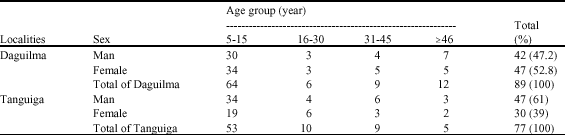Research Article
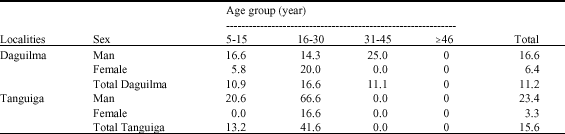
Importance of Malacological Factors in the Transmission of Schistosoma haematobium in Two Dams in the Province of Oubritenga (Burkina Faso)
Institut de Recherche en Sciences de la Sante (Irss), 03 BP 7047, Ouagadougou 03, Burkina Faso
B.G. Kabre
Universite de Ouagadougou, Burkina Faso
D. Dianou
Institut de Recherche en Sciences de la Sante (Irss), 03 BP 7047, Ouagadougou 03, Burkina Faso
B. Savadogo
Institut de Recherche en Sciences de la Sante (Irss), 03 BP 7047, Ouagadougou 03, Burkina Faso
J.N. Poda
Institut de Recherche en Sciences de la Sante (Irss), 03 BP 7047, Ouagadougou 03, Burkina Faso