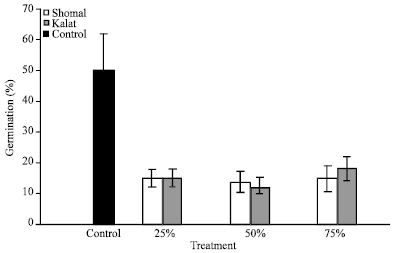
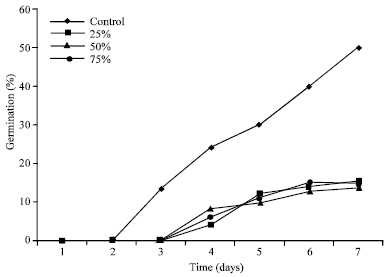
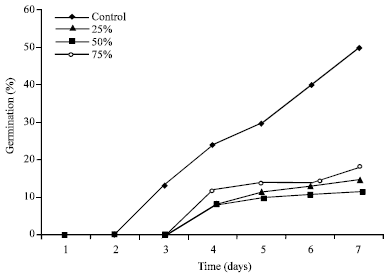
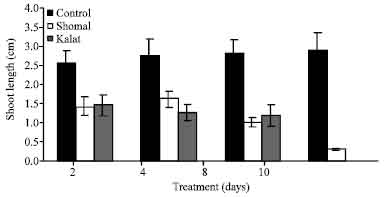
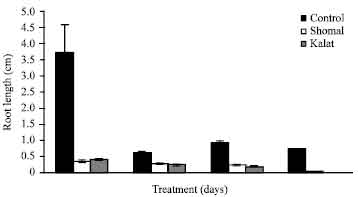
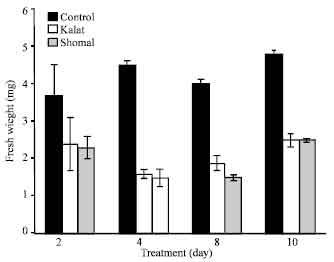
Research Article
Allelopathic Effects of Root Exudate and Leaching of Rice Seedlings on Hedgemustard (Sisybrium officinale)
Department of Biology, Faculty of Sciences, Islamic Azad University, Mashad Branch, Iran
Forugh Abbasi
Department of Biology, Faculty of Sciences, Islamic Azad University, Mashad Branch, Iran
Yalda Ghotbzadeh
Department of Biology, Faculty of Sciences, Islamic Azad University, Mashad Branch, Iran