ABSTRACT
Specific aims of the study were to quantify heavy metal concentration in the coral tissue and to determine the toxic effect of metal on coral Galaxea fascicularis. The concentration of heavy metals in the coral tissues were assessed using Atomic Absorption Spectrophotometer (AAS) technique. Various oceanographic parameters were also measured on sampling sites. Controlled tolerance experiment testing copper were performed on coral organism. Series of exposures at different range concentrations (0.025, 0.050, 0.075 and 0.100 mg L-1 Cu) were conducted for 96 h LC50. Results indicated that low variation existed among some oceanographic parameter in depth. Higher concentrations of Pb and Fe were detected in coral tissues. Short duration (24 h) laboratory assay demonstrated dramatic effects ( tissue bleaching and death) on coral at copper concentrations (0.1 mg L-1 Cu). The LC50-96 was determined to be 0.032 mg L-1 Cu (II). The present experimental results demonstrated that heavy metals can have deleterious effect on coral animal, at relatively low concentrations and for brief exposures.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/rjes.2009.96.102
URL: https://scialert.net/abstract/?doi=rjes.2009.96.102
INTRODUCTION
Coral reefs are the most diverse and important communities in the tropical and subtropical ecosystems on marine environments, providing an essential source of food, tourist income, coastal protection from storm and erosion and source for bioactive metabolite compounds (Kushmaro et al., 1997; Ben-Haim and Rosenberg, 2004; Jensen et al., 2005). About 85,707 km2 or 14% of total corals in the world extending all the way in the Indonesian sea (Tomascik et al., 1997), however, only about 6% of coral reefs in Indonesia are in excellent condition (75 to 100% coral cover). About 70% of the reefs in Indonesia are in poor to fair condition, due primarily to anthropogenic activities (Suharsono, 1998).
Java is by far the most populous island in Indonesia, with approximately 62% of the country`s population and is the most populous island in the world (Anonymous, 2001). Several big cities, Jakarta (capital city), Semarang and Surabaya, are located on the northern coast of Java island. The cities are really highly urbanized, due to the many industries located in those cities. Recent investigations have shown metal contamination in Java coastal waters as a result of industrialization and human development in the coastal zone (Booij et al., 2001; Takarina et al., 2004). As a consequence, most aquatic organisms, including coral reefs, are exposed to high concentrations of metal contaminants.
Heavy metals are well known marine pollutants that originate from such sources as industrial discharges (Gonzalez et al., 1999), urban/agricultural run-off (Guzman and Jimenez, 1992), sewage treatment discharges (Naoum et al., 2001; Stylianou et al., 2007) and anti-fouling paints (Valkirs et al., 2003; Warnken et al., 2004). Extensive research has been directed towards determining the extent and effects of metal pollution in fish (Romeo et al., 1999; Rashed, 2001), sea anemone (Harland and Nganro, 1990; Mitchelmore et al., 2003), mollusks ( Yasoshima et al., 2001; Wang et al., 2005), oysters (Hunter et al., 1995) and mussels (Wong et al., 2000). However, in the tropical marine environment, heavy metals have received little attention with respect to their evidence of metal accumulation and potential toxic effect on reef-building corals (Guzman and Jimenez, 1992). Several studies on metal concentration in corals have been reported by Bastidas and GarcÃa (1999 and 2004), Esslemont et al. (2000) and Mitchelmore et al. (2007), however, no reports was found on the study of heavy metal concentration and their toxic effects on coral Galaxea fascicularis. To our knowledge, this is the first study of heavy metals on coral G. fascicularis, in particular, from Indonesian tropical reef environments.
In the present study the bleaching response is evaluated as a means of assessing stress in corals within the framework of a standardized laboratory-based bioassay. Coral bleaching has been suggested as a potential physiological response which can be used to assess stress (Glynn, 1993). The heavy metal copper was chosen for the tests as it is a common marine pollutant. In addition, Cu is an essential element required by organisms in trace amounts, however, at higher concentrations these heavy metals can exert toxic effects on marine organisms (Kim et al., 2008). Coral G. fascicularis, was chosen as material test due to the most abundant coral species and their resistant properties to high sedimentation in sampling sites. The objectives of the study were to quantify heavy metal concentration (Copper, Lead, Zinc, Cadmium, Chromium and Iron) in the coral tissues of G. fascicularis from the Jepara coastal waters of Java and to determine the toxic effects (LC50-96) of metal on coral organism.
