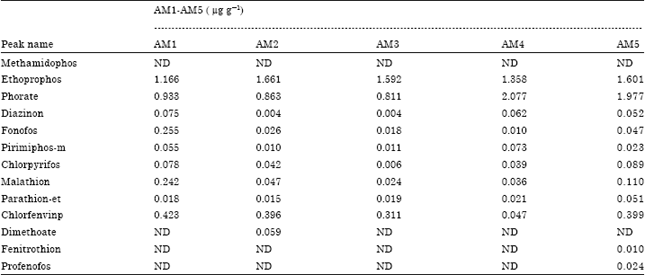 | |
| ND: Not detected; AM1-AM5 represents the five sites at the farm near Ameen Sangari industries | |
| Table 5: | Types and levels of pesticide residues in samples from UCC Technology Farm |
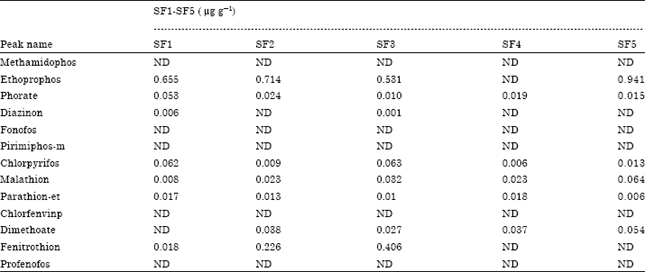 | |
| ND: Not detected; SF1-SF5 represents the five sites at the Unuversity of Cape Coast Technology Village | |
| Table 6: | Summary statistics of pesticide residue concentrations |
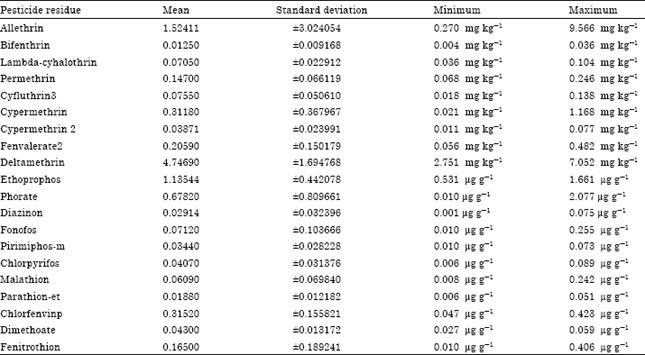 | |
Summary statistics of pesticide residue concentrations: Pesticide residue concentrations show a high degree of variability as shown in Table 6. The highest pesticide residue concentration (9.566 mg kg-1) is attributed to allethrin while the lowest (0.001 μg g-1) is attributed to diazinon. The highest mean concentration of pesticide residue (4.7469 mg kg-1) is attributed to deltamethrin.
Correlation of pesticide residues: Strong associations exist among pesticide residues at different levels of confidence (Table 7). Bifenthrin has strong positive correlations with cypermethrin and malathion at the 0.01 level suggesting a common origin. Lambda-cyhalothrin is strongly correlated with deltamethrin at the 0.05 level.
| Table 7: | Correlation matrix of pesticide residues |
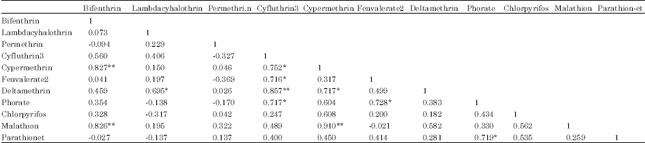 | |
| **Correlation is significant at the 0.01 level (2-tailed). *Correlation is significant at the 0.05 level (2-tailed) | |
Cyfluthrin 3 shows strong positive correlation with cypermethrin, fenvalerate 2 and phorate at the 0.05 level and with deltamethrin at the 0.01 level. Cypermethrin is strongly correlated with deltamethrin, phorate, chlorpyrifos and malathion at different levels of confidence. Fenvalerate 2 shows strong positive with phorate at the 0.05 level whereas parathion-et shows strong positive correlation with phorate at the 0.05 level.
