Research Article
Seed Bank Dynamics in Altitudinal Gradient on an Inselberg in a Nigerian Secondary Forest
Department of Botany, Obafemi Awolowo University, Ile-Ife, Nigeria
S.O. Oke
Department of Botany, Obafemi Awolowo University, Ile-Ife, Nigeria
Environmental factors such as altitude and topography have been recognized as key factors in the structure, function and floristic composition of vegetation in the tropics. Altitude-related changes have been detected in the floristic composition and species richness of vegetation. Seed bank density and species richness have been reported to change along temporal (succession) and spatial (altitude, latitude) gradients (Thompson, 1978; Warr et al., 1993). For example Thompson (1978, 1985) showed that seed bank density decreases with altitude. This is because at high altitude, there is a predominance of slow-growing and long-lived species and environmental conditions (short growing season) are less favourable to seed production (Archibold, 1984; Thompson, 1992). These ideas were reinforced by the results of Ortega et al. (1997) who found that richness and density of seed bank decreased with altitude in Spanish mountain grasslands. At low altitudes, richness is greater and annual species dominate, while at higher altitudes, richness diminishes and perennial dominates (Montalvo et al., 1991a, b). On a local scale, topography cause a water-stress gradient and influences the size of seed banks (Ortega et al., 1997). If the dominant species at high altitude do not form a persistent seed bank, the similarity between the composition of established vegetation and seed bank should decrease with altitude (Peco et al., 1998).
However, an altitude hypothesis has been proposed (Funes et al., 2003). In cold climate (e.g., high mountains or high altitudes) several factors would contribute to the maintenance of many seeds in the soil (Archibold, 1984; Cavieres and Arroyo, 2001). The diversity of both seed predators and fungi tends to be low in high-mountain habitats (Mc Graw and Vavreck, 1989) and low temperatures are associated with low embryonic metabolic rates and show consumption of seed reserves, favouring seed longevity (Murdoch and Ellis, 2000) and thus the formation of persistent seed bank remain in the soil for long periods (Thompson et al., 1997). This is high carry-over of soil seeds from year to year should increase the total seed bank density toward higher altitudes. If this assumption is correct, seed bank density should increase with altitude. Thompson and Grime (1979) suggested that seed bank strategy is independent from the environment. However, Ortega et al. (1997) and Funes et al. (2001) reported different populations of the same species, in relation to changing micro-environmental conditions at the local scale. Furthermore, a temporal pattern in the number of germinable seeds in the soil has been detected with a minimum in spring (Young et al., 1981). Predator mainly takes place in the summer coinciding with the period of maximum insect activity, most germination occurs in the autumn after the first effective rains (Espigares and Peco, 1993).
The aim of this study is to examine the variation in richness and density of soil seed banks and the relationship between the composition of the established vegetation and seed banks at different altitudes on an inselberg in Ile-Ife area of Southwestern Nigeria.
Study Area
The study was carried out on an inselberg in a secondary rain forest in the Biological Gardens of the Obafemi Awolowo University, Ile-Ife (latitude 7°30’ to 7°35’ N, longtitude 4° 0’ to 4°35’ E) southwestern Nigeria. The elevation ranges from 286 to 461 m a.s.l. (Hall, 1969). The forest is a natural regrowth under conservation within and outside the Gardens with minimal human disturbance. The age of the forest cannot be accurately determined since the time of last major disturbance is not known but it is estimated to be about 59 years old (Muoghalu and Okesan, 2005).
The Ife area lies in dry deciduous forest zone (Onochie, 1979). White (1983) described the vegetation as Guineo-Congolian dried forest type. There are two seasons in the area, the rainy season (March to November) and the dry season (November to March). The annual rainfall averaged 1413 mm per year in a 5 year survey (Duncan, 1974) and showed two peaks, one in July and the other in September. The mean annual temperature ranges from 22.5 to 31.4°C.
The area is underlain by rocks of the Basement complex, which are the Precambrain age (De Swardt, 1953). The forest covers the base and lower slope and grassland and woodland cover the upper slope and peak of the inselberg underlain by granite gnesiss. Soil has been classified as Lixisols (FAO/UNESCO, 1974) and Ultisols (USDA, 1975). Soils are usually acidic and contain <10% clay which is kaolinite and hence are characterized by low cation exchange capacity (Ayodele, 1986).
