Research Article
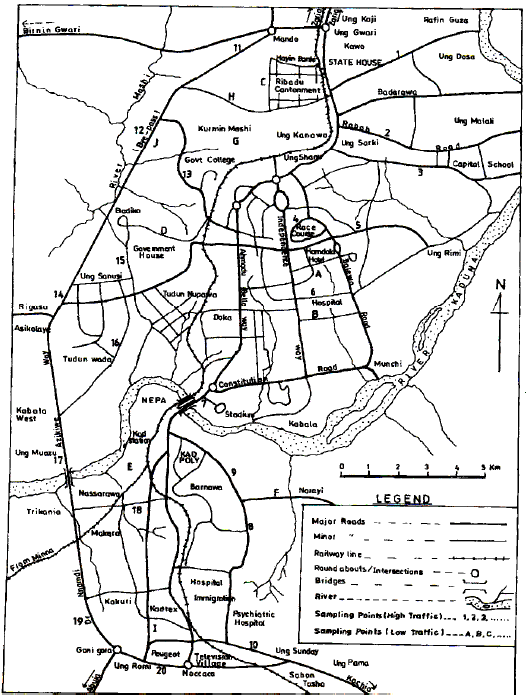
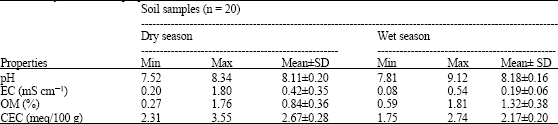
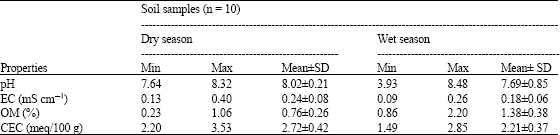
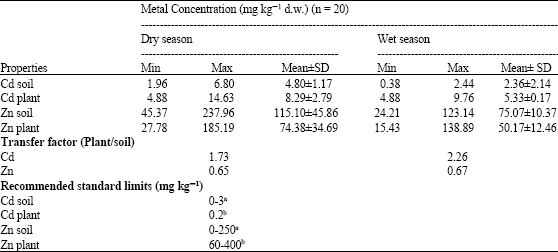
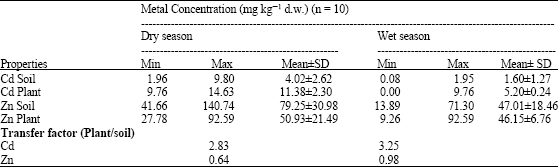
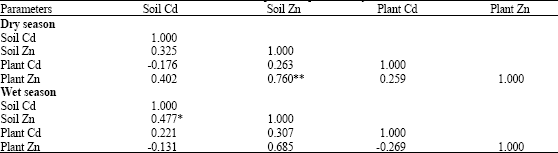
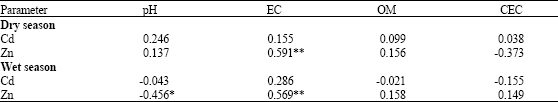
Assessment of Cd and Zn in Roadside Surface Soils and Vegetations along Some Roads of Kaduna Metropolis, Nigeria
Department of Chemistry, Ahmadu Bello University, Zaria, Nigeria
A. Uzairu
Department of Chemistry, Ahmadu Bello University, Zaria, Nigeria
G.I. Ndukwe
Department of Chemistry, Ahmadu Bello University, Zaria, Nigeria
S.G. Adewusi
Department of Chemistry, Crescent University, Abeakuta, Nigeria