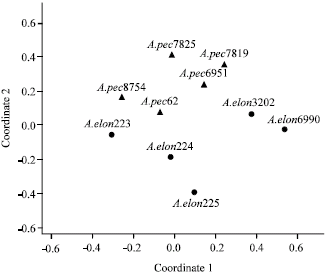
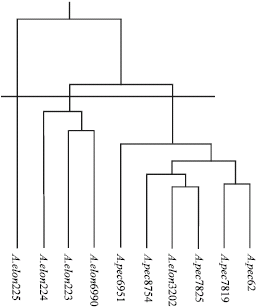
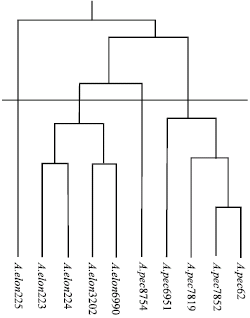
Research Article
Genetic Diversity in Ecotypes of Two Agropyron Species using RAPD Markers
Department of Agronomy and Plant Breeding, Faculty of Agriculture, University of Mohaghegh Ardabili, Ardabil, Iran
A. Asghari
Department of Agronomy and Plant Breeding, Faculty of Agriculture, University of Mohaghegh Ardabili, Ardabil, Iran
M. Shokrpour
Department of Agronomy and Plant Breeding, Faculty of Agriculture, University of Mohaghegh Ardabili, Ardabil, Iran
Mohammaddost Chmanabad
Department of Agronomy and Plant Breeding, Faculty of Agriculture, University of Mohaghegh Ardabili, Ardabil, Iran