Research Article
Histological Alterations Induced by Lead in the Testes of the Quail Coturnix coturnix
Department of Zoology, College of Science, King Saud University, P.O. Box 2455, Riyadh, 11451, Saudi Arabia
Lead intoxication is probably the most common form of heavy metal intoxication and is well documented as one of the most dangerous and insidious poisons (Cheremisinoff, 1990; Schulte et al., 1994; Shy, 1990; Yagminas et al., 1990; Jones, 1991; Fischbein, 1992). Continuous environmental and occupational lead exposure may contribute to haemopoietic, renal, nervous, gastrointestinal and reproductive disorders in man and animals. The absorbed lead is conjugated in the liver and passed to the kidney, where a small quantity is excreted in urine and the rest accumulates in various body organs and interferes with their function (Powell et al., 1999; Jarrar, 2001, 2003; Jarrar and Mahmoud, 1999, 2000a, b, 2002; Taib et al., 2004).
Birds may come in contact with metallic objects that contain lead. In addition, contaminants in prey are often biomagnified in hungry migrating birds (Fair and Ricklefs, 2002). Physical, behavioral, neurological, gastrointestinal and hematological clinical signs are seen in birds with lead intoxication, which may be fatal if not treated promptly (Mateo et al., 1998). Lead accumulation in tissues is affected by the physiological status of the bird and levels of accumulation differ between species (Almansour, 2004, 2007). Lead toxicosis is one of the most commonly diagnosed diseases of captive falcons in Saudi Arabia and is responsible for mortality in both companion and free-ranging birds (Mateo et al., 1998; Samour and Naldo, 2002).
The adverse effects of occupational lead exposure on mammalian male reproductive function remain a controversial subject and show major discrepancies among investigators. There is accumulating evidence that heavy metals inhibit reproduction by interfering with the endocrine system and affecting spermatogenesis and steriodogenesis (Thomas and Brogan, 1983; Saxena et al., 1987; Johansson and Pellicciari, 1988; Rodamilans et al., 1988; Kime, 1998; Ahmad et al., 2003; Yamaguchi et al., 2007). However, most of the lead toxicological data in birds were derived from waterfowl that had ingested lead shot or birds of prey that consumed tissues contaminated with lead (Vyas et al., 2001).
Little is known about the histological alterations induced by chronic lead exposure in avian testes (Dauwe et al., 2004; Mazliah et al., 1989). With this objective, a detailed histological study was undertaken to investigate the effect on the testes of the quail Coturnix coturnix of oral exposure to various concentrations of lead acetate trihydrate in drinking water.
The present investigation was carried out in the Zoology Department, King Saud University over a period of 12 months (October 2006-September 2007).
A total of 60 adult males of the quail Coturnix coturnix of the same age and weighing 185-205 g-1 were used in this study. Birds were randomly divided into five groups of 12 quails each. Four groups, designated groups 1-4, were exposed to ascending concentrations of lead acetate trihydrate (0.1, 0.25, 0.5 and 1.0%) successively in drinking water for 6 months. The fifth group was used as a control. All groups were fed on a standard laboratory animal diet pellet and maintained under standard laboratory conditions that included controlled temperature (23 ± 2 °C) and light, dark periods (12:12). Two quails from each group were dissected at monthly intervals throughout the experimentation period.
After dissection, representative tissue specimens from both testes of each quail were removed rapidly and fixed in neutral buffered formalin (10%). Tissue specimens were then dehydrated in ascending grades of ethanol (70, 80, 90, 95 and 100%), cleared in 2 changes of chloroform, impregnated with 2 changes of molten paraffin wax and finally embedded and blocked out. Tissue sections (4-5 μm) were stained with hematoxylin and eosin (HE) according to Pearse (1985).
Lead acetate trihydrate induced marked changes in the testicular tissue of the exposed quails and included vascular, interstitial and tubular alterations.
Vascular Alterations
The earliest vascular change due to lead intoxication involved the distension of interstitial blood vessels (Fig. 1a). This alteration appeared as early as 2 months post-exposure in all intoxicated birds.