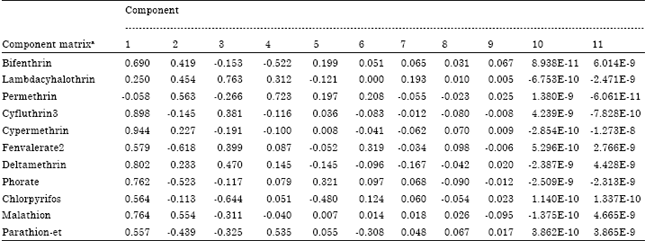
Principal component and cluster analyses: Principal component analysis indicates that the data on pesticide residues is a two-component system explaining 63% of the variability in the data (Table 8). The First component alone explains almost half of the variability in the data whereas first three components extracted explain approximately 80% of the variability in the data. The screen plot (Fig. 2) confirms that the data is a two-component system. From the component matrix (Table 9), component 1 shows strong positive factor loadings on cypermethrin, deltamethrin and cyfluthrin 3 suggesting a common origin whereas component 2 shows strong negative factor loadings on phorate and fenvalerate 2. Component 3 exhibits strong positive factor loadings on lambda-cyhalothrin and strong negative factor loadings on chlorpyrifos.
| Table 8: | Variance of data explained by each component |
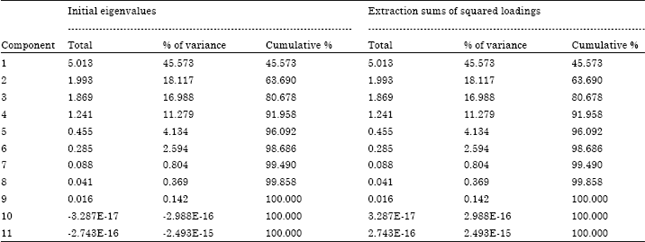 | |
| Extraction method: Principal component analysis | |
| Table 9: | Component matrix of extracted components |
 | |
| Extraction method: Principal component analysis, a11 components extracted | |
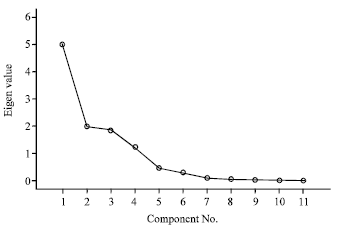 | |
| Fig. 2: | Scree plot showing eigenvalues and number of components extracted |
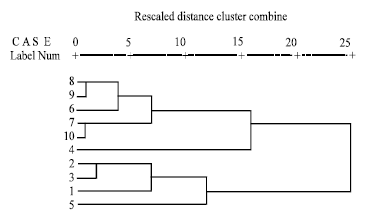 | |
| Fig. 3: | Dendrogram showing each cluster and respective members |
Cluster analysis grouped the sample locations into two spatially dissimilar categories based on the levels of pesticide residues. Cluster one consists of 6 members whereas cluster two consists of 4 members (Fig. 3).
The levels of allethrin detected in samples from both locations were above the MRL for cabbage. This may either be due to early harvest of the cabbage which does not allow most of the allethrin to decompose or be degraded by the cabbage plant or due to differences in soil composition in these sites. Fenvalerate was not detected in any of the samples analysed from the two locations. This may be attributed to the fact that they do not use pesticides that contain fenvalerate as an active ingredient. It may also be due to decomposition or degradation of fenvalerate before the analysis was carried out. Some of the pesticides used on cabbage in the farm behind Ameen Sangari industries include Attak, Desbin, PAWA-karate whereas on the UCC Technology farm Dursban 4 E, Fura 3G, Bossmate 2.5 E, Pyrinex are used. It has been established that the levels of Lambda-cyhalothrin, Chlorpyrifos, Fenitrothion and Cypermethrin residue in tomatoes were above their respective MRL values (Essumang et al., 2008). Consequently, this study confirms the work of Essumang et al. (2008) regarding the observation that some pesticide residues in vegetables are above the allowed MRLs. However, unlike in this study which focused on cabbage, Essumang et al. (2008) experimented on tomatoes. They collected tomato fruits from different market places in Kumasi in the Ashanti region and Cape Coast in the central region of Ghana for analysis. The levels of ethoprophos detected in the cabbage from the vegetable farm behind Ameen Sangari industries were above the maximum residue limit by factors ranging from 58.3 and 80.1. The application of pesticides on tomatoes will be different from the application of pesticides on the cabbage used in this research in terms of dosage and the frequency of pesticide application. There were no detected peaks of fenvalerate and methamidophos in the samples used in this research. Significant levels of fenvalerate and methamidophos have been detected in the analysis of pesticide residues in fruits and vegetables in Sweden (Pihlstrom et al., 2007). This work appears to contradict the work of Pihlstrom et al. (2007) given that neither fenvalerate nor methamidophos were detected in the study. It is important however; to distinguish that Pihlstrom et al. (2007) based their work on market-level sampling whereas