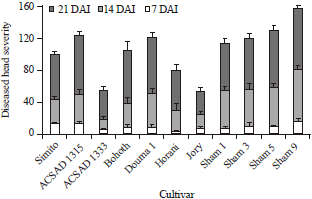
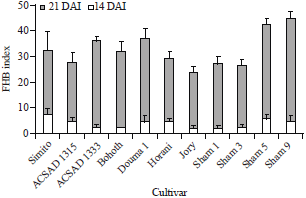
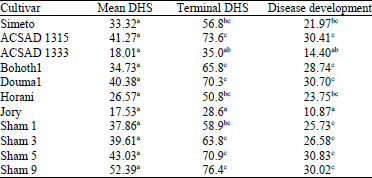
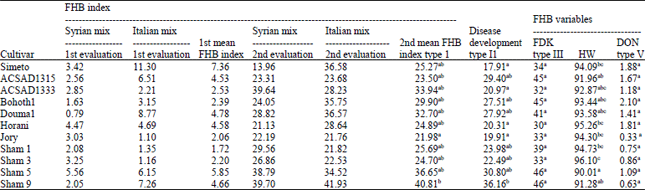
Research Article
Assessment of Different Resistance Types of Syrian Durum Wheat Cultivars Towards FHB Agent
Department of Agricultural Sciences, Alma Mater Studiorum University of Bologna, Viale Giuseppe Fanin 40, 40127, Bologna, Italy
Tonti Stefano
Department of Agricultural Sciences, Alma Mater Studiorum University of Bologna, Viale Giuseppe Fanin 40, 40127, Bologna, Italy
Amato Barbara
Department of Agricultural Sciences, Alma Mater Studiorum University of Bologna, Viale Giuseppe Fanin 40, 40127, Bologna, Italy
Nipoti Paola
Department of Agricultural Sciences, Alma Mater Studiorum University of Bologna, Viale Giuseppe Fanin 40, 40127, Bologna, Italy
Pisi Annamaria
Department of Agricultural Sciences, Alma Mater Studiorum University of Bologna, Viale Giuseppe Fanin 40, 40127, Bologna, Italy
Prodi Antonio
Department of Agricultural Sciences, Alma Mater Studiorum University of Bologna, Viale Giuseppe Fanin 40, 40127, Bologna, Italy