Data Collection
Three sample plots designated A, B and C, each 25x25 m, were established at 80 m intervals along an altitudinal gradient from the base 290 m a.s.l. to the peak 461 m a.s.l. of the inselberg. Within each plot all shrubs, trees and herbaceous species were identified to species level and enumerated. Species nomenclature follows those of Hutchinson and Dalziel (1954-1972). In each of the three plots, twenty replicate soil samples were randomly collected to a depth of 15 cm using a soil auger of diameter 8.5 cm, taking into account that most viable seeds are located within the first few cm (Funes et al., 2001). The soil samples were collected in March 2004 for the dry season and October 2004 for the rainy season in order to capture both the transient seed bank of the present year and persistent fraction of the seed bank. The samples were put in polythene bags, labeled and were then transferred to the laboratory where they were spread on tables to dry. The soil samples were transferred after drying into porous plates and placed in the screen house in April 2004 (dry season collection) and November 2004 (rainy season collection) where they were watered daily and monitored for seedling emergence. There were ten plates for each plot for each for each season making a total of sixty plates per for the two seasons. Plates were kept in the screen house for 6 months and whenever possible seedlings were identified at an early stage and removed from the plates. When flowers and or fruits were required for correct identification, seedlings were transplanted into separate plots and grown on natural unfertilized soil within the same screen house. Most of the seedlings were identified to species level. Once in a while, the soil in each plate was turned over and mixed so as to aid seed germination. The germination method is considered the most appropriate for studying the composition of species in the soil seed bank, particularly in natural systems with high floristics richness (Gross, 1990).
The total number of individuals and species in each plate for each plot for both dry and rainy seasons collection was determined. The percentage contribution of each species to the seed bank were also determined seasonally. Sorenson index of similarity was also used to compare the similarity in species composition among the plots in each sampling gradient. To assess the contribution of the seed bank to the various forest plant community, the occurring species in seed bank and the established vegetation were compared.
Standing Vegetation Composition
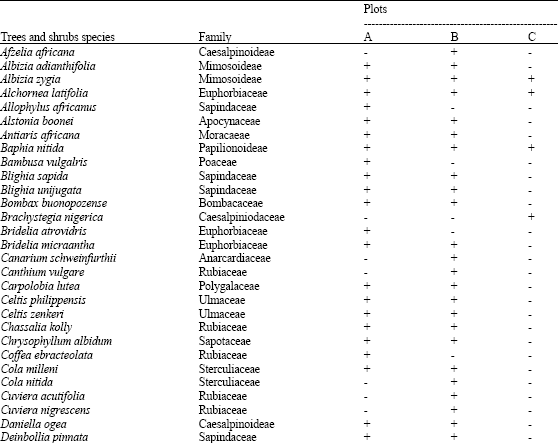
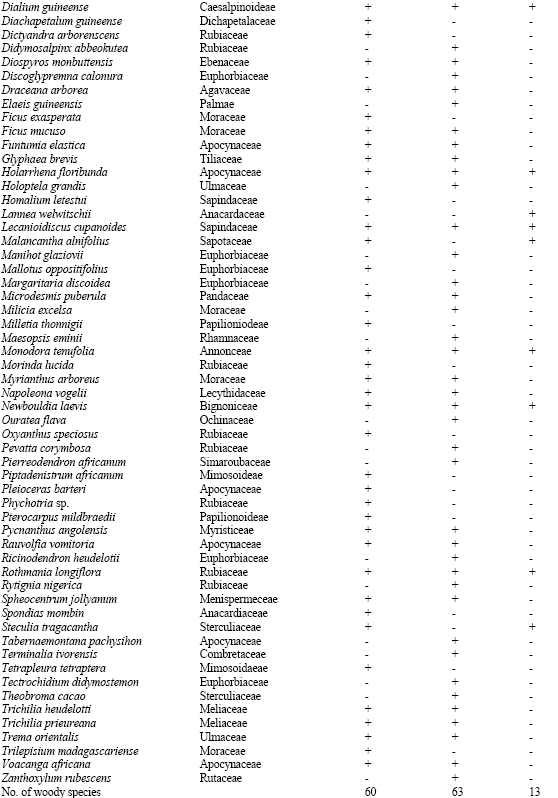
There were 126 plant species encountered in 117 genera and 49 families while 5 plant species were unidentified in the vegetation at all the three altitudes (plots). The plant species in these three study plots consist of a total of 86 woody species (68.5%), 5 grass species (3.9%), 11 forbs (8.7%) and 24 climber species (18.8%) (Table 1). The common woody species in the standing vegetation at all the three altitudes are: Albizia zygia, Alchornea latifolia, Baphia nitida, Dialium guineensis, Holarrhena floribundia, Lecaniodiscus cupanoides, Monodora tenufolia, Newbouldia laevis and Rothmania longiflora while no grass, forb and climber species was common in the vegetation at the three altitudes (plots).
