ABSTRACT
Selenium (Se) is a metalloid and now becomes dietary important for human. The genotoxic potentiality and genetics of tolerance for this heavy metal is more or less lacking. The present communication deals with the cytological response of wheat (Tritium aestivum) and safflower (Carthamus tinctorius) to Se treatment. The 10-3 M concentration of SeO2 and 10-4 M concentration of SeO2 was used as a source of Se for treating wheat and safflower respectively. Control experiments were also conducted using Hoagland’s solution and normal mitotic pattern was observed. Tritium aestivum had 2n = 42 and C. tinctorius had 2n = 24. The accessions exhibited differential cytological response to Se. Although, this heavy metal was mito-toxic to both wheat and safflower but genotoxicity was more pronounced in the accessions of wheat. In response to the Se treatment, both wheat and safflower were displaying more or less similar types of mitotic anomalies. Presented data will help to understand tolerance and effects of Se in plants.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/rjes.2011.82.87
URL: https://scialert.net/abstract/?doi=rjes.2011.82.87
INTRODUCTION
Heavy metals like Pb, Hg, Se, As, Cd, Co, Cu, Ni, Zn and Cr are phytotoxic above their threshold levels. They infect the environment by affecting soil properties its fertility, biomass and crop yields and ultimately human health. Se, a chemical analog of S, is thought to be assimilated and volatilized by plants using the same enzymes of the S assimilation pathway (Brown and Shrift, 1982; Terry and Zayed, 1994). Controversy exists whether selenium is an essential plant micronutrient; however, there is evidence that trace amounts of selenium can enhance the growth of some plant species. Lower concentrations of selenium inhibit lipid peroxidation in Lolium perenne and this decrease coincides with increased growth (Hartikainen et al., 2000). Se was also able to promote the growth of UV-stressed lettuce (Latuca sativa) seedlings and delayed the death of plants subjected to severe UV-stress (Pennanen et al., 2002). Low concentrations of selenium decreased the growth but increased the multiplication rate in Lemna minor (Severi, 2001). X-ray Absorption Spectroscopy (XAS) showed that wild-type Indian mustard treated with selenate accumulated Se, mainly in the form of selenate, in root and shoot tissue, whereas selenite- or SeMet-treated plants accumulated an organoselenium compound similar to SeMet (De Souza et al., 1998; Zayed et al., 1998). SeMet is volatized much more readily than selenate or selenite; the rate of Se volatilization measured separately from roots and shoots of Indian mustard supplied with SeMet was 48 to100 times higher than plants supplied with selenate or selenite (Zayed et al., 1998).
Astragalus bisulcatus has the capacity to accumulate Se to high concentrations but it has a slow growth rate. It has been proposed that in Selenocysteine Methyltransferase (SMT) Specifically Methylates Selenocysteine (SeCys) to produce the nonproteinamino acid methylselenocysteine MetSeCys, which causes a reduction in the intracellular concentrations of SeCys and selenomethionine (SeMet), thus preventing their incorrect insertion into protein (Madaan, 2008). Brassica juncea over expressing the A. bisulcatus SMT gene, exhibited a greatly increased accumulation of MetSeCys and tolerance to Se compounds, in particular selenite (LeDuc et al., 2004).
MATERIALS AND METHODS
During the present study (2006), 163 accessions of wheat (Tritium aestivum) and 234 accessions (Carthamus tinctorius) were tested for their cytological response to selenium (Se) treatment. At least fifty seeds each of both wheat and safflower were grown in separate sterilized petriplates lined with cotton pads, sandwiched between filter papers for raising different control and treated sets. Selenium dioxide (SeO2) was used as source of Se. Control sets were raised in Hoagland’s solution lacking selenium. The seeds of wheat were grown in 10-3 M whereas of safflower in 10-4 M solution of the SeO2, prepared in Hoagland’s solution. Both control and treated sets were raised in Calton’s Seed Germinator in total darkness at 20°C. Root tip samples, collected from treated and control sets after the 48 h of the germination of seeds, were fixed in acetic alcohol (3 parts absolute ethanol+1 part glacial acetic acid) fir at least 48 h and stored in 70% ethanol in refrigerator. Wheat root tips were smeared and squashed in 1.5% aceto-carmine while of the safflower in 1% aceto-orcein. Genotoxic effects of Se were quantified by working out mitotic index (MI), active mitotic index (AMI), type of mitotic anomalies and Total Mitotic Anomaly (TMA).
