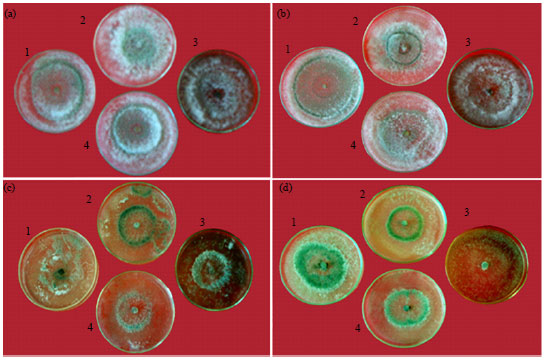
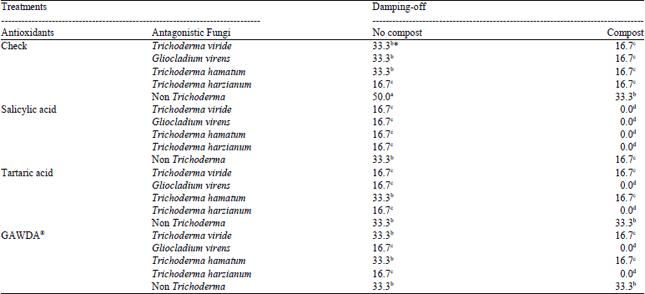
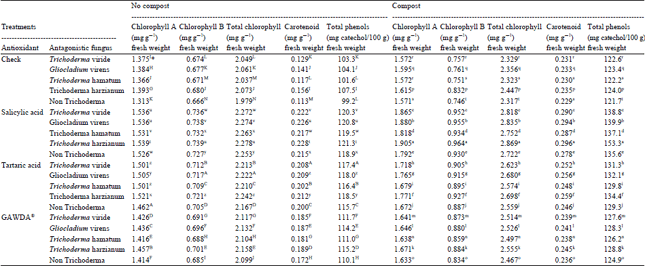
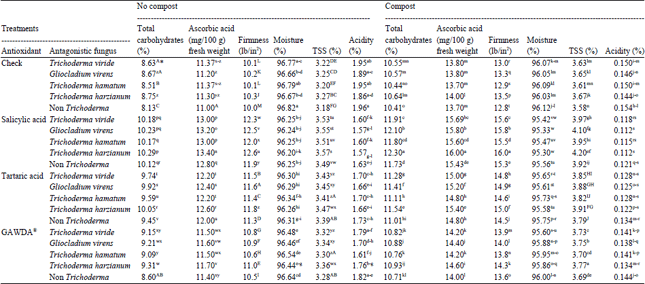
Research Article
Enhancing Systemic Acquired Resistance in Cucumber to Control Root Rot and Wilt Diseases with Reference to Yield and Quality
Department of Plant Pathology, Faculty of Agriculture, Mansoura University, El-Mansoura, 35516, Egypt
M.A. El- Metwally
Plant Pathology Research Institute, Agricultural Research Center, Giza, Egypt
Elsherbiny A. Elsherbiny
Department of Plant Pathology, Faculty of Agriculture, Mansoura University, El-Mansoura, 35516, Egypt
K.N.M. Eisa
Plant Pathology Research Institute, Agricultural Research Center, Giza, Egypt