| Table 1: | Plant species composition of the study plots along altitudinal gradient on an inselberg in Ile-Ife area of Southwestern Nigeria |
 | |
 | |
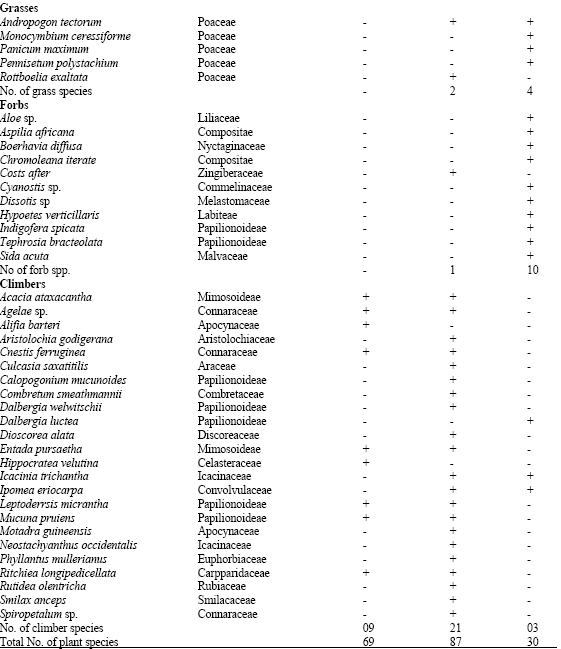 | |
| + = Species present, - = Species absent | |
Species Richness and Density of Soil Seed Bank
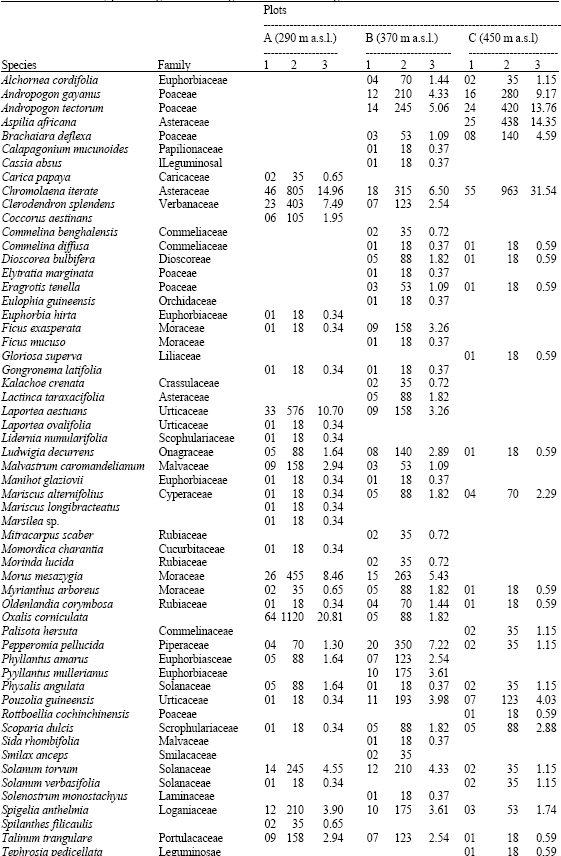
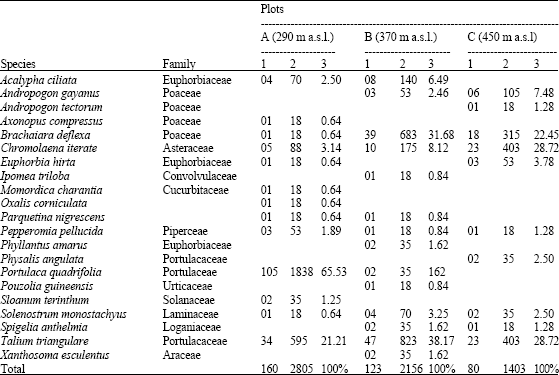
In total 71 species belonging to 27 families germinated from the soil samples. The dominant plant families were Poacea (7 species), Euphorbiacea (6 species), Asteracea and Moracea (5 species each). The number of species found per sample was higher in the dry season soil collection than rainy season soil collection. The species richness of the seed bank changed significantly with altitude both in dry and rainy season (p<0.05). In the dry season soil collection the lower altitudes showed significantly more species than the other higher altitudes (Table 2). In the rainy season soil it follows the same trend but less gradual (Table 3) Soil seed bank density decreased with altitude both in the dry and rainy season soil collections (Table 2 and 3), although, this difference was significant (p<0.05) only in the dry season soil collection.