this study focused on cabbage obtained at the farm gate. Again, Ripley et al. (2000) reported pesticide residues in vegetables (cabbage) and fruits in Canada; however, the levels were far below the MRLs. EL-Saeid (2003) used Supercritical Fluid Extraction (SFE) in the analysis of pesticide residues unlike in this study where Gas Chromatography was used. Also, EL-Saeid (2003) sampled canned foods, fruits and vegetables whereas this study sampled fresh cabbage. Ahn et al. (2011) has shown that diazinon and chlopyrifos residues were stable up to 30 months and this needs public attention. An integrated approach to pesticide use should be encouraged and this should fit broadly in a framework in which certain criteria are used for pesticides selection, specific instructions are followed for their application on crops and residue analysis is used as one of the tools for enforcement (Damalas and Eleftherohorinos, 2011). Pesticides that are selected for use on vegetables should be:
| • | Biologically effective (high selectivity, fast impact, optimal residual effect, good plant tolerance, low risk of resistance) |
| • | User friendly (low acute toxicity and low chronic toxicity, optimum formulation, safe packaging, easy application method, long store stability) |
| • | Environmentally friendly/compatible (low toxicity to non-target organisms, fast degradation in the environment, low mobility in the soil, no residues in food and fodder above the MRLs, low application rate) |
Presence of pesticide residues in vegetables and fruits is an indicative change in use pattern of pesticides in Ghana where shift has taken place from organochlorines to the easily degradable groups of pesticides over the last few years. Monitoring studies are imperative to know actual status of contamination due to toxic pesticide residues for future policies as well as to strengthen the confidence of consumer in quality of food. It is therefore, suggested that such studies may be extended to other fruits grown in different agro-climatic regions of Ghana. These issues are central to minimising the risk posed to human health on exposure to pesticides in food.
Pesticides are extensively used in agricultural production to check or control pests, diseases weeds and other plant pathogens in an effort to reduce or eliminate yield losses and preserve high product quality. This study assessed the types of pesticides used by cabbage farmers and the levels of pesticide residues in detected in the cabbage samples of which 9 are pyrethroids and 12 are organophosphates. The cabbage samples from two vegetable growing hubs in Cape Coast, Ghana. Twenty-one pesticide residues were results are as follows: Allethrin (1.52411±3.024054 mg kg-1), deltamethrin (4.74690±1.694768 mg kg-1), cypermethrin (0.31180±0.367967 mg kg-1), fenvalerate 2 (.20590±0.150179 mg kg-1), permethrin (0.14700±0.066119 mg kg-1), ethoprophos (1.13544 μg g-1±0.442078), phorate (0.67820 μg g-1±0.809661), chlorfenvinp (0.31520±0.155821 μg g-1) and fenitrothion (0.16500±0.189241 μg g-1). High levels of pesticide residue were observed for allenthrin (9.566 mg kg-1) and phorate (2.077 μg g-1). Methamidophos and fenvalerate were not detected in any of the samples. The level of Allethrin detected in some cabbage samples was higher than the Maximum Residual Limit (MRL) by a factor of 20. At least one pesticide residue was detected in more than half the samples analyzed. Broadly, this could be attributed to wrong application techniques, badly maintained or totally unsuitable spraying equipment, inadequate storage practices and often the reuse of old pesticide containers for food and water storage. Certainly, this suggests the need to enforce regulations and educate vegetable farmers regarding the proper use and management of pesticides.
Dr h D joshi Reply
i am also working on this field but i need standard pesticide for identification do u have any source? I have limited budget