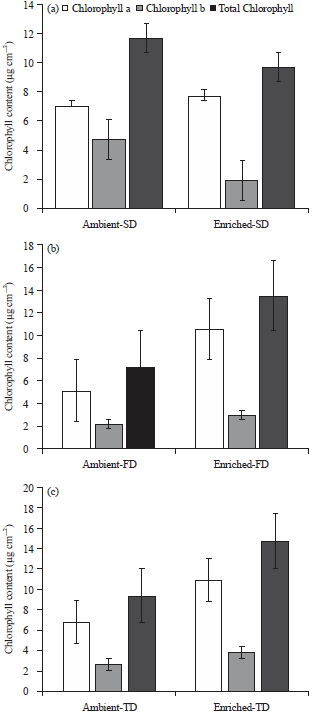
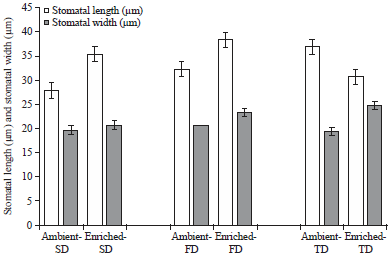
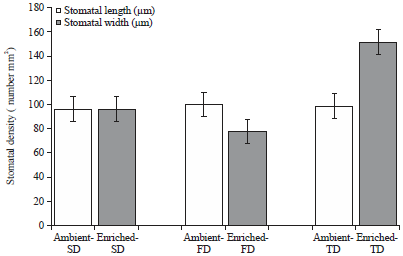
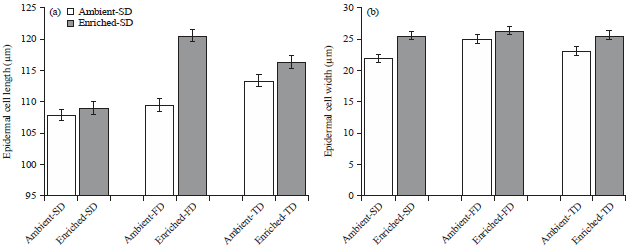
Research Article
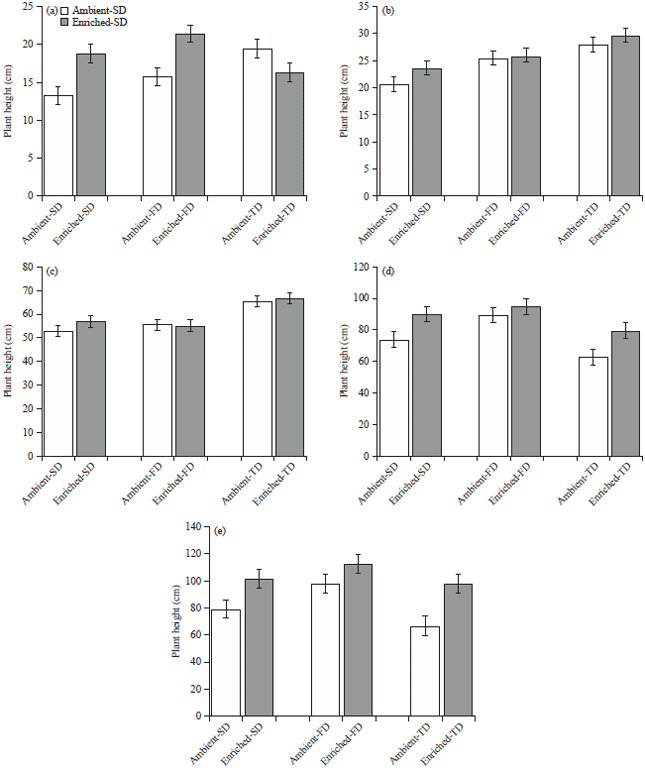
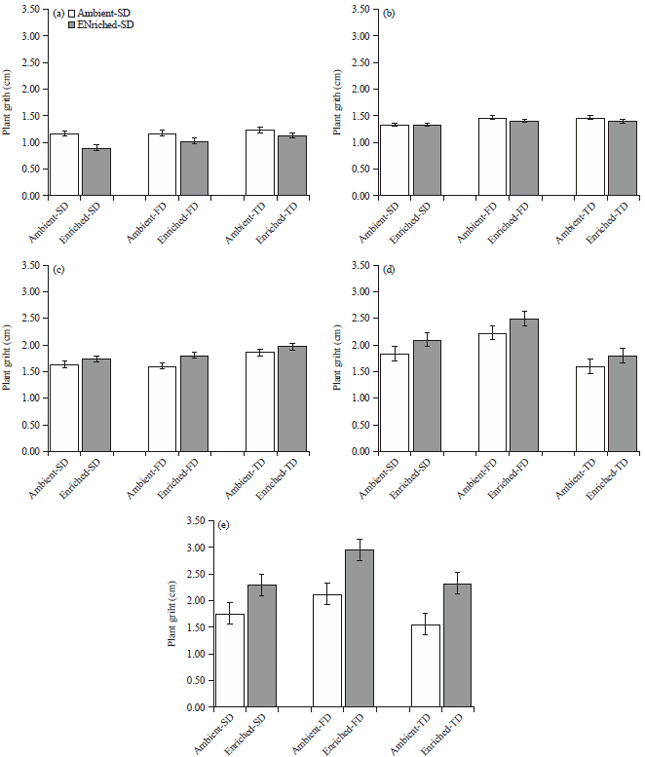
Elevated CO2 Improves Vegetative Growth of Water-stressed Zea mays L. Under Low Photosynthetic Photon Flux Density
Department of Biology, College of Science, United Arab Emirates University, Al Ain, UAE
LiveDNA: 1.19883
Shaijal Babu Thruppoyil
Department of Biology, College of Science, United Arab Emirates University, Al Ain, UAE
LiveDNA: 91.19851
Abdul Rasheed Palakkott
Department of Biology, College of Science, United Arab Emirates University, Al Ain, UAE