MI, AMI and TMA were calculated using the following formulae:
 |
A parameter called Response Coefficient (RC) was calculated, using the following formula for estimating the toxicity imposed by Se treatment.
where, VT is value of treated set, VC is value of control set.
The negative value of RC’s signified inhibition while positive values indicated stimulation. On the basis of RCs of the above mentioned parameters, accessions were categorized into five categories A_E. A=> -0.20; B = -0.20 to -0.39; C = -0.40 to -0.59; D = -0.60 to -0.79; E =< -0.80. Category A comprised of Tolerant (T), category E of Non-Tolerant (NT) and categories B-D of partially tolerant accessions.
RESULTS AND DISCUSSION
Wheat and Safflower exhibited differential cytological response against Se. The MIs could not be affected in both the crops. While in about 36% accessions of safflower Se could not adversely affect AMI, in wheat this could adversely effect only about 3% accessions. Further, about 8% accessions of safflower were having greater then 80% reduction in AMI but only about 4% accessions of wheat showed this behavior (Table 1).
Se could induce mitotic anomalies in 65% accessions of wheat while in safflower it could induce only on about 7% accessions; this clearly concluded that selenium was more toxic to wheat (monocot) than to safflower (dicot). Frequency distributions of the accessions (%) of wheat and safflower having different frequency (%) of TMA are shown in Fig. 1a and b. Different types of mitotic anomalies induced by Se in the two crops included: presence of c-metaphase (Fig. 2, 9), chromosomal erosion (Fig. 3), lagging of chromosomes (Fig. 4, 5), clumping of chromosomes at metaphase (Fig. 6, 8), chromatin bridge at anaphase (Fig. 7, 12), formation of groups of chromosomes at metaphase (Fig. 10), restitution nucleus formation (Fig. 11), presence of hyperploid cells (Fig. 13), formation of micro-nuclei at telophase (Fig. 14) and transcellular migration of chromatin material between neighbouring cells (Fig. 15).
Hershbach began growing plants like mustard that removed the toxic selenium compounds by concentrating them into its harvestable parts and thereby prevented the contamination of water sources. The strategy was later on named as phytoremediation.
| Table 1: | Overall assortment (%) of the accessions (wheat and safflower) on the basis of AMIs to different classes of response coefficient for Se treatment |
 | |
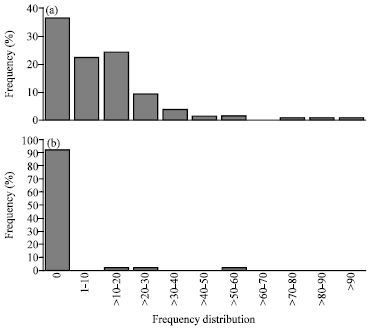 | |
| Fig. 1: | (a, b) Frequency distributions of the accessions (%) of wheat and safflower having different frequencies of Total Mitotic Anomaly (TMA) |
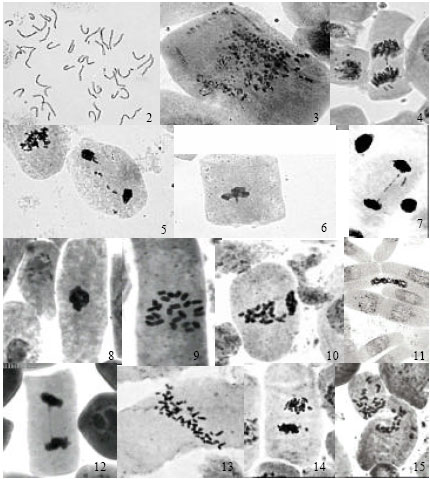 | |
| Fig. 2-15: | Photomicrograph of mitotic anomalies induced by Se. (2-7 wheat) 2: C-metaphase, 3: Chromosomal erosions, 4-5: Lagging of chromosomes, 6: Clumping, 7: Chromatin bridge. (8-15 safflower) 8: Clumping, 9: C-metaphase, 10: Grouping at metaphase, 11: Restitution nucleus, 12: Chromatin bridge, 13: Hyperploid cell, 14: Micronuclei, 15: Transcellular migration |