MATERIALS AND METHODS
Sample Collection
Sampling sites were located on Jepara coastal waters (S 06034` 44.1", E 1100 37` 47.4"), Java Sea. Figure 1 shows the sampling sites. Specimens of the corals for this analysis were collected randomly during rainy season of 2008 (September) by scuba diving at depths of 2 to 3 m, broken away with chisel and hammer and placed in plastic bag submerged in sea water. Upon collection coral fragments were put into sterile plastic bags (Whirl-Pak, Nasco, USA) and immediately brought to the laboratory with dry-ice. In laboratory, corals were dried in room temperature, ground and sieved. Fraction of particles less than 2 mm size was used for chemical analysis. Oceanographic parameters such as temperature, salinity, turbidity, conductivity, pH and dissolved oxygen concentration were measured by using Water Quality Checker, produced by Horiba Co. Ltd, Japan. Wave Recorder produced by Sountex, USA was used to measure current speed and orientation.
 | |
| Fig. 1: | Sampling site of Jepara coastal waters, Java Sea |
Metal Analysis
Coral G. fascicularis was collected and identified according to Veron (1986). The collected specimens were cleaned in running water to remove organic materials, dried in the room temperature and powdered, then 1 g of each sample was digested a mixture of HF, HNO3 and HClO4 acids (Chester et al., 1994). After the complete digestion, each sample was diluted to 50 mL and the trace metals were determined as μg g-1 using AAS technique (PE-3110). The measurements accuracy was checked by applying two replicates in each sample.
Copper Toxicity Test and 96 h LC50
Corals were placed in experimental aquariums 1 day prior to the start of the experiment. Dilutions of the copper were prepared with filtered sea water to give final concentrations of Cu (II) of 10, 1,0, 0.1, 0.01 and 0.001 mg L-1. Ten small fragments (2x2 cm) of G. fascicularis were placed in 7.5 L of each of the solutions for 48 h. All test solutions and control were aerated throughout the experimental period. Coral mortality was investigated visually on percentage bleaching. Based on the results obtained in the range-finding experiment of copper tolerance, groups of ten small G. fascicularis colonies were exposed to copper concentrations in sea water of 0, 0.025, 0.050, 0.075 and 0.10 mg L-1 Cu were conducted to determine 96 h median lethal concentrations (LC50) graphically. The corals were inspected regularly and their condition recorded. All test solutions and control were also aerated throughout the experimental period. When polyp tissue could no longer be seen within the calices, the corals were considered dead.
RESULTS AND DISCUSSION
Oceanographic Parameters and Heavy Metal Analysis
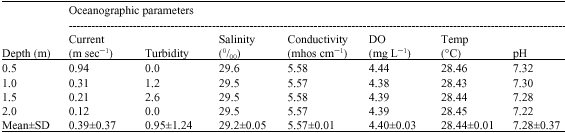
The physico-chemical variables of the present study areas are subjected to wide spatial temporal variation. Rainfall is the most important cyclic phenomenon in Jepara as it brings about important changes in the physical and chemical characteristics of the Jepara coastal zone. During the study Jepara coastal water temperature was 28.44°C. Salinity range was 29.52 0/00. The pH value of water sample was 7.28. Dissolved Oxygen (DO) concentration was varied from 4.38 to 4.44 ml L-1. The current value was varied from 0.12 to 0.94 m sec-1 in depth. Results showed that oceanographic parameters such as pH, temperature, DO, salinity and conductivity were similar with increasing depth (Table 1).
| Table 1: | Oceanographic parameters of sampling site |
 | |
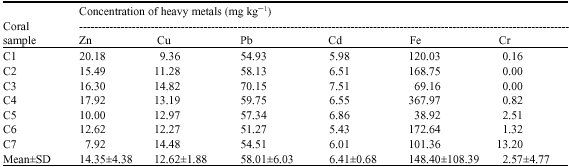
| Table 2: | Heavy metal concentrations in coral tissues |
 | |
Comparing with earlier study (Takarina et al., 2004) shows that Jepara coastal waters had high DO and low conductivity. Water quality parameters, such as DO, temperature, pH, were known to influence the availability and accumulation of metal by marine organisms (Shuhaimi-Othman et al., 2006; Gorski and Nugegoda, 2006). Furthermore, the toxicity and bioavailability to marine organisms is greatly influenced by physico-chemical condition in which the heavy metals is present (Gorski and Nugegoda, 2006).
