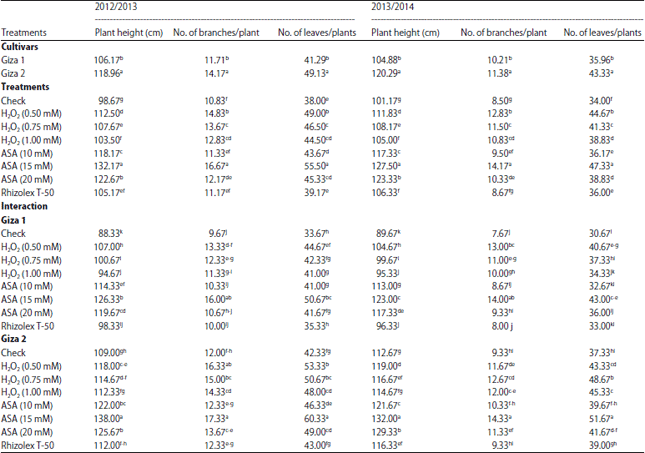
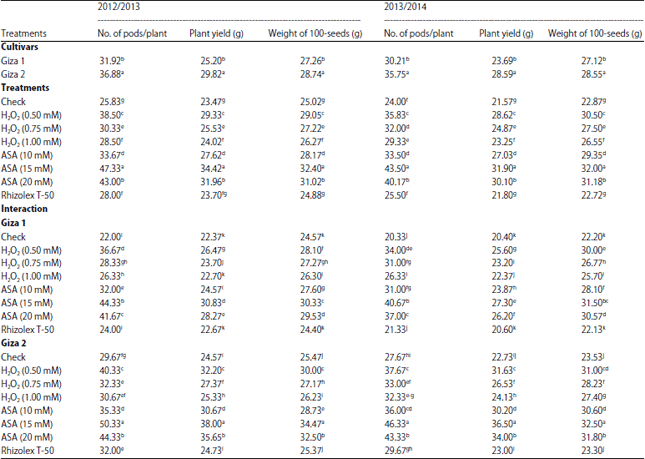
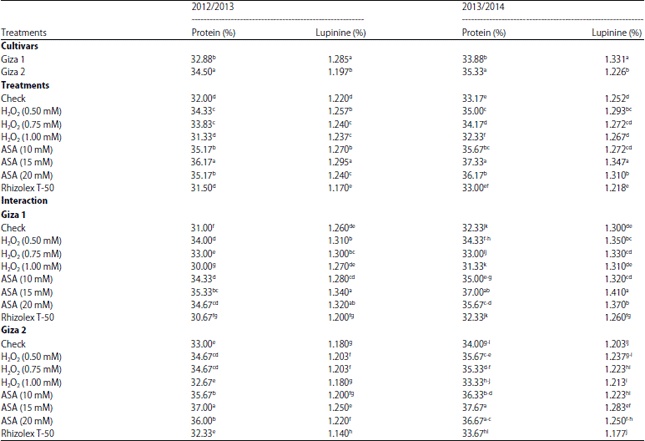
Research Article
Hydrogen Peroxide and Acetylsalicylic Acid Induce the Defense of Lupine Against Root Rot Disease
Plant Pathology Research Institute, Agricultural Research Center, Giza, Egypt
M.A. El-Metwally
Plant Pathology Research Institute, Agricultural Research Center, Giza, Egypt
LiveDNA: 20.359
Naglaa T. Mohamed
Plant Pathology Research Institute, Agricultural Research Center, Giza, Egypt