Research Article
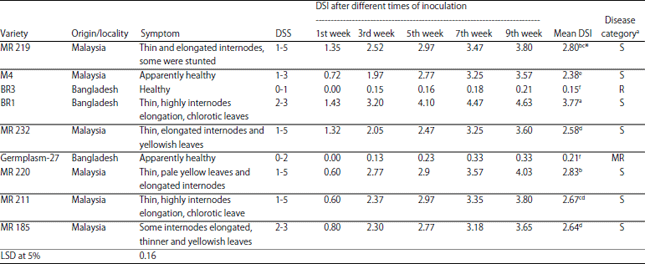
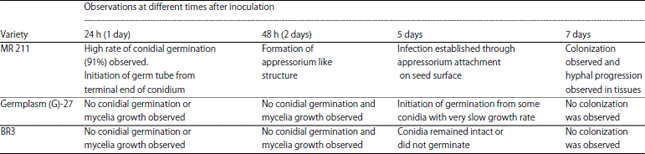
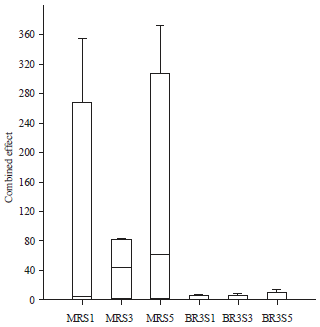
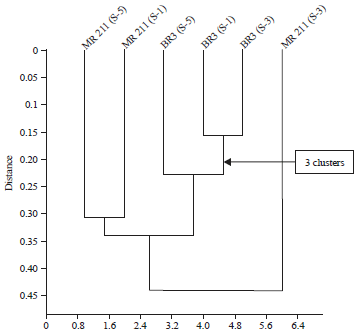
Varietal Screening and Infection Process of Fusarium proliferatum in Rice Varieties
Institute of Tropical Agriculture, Universiti Putra Malaysia, 43400 Serdang, Selangor, Malaysia
LiveDNA: 880.31808
Sariah Meon
Department of Plant Protection, Universiti Putra Malaysia, 43400 Serdang, Selangor, Malaysia
Hawa Jaafar
Department of Crop Science, Universiti Putra Malaysia, 43400 Serdang, Selangor, Malaysia
Zainal Abidin B.M. Ahmad
Department of Plant Protection, Universiti Putra Malaysia, 43400 Serdang, Selangor, Malaysia