There is variation and high concentrations of metals were determined for the coral tissue. Except for the Chrom (Cr) on sample C2 and C3 all other trace metals were detectable. Result for the mean of metals in the coral tissues were Zn 14.35±4.38 mg kg-1, Cu 12.62±1.88 mg kg-1, Pb 58.01±6.03 mg kg-1, Cd 6.41±0.68 mg kg-1, Fe 148.40±108.39 mg kg-1 and Cr 2.57±4.77 mg kg-1. Result also showed that Fe and Pb concentrations in coral were very high (Table 2). The causes of higher concentration of heavy metals in corals is likely to be restricted to the areas immediately surrounding the site of release, such as locations of industrial discharge, sewage outfalls and urban/agricultural runoff. Jepara coastal waters was surrounded by harbour, shipyard, high density of coastal settlements, wood industries and high intensity of agricultural activities. These heavy metals may also be derived from some of these anthropogenic heavy metals which found in Jepara nearshore waters. Corals absorb metals across their surface`s tissue, then metals are suspended or floating in the surrounding water will be absorbed.
These higher values in corals are not surprisingly, Brown (2000) suggested that tropical cnidarian species could increase their sensitivity to pollutants compared to their temperate counterparts. Furthermore, Barka (2007) stated that marine invertebrates accumulate trace metals in their tissues at levels several fold higher than those in their surroundings and are still able to survive.
Copper Toxicity Test and 96 h LC50
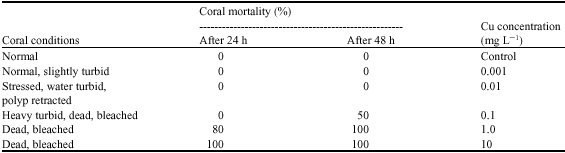
In the range-finding experiment, fragments of G. fascicularis from assay exposed to 10 mg L-1 copper turned from a normal dark green colour to a lighter colour within 5 h and began to discolour after 7 h. Between 13 and 15 h, all fragments exposed to 10 mg L-1 copper died and 8 of the 10 fragments in the 1 mg L-1 copper treatment died. The discolouration appeared to be less extensive on the fragments exposed to 0.1 mg L-1 Cu and 5 of the 10 fragments died after 48 h. No mortalities were observed at any test concentration. Result of range-finding tolerance of G. fascicularis to copper concentrations was 0.01 to 0.1 mg L-1 . The condition of the test corals after 48 h exposure is given in Table 3. Based on the results obtained in the initial–finding study, a second series of exposures at different range concentrations were conducted for 96 h LC50. Fifty percent coral mortality figure was achieved for G. fascicularis colonies at copper concentrations of 0.025, 0.05, 0.075 and 0.10 mg L-1 Cu. Figure 2 plots the time taken for 50% of colonies to die at each experimental copper concentration and results a 96h LC50 of 0.032 mg L-1.
| Table 3: | Results of range-finding toxicity of coral G. fascicularis to copper after 48 h |
 | |
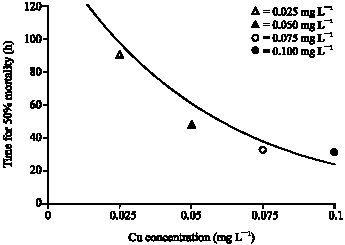 | |
| Fig. 2: | Time for 50% mortality of G. fascicularis at various Cu concentrations |
Bleaching of corals following exposure to heavy metals has been described in the earlier studies. Howard et al. (1986) reported a 96h LC50 for Montipora verucosa exposed to 0.048 mg L-1 Cu. While Jones (1997) demonstrated the loss of zooxanthellae from Acropora formosa exposed at 0.01 to 0.04 mg L-1 Cu for 48 h. Mitchelmore et al. (2007) in their study showed 0.05 mg L-1 Cu exposure caused severe stress for P. damicornis. It seems that acute toxicity for coral exposed to copper depend on the metal, species and life-stage. The present study showed that result a 96 h LC50 for G. fascicularis was 0.032 mg L-1 Cu. No measurements were made of the uptake and partitioning of copper during the toxicity tests described here. However, McConchie and Harriott (1992) stated that coral tissue parts had more potential use in metal pollution monitoring than that of coral skeletal parts. Even the effect of copper was only examined on G. fascicularis, the study has indicated that measuring coral bleaching has considerable potential as a means of assessing stress.
