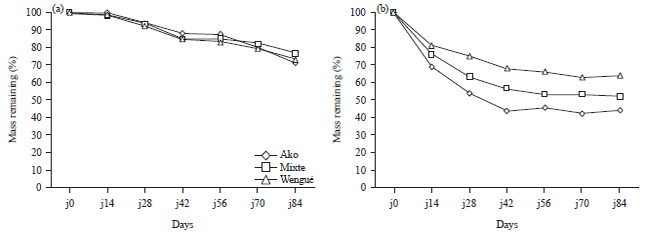
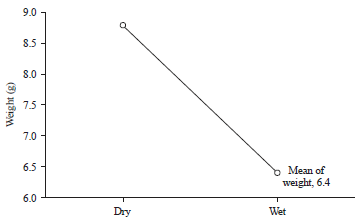
Research Article
Litter Fall, Standing Litter and Leaves Decomposition Within Urban Tropical Forest of Zoological Park of Brazzaville, Republic of the Congo
Laboratoire de Geomatique et Ecologie Tropicale Appliquee, Ecole Normale Superieure, Universite Marien N`GOUABI, BP 237 Brazzaville, Congo
LiveDNA: 682.23113
Malonga Marini Urielle
Laboratoire de Geomatique et Ecologie Tropicale Appliquee, Ecole Normale Superieure, Universite Marien N`GOUABI, BP 237 Brazzaville, Congo
Milandou Jodrhy
Laboratoire de Geomatique et Ecologie Tropicale Appliquee, Ecole Normale Superieure, Universite Marien N`GOUABI, BP 237 Brazzaville, Congo
Madingou Mady Parfait
Laboratoire de Geomatique et Ecologie Tropicale Appliquee, Ecole Normale Superieure, Universite Marien N`GOUABI, BP 237 Brazzaville, Congo
Mavoungou Naiving
Laboratoire de Geomatique et Ecologie Tropicale Appliquee, Ecole Normale Superieure, Universite Marien N`GOUABI, BP 237 Brazzaville, Congo
Stevanche Nzingoula
Laboratoire de Geomatique et Ecologie Tropicale Appliquee, Ecole Normale Superieure, Universite Marien N`GOUABI, BP 237 Brazzaville, Congo
Binsangou Stoffenne
Laboratoire de Geomatique et Ecologie Tropicale Appliquee, Ecole Normale Superieure, Universite Marien N`GOUABI, BP 237 Brazzaville, Congo