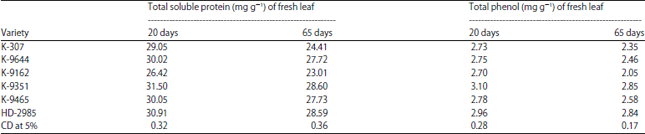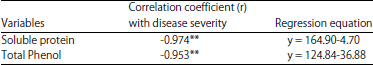Research Article
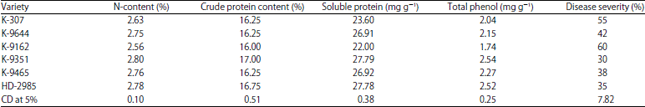
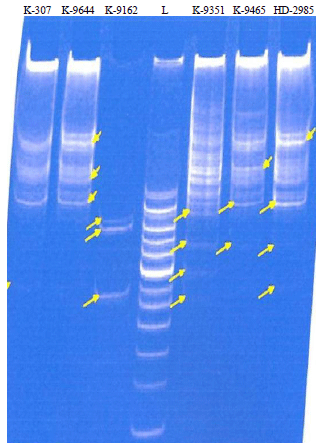
Variable Disease Response to Spot Blotch in Different Eat Varieties and it Assessment at Biochemical and Genetics Level
Department of Plant Pathology, Chandra Shekhar Azad University of Agriculture and Technology, Kanpur 208002, Uttar Pradesh, India
LiveDNA: 91.16360
Mohd. Rajik
Department of Plant Pathology, Chandra Shekhar Azad University of Agriculture and Technology, Kanpur 208002, Uttar Pradesh, India
Muneeshwar Sharma
Shere-Kashmir University of Agricultural and Sciences, Jammu and Kashmir, India
Prem Naresh
Department of Plant Pathology, Chandra Shekhar Azad University of Agriculture and Technology, Kanpur 208002, Uttar Pradesh, India
Upesh Kumar
Department of Plant Pathology, Chandra Shekhar Azad University of Agriculture and Technology, Kanpur 208002, Uttar Pradesh, India
Kishan Lal
Department of Plant Pathology, Chandra Shekhar Azad University of Agriculture and Technology, Kanpur 208002, Uttar Pradesh, India
Ravindra Singh
Department of Plant Pathology, Chandra Shekhar Azad University of Agriculture and Technology, Kanpur 208002, Uttar Pradesh, India