Phytoremediation is an inexpensive, efficient and environment-friendly technology for the remediation of inorganic Se (Terry and Zayed, 1998). Selenium has been called as essential toxin, as it is required for certain cell processes and enzymes, but it becomes deleterious at greater doses (Russo et al., 1997; Patterson and Lavender, 1997; Knekt et al., 1998; Fleet, 1997; Shamberger, 1985; Young and Lee, 1999; Burguera et al., 1990; Clark et al., 1996; Combs and Liu, 2001; Burk, 2001; Combs et al., 1997). It prevents the oxidation of Low-Density Lipoproteins (LDL) as it is an anti-oxidant, which further prevents the formation of plaque in the coronary arteries and thus saves from heart diseases (Ozer et al., 1995; Lapenna et al., 1998; Neve, 1996; Coppinger and Diamond, 2001). Body immune system naturally makes free radicals that can also harm healthy tissue. Se as an anti-oxidant, may help in controlling levels of free radicals and help to relieve symptoms of arthritis (Aaseth et al., 1998). Indeed recent experiments with transgenic plants demonstrated that the proposed Se assimilation and volatilization pathway (Terry and Zayed, 1994) uses enzymes of the S assimilation and volatilization pathway. Phtyoremediation through Brassica juncea, Arabidopsis thaliana, Astragalus bisulcatus (Pilon-Smits and Pilon, 2002; Mudgal et al., 2010; Madaan and Mudgal, 2009) has proved useful.
REFERENCES
- Brown, T.A. and A. Shrift, 1982. Selenium: Toxicity and tolerance in higher plants. Biol. Rev., 57: 59-84.
Direct Link - Hartikainen, H., T.L. Xue and V. Piironen, 2000. Selenium as an anti-oxidant and pro-oxidant in ryegrass. Plant Soil, 225: 193-200.
CrossRefDirect Link - Pennanen, A., T.L. Xue and H. Hartikanien, 2002. Protective role of selenium in plants subjected to severe UV irradiation stress. J. Applied Bot., 76: 66-76.
Direct Link - Severi, A., 2001. Toxicity of selenium to Lemna minor in relation to sulphate concentration. Physiologia Plantarum, 113: 523-532.
Direct Link - De Souza, M.P., E.A.H. Pilon-Smits, C.M. Lytle, S. Hwang and J. Tal et al., 1998. Rate limiting steps in selenium accumulation and volatilization by Indian mustard. Plant Physiol., 117: 1487-1494.
Direct Link - Zayed, A.M., C.M. Lytle and N. Terry, 1998. Accumulation and volatilization of different chemical species of selenium by plants. Planta, 206: 284-289.
CrossRefPubMedDirect Link - LeDuc, D.L., A.S. Tarun, M. Montes-Bayon, J. Meija and M.F. Malit et al., 2004. Overexpression of selenocysteine methyltransferase in Arabidopsis and Indian mustard increases selenium tolerance and accumulation. Plant Physiol., 135: 377-383.
Direct Link - Russo, M.W., S.C. Murray, J.I. Wurzelmann, J.T. Woosley and R.S. Sandler, 1997. Plasma selenium levels and the risks of colorectal adenomas. Nutr. Cancer, 28: 125-129.
PubMedDirect Link - Patterson, B.H. and O.A. Levander, 1997. Naturally occurring selenium compounds in cancer chemoprevention trails: A workshop summary. Cancer Epidemiol. Biomarkers Prev., 6: 63-69.