| Table 2: | Density of species (seedlings (cm-2) and seeds (m-2)) that emerged from the dry season soil collection in the three study plots along an altitudinal gradient on an inselberg |
 | |
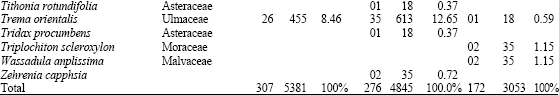 | |
| 1= Seedling (cm-2), 2 = Seedling (m-2), 3 = SB (%) | |
| Table 3: | Density of species (seedlings (cm-2) and seeds (m-2)) that emerged from the rainy season soil collection in the three study plots along an altitudinal gradient on an inselberg |
 | |
| 1= Seedling (cm-2), 2 = Seedling (m-2), 3 = SB (%) | |
Furthermore, seed density was significantly greater in the dry season than in rainy season (p<0.05). As altitude increased, the number of seed m-2 of the dry season seed banks dropped from 5381 to 3053 (Table 2) and in the rainy season, this number fell from 2805 to 1403 (Table 3).
Established Vegetation and Soil Seed Bank
At all altitudes, both on the dry and rainy seasons, the Sorensen similarities between the established vegetation and the soil seed bank were low. One hundred and six species present in the vegetation were absent from the seed banks at all the three altitudes. Moreover, 61 species occurring in the seed bank were absent in the established vegetation. Only 10 species were found both in established vegetation and in the seed bank and these include 6 trees/shrubs, 2 forbs, 1 grass and 1 climber.
The decrease in seed density in the dry and rainy seasons germinable seed banks as altitude increases seems to be related to the change of species communities determined by climatic variation. The low temperatures at the high end of the gradient cause a shortening of the reproductive period (Ortega et al., 1993). These species probably reduce their seed production at the expense of increasing their vegetative growth as a form of alternative reproduction in unfavourable situations. The negligible contribution of perennial species to seed banks has already been described by Major and Pyolt (1966) and Thompson (1985) have also observed smaller seed banks as altitude increases in British pastures. The fall in species richness (21) of the seed banks when the pasture altitude rises is also probably linked to the fall in richness found in the vegetation (Mantalvo et al., 1991b). The floristic composition of the standing vegetation in this study site falls from 69 to 30 as the altitude rises (461 m.a.s.l). The change in microclimate and fertility associated with topography gradient (Pope and Lioyd, 1975) in which the low zones have a more favourable environment for the development of vegetation may also be responsible for the decline in species and density of seed bank as altitude rises. Furthermore, the accumulation of water in the low slope zones, which can be regarded as positive factor in the low positions on the altitudinal gradient can contribute to higher species diversity and density seed bank because it can cause the accumulation of the seeds (Ortega et al., 1997). The patterns found by Ortega et al. (1997) and Thompson (1978, 1985) whereby seed bank density falls as altitude increases were repeated in the study but does not agree with the finding of Funes et al. (2003) whereby seed bank richness increased with altitude. We found a low degree of similarity between the composition of established vegetation and that of soil seed banks along the gradient. Present data confirm the results obtained in several part of the world, where a very low degree of association has been found between the composition of seed banks and that of the standing vegetation (Major and Pyott, 1966; Abrams 1988, D’ Angela et al., 1988; Bakker, 1989, Warr et al., 1993; Oke et al., 2006).
The relation between seed bank density, diversity and altitude has been proven and seasonal variation of seed numbers is environment-dependent for most species. At low altitudes, richness is greater and annual species dominate while at higher altitudes richness diminishes and perennial dominates
SANGITA GANGULI (research scholar in visva bharati) Reply
the paper was intersting and has opened a wide way in forest research.
Esemuze Lucky Reply
The paper gave a well articulated information on the spatial characteristics of vegetation distribution along altitudinal gradient. Found it very useful for my research.