CONCLUSION
Variations of Zn, Cd, Cu, Fe, Pb and Cr concentrations in tissues of the scleractinian coral G. fascicularis were measured to provide information for designing bio-assay surveys involving this coral. Inter-colony variations were measured using single portions (tissue) from different colonies of a coral community. The application of tissues for heavy-metal bio-assays is compromised by consistent inter-colony variation. Comparisons with earlier studies show that G. fascicularis to be the more sensitive to heavy metal toxicity, however this organism could survive in higher concentrations for longer periods of time than the other coral species. The finding of this study suggest that the metal concentrations in coral tissue might be sensitive to differences in environmental metal load. Future study will need to establish study for quantitatively assessing both the acute and chronic toxic influence of heavy metals on coral physiology from polluted and unpolluted sampling sites.
ACKNOWLEDGMENT
This study was supported by grant from Directorate General of Higher Education (Dikti), Indonesian Ministry of National Education under competent research grant scheme (HIBAH KOMPETENSI, No: 013/HIKOM/DP2M/2008).
REFERENCES
- Barka, S., 2007. Insoluble detoxification of trace metals in a marine copepod Tigriopus brevicornis (Muller) exposed to copper, zinc, nickel, cadmium, silver and mercuri. Ecotoxicology, 16: 491-502.
CrossRef - Bastidas, C. and E. García, 1999. Metal content on the reef coral Porites astreoides: An evaluation of river influence and 35 years of chronology. Mar. Pollut. Bull., 38: 899-907.
CrossRef - Bastidas, C. and E.M. García, 2004. Sublethal effects of mercury and its distribution in the coral Porites astreoides. Mar. Ecol. Prog. Ser., 267: 133-143.
Direct Link - Booij, K., M.T. Hillebrand, R.F. Nolting and J. Van Ooijen, 2001. Nutrients, trace metals and organic contaminants in Banten Bay, Indonesia. Mar. Pollut. Bull., 42: 1187-1190.
Direct Link - Brown, E.B., 2000. The significance of pollution in eliciting the “bleaching” response in symbiotic cnidarians Int. J. Environ. Pollut., 13: 392-415.
CrossRefDirect Link - Chester, R., F.G. Lin and A.S. Basaham, 1994. Trace metal solid state speciation changes associated with the down-column fluxes of oceanic particulates. J. Geol. Soc., 151: 351-360.
CrossRefDirect Link - Esslemont, G., V.S. Harriott and D.M. McConchie, 2000. Variability of trace-metal concentration within and between colonies of P. damicornis. Mar. Pollut. Bull., 40: 637-642.
CrossRef - Glynn, P.W., 1993. Coral reef bleaching: Ecological perspectives. Coral reef bleaching: Ecological perspectives. Coral Reefs 12: 1-17.
CrossRef - Gonzalez, H., M. Pomares, M. Ramirez and I. Torres, 1999. Heavy metals in organisms and sediments from the discharge zone of the submarine sewage outfall of Havana City, Cuba. Mar. Pollut. Bull., 38: 1048-1051.
CrossRef - Gorski, J. and D. Nugegoda, 2006. Sublethal toxicity of trace metals to larvae of the blacklip abalone, Haliotis rubra. Environ. Toxicol. Chem., 25: 1360-1367.
CrossRef - Guzman, H.M. and C.E. Jimenez, 1992. Contamination of coral reefs by heavy metals along the Caribbean coast of central America (Costa Rica and Panama). Mar. Pollut. Bull., 24: 554-561.