Hyperplasia of endothelial lining cells in some testicular arteries was observed (Fig. 1b). This change was recorded at the 0.5% lead acetate level for 2 months and more of exposure. Testicular arteriolar branches showed hypertrophy of endothelial lining cells associated with vacuolation of the tunica media, with perivascular edema discerned in testicular tissues after 0.5% lead acetate trihydrate exposure for 2 months and more (Fig. 1c).
Leydig Cell Hyperplasia
Occasional Leydig cell hyperplasia associated with fibroblast proliferation was discerned in birds that received 0.1% lead acetate trihydrate at 2 months post-exposure (Fig. 1d). This alteration was more frequent for higher concentrations at 3 months of exposure to lead acetate trihydrate.
Interstitial Edema
Mild edema between seminiferous tubules appeared in quails that received at least 0.25% lead acetate for 3 months and more. Edema was evidenced by expansion of intertubular connective tissue associated with separation of seminiferous tubules due to accumulation of edematous fluid (Fig. 1e, f).
Thickening of Somniferous Tubular Basement Membranes
The basement membranes of some seminiferous tubules thickened (Fig. 2a). This alteration was seen in the testes of quails that received 0.5 and 1% lead acetate for 4 months and more. The thickening of tubular basement membranes was associated with fibroblast proliferation (Fig. 2b) and a large number of the proliferated fibroblasts were observed in conjunction with the outermost layer of the thickened basement membranes. This alteration was first recognized in birds exposed to 0.5% lead acetate trihydrate for 4 months and was more frequent in birds intoxicated with 1% lead acetate trihydrate for 6 months.
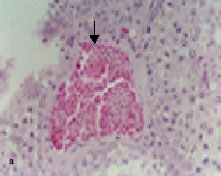 � � 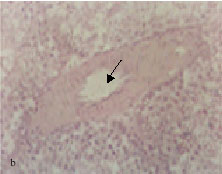 | |
Fig. 1: | Light micrograph sections stained hematoxylin and eosin showing: Distension of an arterial branch, 5%lead acetate trihydrate for 2 months (arrow) Hyperplasia of the endothelial cells, 0.5% lead acetate trihydrate for 3 months (arrow) Hypertrophy and hyperplasia of the endothelial lining associated with vacoulation of the tunical media of an arteriolar branch (arrows), 1% lead acetate trihydrate for 3 months Leydig cell hyperplasia associated with fibroblast proliferation (arrow), 0.25% lead acetate trihydrate for 2 months Perivascular edema accompanied by separation of seminiferous tubules (arrow), 0.25% lead acetate trihydrate for 3 months Accumulation of edematous fluid between seminiferous tubules (arrow), 0.5% lead acetate trihydrate for 5 months |
a: b: c: d: e: f: | |
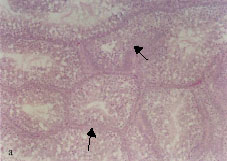 � � 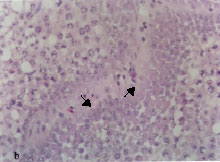 | |
| Fig. 2: | Light micrograph sections stained with hematoxylin and eosin, showing: Thickening of the seminiferous tubular basement membrane (arrow), 0.5% lead acetate trihydrate for 4 months Fibroblast proliferation (arrows) associated with thickening of the seminiferous tubular basement membrane, 0.5% lead acetate trihydrate for 5 months Spermatid giant cells (arrows), 1% lead acetate trihydrate for 6 months Pyknosis of spermatocyte nuclei (arrow), 0.5% lead acetate trihydrate for 5 months Desquamated spermatocytes in the lumens of seminiferous tubules (arrows), 1% lead acetate trihydrate for 6 months Marked atrophy of seminiferous tubules (arrows), 1% lead acetate trihydrate for 6 months |
a: b: c: d: e: f: | |
Degenerative Tubular Changes
Multinucleated spermatid giant cells were detected in seminiferous tubules of birds exposed to 0.5% and more of lead acetate trihydrate at the last month of exposure (Fig. 2c).