PubMedDirect Link - Knekt, P., J. Marniemi, L. Teppo, M. Heliovaara and A. Aromaa, 1998. Is low selenium status a risk factor for lung cancer? Am. J. Epidemiol., 148: 975-982.
Direct Link - Fleet, J.C., 1997. Dietary selenium repletion may reduce cancer incidence in people at high risk who live in areas with low soil selenium. Nutr. Rev., 55: 277-290.
PubMedDirect Link - Young, K.L. and P.N. Lee, 1999. Intervention studies on cancer. Eur. J. Cancer Prev., 8: 91-103.
PubMedDirect Link - Burguera, J.L., M. Burgera, M. Gallignani, O.M. Alarcon and J.A. Burgueera, 1990. Blood serum selenium in the province of merida, venezuela related to sex, cancer incidence and soil selenium content. J. Trace Element Electrolytes Health Dis., 4: 73-77.
PubMedDirect Link - Clark, L.C., G.F. Combs Jr., B.W. Turnbull, E.H. Slate and D.K. Chalker et al., 1996. Effects of selenium supplementation for cancer prevention in patients with carcinoma of the skin: A randomized controlled trial. J. Am. Med. Assoc., 276: 1957-1963.
CrossRefPubMedDirect Link - Combs, G.F. Jr., L.C. Clark and B.W. Turnbull, 1997. Reduction of cancer risk with oral supplement of selenium. Biomed. Environ. Sci., 10: 227-234.
PubMedDirect Link - Burk, R.F., 2001. Selenium: Recent clinical advances. Curr. Opin. Gastroenterol., 17: 162-166.
Direct Link - Combs, G.R. and L. Liu, 2001. Selenium as a Cancer Preventive Agent. In: Selenium: Its Molecular Biology and Role in Human Health, Hatfield, D.L., M.J. Berry and V.N. Gladyshev, (Eds.). 2nd Edn., Kluwer Academic Publishers, Norwell, US., ISBN: 978-0-387-33826-2, pp: 249-264.
Direct Link - Ozer, N.K., D. Boscoboinik and A. Azzi, 1995. New roles of low density lipoproteins and vitamin E in the pathogenesis of atherosclerosis. Biochem. Mol. Biol. Int., 35: 117-124.
Direct Link - Lapenna D., S. de Gioia, G. Ciofani, A. Mezzetti and S. Ucchino et al., 1998. Glutathione-related antioxidant defences in human atherosclerosis plaques. Circulation, 97: 1930-1940.
PubMedDirect Link - Neve, J., 1996. Selenium as a risk factor for cardiovascular diseases. J. Cardiovascular Risk, 3: 42-70.
PubMedDirect Link - Coppinger, R.J. and A.M. Diamond, 2001. Selenium Deficiency and Human Disease. In: Selenium: Its Molecular Biology and Role in Human Health, Hatfield, D.L. (Eds.). Kluwer Academic Publishers, Norwell, Massachusetts, ISBN: 0-7923-7335-9, pp: 219-233.
Direct Link - Aaseth, J., M. Haugen and O. Forre, 1998. Rheumatoid arthritis and metal compounds-perspectives on the role of oxygen radical detoxification. Analyst, 123: 3-6.
Direct Link - Pilon-Smits, E. and M. Pilon, 2002. Phytoremediation of metals using transgenic plants. Crit. Rev. Plant. Sci., 21: 439-456.
CrossRefDirect Link - Mudgal, V., N. Madaan and A. Mudgal, 2010. Heavy metals in plants: Phytoremediation: Plants used to remediate heavy metal pollution. Agric. Biol. J. N. Am., 1: 40-46.
Direct Link - Madaan, N. and V. Mudgal, 2009. Differential tolerance behaviour of safflower accessions to some heavy metals. Int. J. Applied Environ. Sci., 4: 413-420.
Direct Link








