Research Article
Bioremediation of Malathion from Environment for Pollution Control
Department of Food Technology, Guru Nanak Institute of Technology,Kolkata-700114, India
A single pesticide cannot control all the pests. The first chemical used to combat plant pathogens fungi and to poison plant-eating insects were inorganic materials containing copper, sulfur, arsenic and other substances. Some naturally occurring organic insecticides e.g., Protenone were also used. These eventually entered in soil and toxic elements some times accumulated to undesirable levels. Synthetic organic chemicals, many of which contained phosphorus, mercury, arsenic and chlorine bound to the organic molecules, largely replaced these early pesticides. These organic pesticides were much more specific in their action and were instrumental in raising crop yields. They too entered the soil. All organic compounds that occur naturally and most of those synthesized by man are decomposed in soil by a combination of chemical and biological action but the rate of decomposition vary with the compound. Organic pesticides are attacked by soil bacteria of several genera and eventually decomposed. But this is not true in case of a large number of synthetic chemicals. In recent years people have added to the number of slowly degradable or non-biodegradable compounds in the soil by adding different pesticides in many parts of world. The chemical nature of herbicides and insecticides covers an extremely broad range of organic compounds, organic acids, nitro phenols, chlorinated organic acids and other organic substances (William et al., 1973).
Malathion is an organophosphorus pesticide having molecular formula C10H19O6PS2 and structure shown in Fig. 1 (Tomlin, 1994).
It is an insecticide used for many insects including aphids, spider mites, scale insects, house fly and mosquitoes as well as large number of other sucking and chewing insects attacking fruits, vegetables, ornamentals and stored products. It has a cholinesterase activity and it kills the insects by affecting their nervous system.
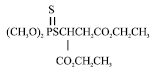 | |
| Fig. 1: | Chemical structure of malathion [S-1, 2-bis (ethoxycarbonyl) ethyl o,o-dimethyl phosphorodithioate] |
Malathion comes in two forms; a pure form of colorless liquid and a technical grade solution (brownish-yellow liquid), which contains Malathion (greater than 90%) and impurities in a solvent. The technical grade is approximately 10 times stronger in causing death to laboratory animals. The FDA and EPA allow a maximum amount of 8 mg L-1 of Malathion to be present as a residue an specific crops used as foods. Because Malathion can be dangerous to humans, EPA requires that a certain time must pass between the time of application of the insecticide and entry by a worker into a field where the chemical has been applied. Usually at least 12 h must pass between application and entry, but in some cases such as when workers are entering a field to hand harvest or hand prune the crops, time period as long as 6 days must pass between application and entry into the field. In this way, exposure to Malathion can be controlled and accidental exposures can be prevented (ATSDR, 2001).
Malathion contamination may be originated from two sources.
| • | Point sources |
| • | Non-point sources |
Point Sources
These sources arise from daily/routine handling operation and waste disposal. These include means of transportation, agrochemical operation. These have definite identity with constant volume and fixed composition of the effluent discharged.
Non-Point Sources
The sources are diffusion, situated far beyond the sight having waste of unknown composition and volume. Also arises from the course of routine operating practices, which include application of pesticides to the field and tank loading, mixing and rinsing (Boopathy, 2000). General means of removal of pesticides from soil and water are adsorption, photodecomposition, leaching and volatilization. But the advantage of the Bioremediation process is, it avoids the use of costly chemicals and lengthy steps of physical and chemical removal process.
Bioremediation process can be either biodegradation or biosorption. When biodegradation occurs it can follow any of the following mechanisms.
| • | The pesticide can serve as a substrate for growth and energy |
| • | The xenobiotic compound can undergo co-metabolism, i.e., the microorganism can transform the pesticide but cannot derive energy for growth from it |
| • | The entire pesticide molecule or an intermediate of it can be conjugated with naturally occurring compound |
| • | The pesticide is incorporated and accumulated within the organism |
When biosorption occurs the pesticide molecule will be either absorbed in to the cell or it will remain adsorbed on the surface of the organism.
Bioremediation of Malathion
Malathion is degraded by carboxyesterase enzyme and it is detected in several fungi like Aspergillus sp., Penicillum sp. and Rhizoctonia sp. (Mostafa et al., 1972). Omar (1998) and Hasan (1999) also demonstrated the same type of fungal utilization and degradation of Malathion. Several Aspergillus sp. showed greatest potential for utilizing Malathion as phosphorous and carbon source. Not only the fungal cells but also the bacterial cells are also capable of biodegradation of Malathion (Kamal et al., 2008) found that a strain of Bacillus thuringiensis MOS-5(Bt) isolated from agricultural waste water was able to degrade Malathion cometabolically. The major degraded products were Mal-Monocarboxylic Acid (MMA) and Mal-Dicarboxylic Acid (MDA). Xie et al. (2009) found a bacterium, Acinetobacter johnsonii MA19, that could degrade Malathion with cometabolism. The bacterium A. johnsonii MA19 was found to have the capability for malathion biodegradation and environmental bioremediation when some suitable conventional carbon sources are supplied Tsezos and Bell (1991) studied the fate of Malathion following interaction with live and dead microbial biomass of Rhizopus arrhizus. The experimental results from equilibrium, kinetic and carbon-14 studies suggested that Malathion is adsorbed and then chemically decomposed by the microbial biomass into water soluble products. This chemical transformation that was effected by the microbial biomass is not an active metabolic process as it is also exhibited by dead cells and isolated microbial cell walls. Cells of isolated Bacillus sp. S14 was found to have the capability of removal of Malathion from dilute aqueous solution and the capability of this removal by dead cell (Adhikari et al., 2010) indicates that this process is not an active metabolic process but fully controlled by the functional groups present on the microbial surface.
Bioremediation technique for removal of Malathion is nothing but a very effective and interesting field with the advantages like it is a natural process with less cost requirement compared to other methods and this process can be applicable at the place where the problem is located. Although, this technique has some disadvantages like formation of undesirable degraded products, desorption of Malathion and disposal of it, disposal of cells containing Malathion in adsorbed/absorbed condition. These problems can be solved with further monitoring of the process and there are lots of scopes for research work in this field.