Pyknosis of Spermatocyte Nuclei
Some spermatocytes of intoxicated birds showed nuclear pyknosis (Fig. 2d). The pyknotic spermatocytes were first recognized in birds exposed to 1% lead acetate trihydrate for 3 months and thereafter became more frequent.
Spermatocyte Desquamation
Accumulation of desquamated spermatocytes in association with cellular debris was observed in the lumens of seminiferous tubules of intoxicated birds (Fig. 2e). Desquamation of spermatocytes was apparent in birds that received 1% acetate trihydrate for 6 months.
Atrophy of Seminiferous Tubules
Occasional tubular atrophy was observed after 3 months of exposure to 0.5 and 1% lead acetate trihydrate. However, marked atrophy of seminiferous tubules with complete arrest of spermatogenesis was detected in some birds exposed to 1% lead acetate trihydrate at the last month of exposure (Fig. 2f).
No comparable histological findings were detected in the testes of control birds.
The results of the present investigation showed that subtoxic chronic lead exposure caused progressive vascular, tubular and interstitial testicular damage. These findings are in agreement with that reported by Veit et al. (1983), who reported degeneration of seminiferous tubules and interstitial cells after the ringed turtle dove Streptopelia risoria was exposed orally to lead. Moreover, lead chronic intoxication caused histological changes in mature testes of the rooster (Mazliah et al., 1989). In the current study, the early changes in testicular tissue due to lead intoxication mainly comprised arterial congestion and Leydig cell hyperplasia. Arterial congestion was most likely the result of direct damage exerted by lead on the vascular walls. The hyperplasia was possibly an early adaptive cellular response to lead-induced cytotoxicity.
The present results indicate that the observed nuclear pyknosis of spermatocytes was an initial necrotic change induced by lead exposure. This nuclear alteration was associated with testicular tubular cell degeneration and necrosis, tubular atrophy, desquamation of the germinal epithelium and complete arrest of spermatogenesis. These tubular changes might indicate the susceptibility of the seminiferous epithelium to lead toxicity with a possible effect on the intercellular junction between the strata of the germinal cells. The appearance of intratubular spermatid giant cells in the tissues of intoxicated birds may indicate lead-induced spermatocyte degeneration. Spermatid giant cells are known to develop as a result of the failure of cytokinesis during meiotic division or the disruption of cytoplasmic bridges connecting germ cell clones (Stainley and Akbarsha, 1992).
The findings of the present work are basically in agreement with those of previous investigations conducted on the testes of some mammalian models. The testicular tissue of albino rats exposed to lead acetate through intraperitoneal inoculation presented similar histological changes (Ahmad et al., 2003). On the other hand, Bogden et al. (1992) reported neither effect of lead on Sertoli cell function nor any evidence of seminiferous tubular atrophy in adult Sprague rats after 10 weeks of exposure to 1% lead acetate. Additionally, rabbits exposed to lead manifested no change in sperm count and testicular morphology was almost normal (Willems et al., 1982). The discrepancies among investigators regarding the effects of lead on spermatogenesis and testicular histology could be due to variations in the level of exposure, duration, route of administration and animal species used in experiments. However, occupational exposure to lead has been shown to compromise human fertility through lead-induced testicular toxicity followed by hypothalamic or pituitary disturbances in the long term (Thomas and Brogan, 1983; Rodamilns et al., 1988) Some environmentalist reported that environmental exposure to lead is more widespread than appreciated and is associated with the production of human male subfertility (Richard et al., 2004). Other investigators concluded that lead exposure during the growing period might result in reproductive impairment during adulthood (Saxena et al.,1987; Johansson and Pellicciari, 1988).
In conclusion, the results of the present study indicate that chronic exposure to lead is capable of inducing significant testicular histological changes in the quail Coturnix cotunix. Moreover, our investigation has demonstrated that this quail species is a suitable avian model for the study of lead toxicity.
The present research was supported by the Research Center, College of Science, King Saud University (Research Project No. Zoo/2008). The author gratefully acknowledges this support.