CrossRef - Harland, A.D. and N.R. Nganro, 1990. Copper uptake by the sea anemone Anemonia viridis and the role of zooxanthellae in metal regulation. Mar. Biol., 104: 297-301.
CrossRef - Hunter, C.L., M.D. Stephenson, R.S. Tjeerdema, D.G. Crosby and G.S. Ichikawa et al., 1995. Contaminants in oysters in Kaneohe Bay, Hawaii. Mar. Pollut. Bull., 30: 646-654.
CrossRef - Jones, R.J., 1997. Zooxanthellae loss as a bioassay for assessing stress in corals. Mar. Ecol. Prog. Ser., 149: 163-171.
Direct Link - Kim, B.E., T. Nevitt and D.J. Thiele, 2008. Mechanisms for copper acquisition, distribution and regulation. Nature Chem. Biol., 4: 176-185.
CrossRef - Kushmaro, A., E. Rosenberg, M. Fine and Y. Loya, 1997. Bleaching of the coral Oculina patagonica by Vibrio AK-1. Mar. Ecol. Prog. Ser., 147: 159-165.
Direct Link - Mitchelmore, C.L., A.E. Verde, A.H. Ringwood and V.M. Weis, 2003. Differential accumulation of heavy metals in the sea anemone Anthopleura elegantissima as a function of symbiotic state. Aquat. Toxicol., 64: 317-329.
CrossRef - Naoum, C., D. Fatta, K.J. Haralambous and M. Loizidou, 2001. Removal of heavy metals from sewage sludge by acid treatment. J. Environ. Sci. Health, 36: 873-881.
CrossRef - Rashed, M.N., 2001. Monitoring of environmental heavy metals in fish from Nasser Lake. Environ. Int., 27: 27-33.
CrossRefPubMedDirect Link - Romeo, M., Y. Siau, Z. Sidoumou and M. Gnassia-Barellia, 1999. Heavy metal distribution in different fish species from the Mauritania coast. Sci. Total Environ., 232: 169-175.
CrossRefDirect Link - Shuhaimi-Othman, M., D. Pascoe, U. Borgmann and W.P. Noorwood, 2006. Reduced metals concentrations of water, sediment and Hyallela azteca from lakes in the vicinity of Sudbury metal smelters, Ontario, Canada. Environ. Monit. Assess., 117: 27-44.
CrossRef - Stylianou, M.A., D. Kollia, K.J. Haralambous, V.J. Inglezakis, K.G. Moustakas and M.D. Loizidou, 2007. Effect of acid treatment on the removal of heavy metals from sewage sludge. Desalination, 215: 73-81.
CrossRefDirect Link - Takarina, N.D., D.R. Browne and M.J. Risk, 2004. Speciation of heavy metals in coastal sediments of Semarang, Indonesia. Mar. Pollut. Bull., 49: 861-868.
CrossRefDirect Link - Valkirs, A.O., P.F. Seligman, E. Haslbeck and J.S. Caso, 2003. Measurement of copper release rates from antifouling paint under laboratory and in situ conditions: Implications for loading estimation to marine water bodies. Mar. Pollut. Bull., 46: 763-779.
CrossRef - Wang, Y., L. Liang, J. Shi and G. Jiang, 2005. Study on the contamination of heavy metals and their correlations in mollusks collected from coastal sites along the Chinese. Bohai Sea, 31: 1103-1113.
CrossRef - Warnken, J., R.J.K. Dunn and P.R. Teasdale, 2004. Investigation of recreational boats as a source of copper at anchorage sites using time-integrated diffusive gradients in thin film and sediments measurements. Mar. Pollut. Bull., 49: 833-843.
CrossRef - Wong, C.K.C., R.Y.H. Cheung and M.H. Wong, 2000. Heavy metal concentrations in green-lipped mussels collected from Tolo Harbour and markets in Hong Kong and Shenzhen. Environ. Pollut., 109: 165-171.
CrossRefDirect Link - Yasoshima, M., M. Matsuo, A. Kuno and B. Takano, 2001. Studies on intake of heavy metals by Bradybaena similaris, land snails, by XAFS measurement. J. Synchrotron Rad., 8: 969-971.
CrossRef








